Abstract
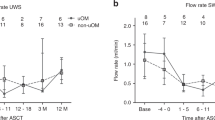
Oral mucositis (OM) is a common side effect experienced during haematopoietic SCT (HSCT), and it can have a significant impact on the quality of life of patients. A descriptive nurse-led study was undertaken in 19-member centres of the Italian national transplant group (GITMO) evaluating incidence, severity and duration of OM in patients undergoing HSCT. Data from 1841 patients between 2002 and 2006 was analyzed. Initial medical history and oral cavity assessment was performed. Assessment was repeated on the day of transplant, then daily, using the WHO (World Health Organisation) oral toxicity scale. A total of 71% of the patients evaluated developed mucositis and 21.6% developed severe mucositis. Duration of OM in most cases lasted for 10–14 days and resolved along with marrow reconstitution. Oral mucostitis is a frequent side effect in patients undergoing HSCT. The onset of severe mucositis seems to be related to the conditioning regimen used. This database provides a descriptive overview of the incidence and severity of mucositis and has encouraged participating centres to adopt routine evaluation and measurement of the oral cavity. The assessment tools are still used in some centres, providing a basis for further collaborative research projects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Sonis ST, Elting LS, Keefe D, Peterson DE, Schubert M, Hauser-Jensen M, et al., for the Mucositis Study Section of the multinational Association of Supportive Care in Cancer—MASCC —and the International Society of Oral Oncology—ISOO. Perspectives on cancer therapy-induced mucosal injury: pathogenesis, measurement, epidemiology, and conseguences or Patients. Cancer 2004; 100: 1995–2025.
Stiff P . Mucositis associated with stem cells transplantation: current status and innovative approaches to management. Bone Marrow Transplant 2001; 27 (suppl 2): S3–S11.
Gabriel DA, Shea T, Olajida O, Serody JS, Comeau T . The effect of oral mucositis on morbility and mortality in bone marrow transplant. Semin Oncol 2003; 30: 76–83.
Rubenstein EB, Peterson DE, Schubert M, Keefe D, McGuire D, Epstein J, et al., for the Mucositis Study Section of the Multinational Association of Supportive Care in Cancer—MASCC —and the International Society of Oral Oncology—ISOO. Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer 2004; 100: 2026–2046.
The Joanna Briggs Institute. Prevention and treatment of oral mucositis in cancer patients. Best Practice 1998; 2: 1–6.
Sonis ST . Mucositis as a biological process: a new hypotesis for the development of chemotherapy-induced stomatotoxicity. Oral Oncol 1998; 34: 39–43.
Sonis ST . Mucositis: the impact, biology and therapeutic opportunities of oral mucositis. Oral Oncol 2009; 45: 1015–1020.
Molinterni A, Zambetti M . Danni da farmaci. In: Bonadonna G, Robustelli Della Cuna G, Valgusa P (eds). Medicina Oncologica, 7th edn. Masson (ed). Milano, 2004, pp 1647–1661.
Salgarello MC . Le evidenze scientifiche nella prevenzione e nel trattamento della mucosite da chemioterapia e radioterapia. Assistenza Infermieristica e Ricerca 2003; 22: 95–106.
Tish Knobf M . Assistenza infermieristica in oncologia medica. In: Bonadonna G, Robustelli Della Cuna G, Valgusa P (eds). Medicina Oncologica, 7th edn. Masson Ed. Milano, 2004, pp 559–579.
Schubert MM, Peterson DE, Lloid ME . Complications and their management—oral complications. In: Blume KG, Forman SJ, Appelbaum FR (eds). Thomas' Haematopoietic Cell Transplantation, 3rd edn. Blackwell Publishing Ltd: Malden, MA, 2004, pp 911–925.
Gori E, Arpinati M, Bonifazi F, Errico A, Mega A, Alberani E et al. Cryotherapy in the prevention of oral mucositis in patients receiving low-dose methotrexate following myeloablative allogenic stem cell transplantation: a prospective randomized study of the Gruppo Italiano Trapianto di Midollo Osseo nurses group. Bone Marrow Transplant 2007; 39: 347–352.
Reuscher TJ, Sodeifi A, Scrivani SJ, Kaban L, Sonis ST . The impact of mucositis of alpha Hemolytic streptococcal infections in patients undergoing autologous bone marrow transplantation for hematologic malignancies. Cancer 1998; 82: 2275–2281.
www.gitmo.net-area riservata infermieristica—studi—in corso—mucosite—Feraut C, Gobetto G, Vagliano L. Database sull'incidenza e la gravità della mucosite orale nel paziente sottoposto a trapianto di cellule staminali emopoietiche.
Shea TC, Brunner R, Wilej JM . An expanded phase I/II trial of cyclophosphamide, etoposide, and carboplatin plus total body irradiation with autologous marrow or stem cell support for patients with hematologic malignancies. Biology Blood Marrow Transplant 2003; 9: 443–452.
Bolwell BJ, Kalaycio M, Sodecks R, Andresen S, Kuczkowski E, Bernhard L et al. A multivariable analysis of factors influencing mucositis after autologous progenitor cell transplantation. Bone Marrow Transplant 2002; 30: 587–591.
Cesaro S, Brugiolo A, Cecchetto G, Badon P . Il bambino affetto da malattie dell'apparato emolinfopoietico. In: Badon P, Cesaro S (eds). Manuale di Nursing Pediatrico, 1st edn. Casa Editrice Ambrosiana: Milano, 2002, pp 679–799.
Filicko J, Lazarus HM, Flomenberg N . Mucosal injury in patients undergoing hematopoietic progenitor cell transplantation: new approaches to prophilaxis and treatment. Bone Marrow Transplant 2003; 31: 1–10.
Woo SB, Sonis ST, Monopoli MM, Sonis AL . A longitudinal study of oral ulcerative mucositis in bone marrow transplant recipients. Cancer 1993; 72: 1612–1617.
Acknowledgements
We would like to thank all those who participated in the study (for data collection and the revision of this article). We would also like to thank EM Mollo Bsc for her precious help with data elaboration.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Vagliano, L., Feraut, C., Gobetto, G. et al. Incidence and severity of oral mucositis in patients undergoing haematopoietic SCT—results of a multicentre study. Bone Marrow Transplant 46, 727–732 (2011). https://doi.org/10.1038/bmt.2010.184
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.184
Keywords
This article is cited by
-
A randomized phase II study of acyclovir for the prevention of chemotherapy-induced oral mucositis in patients undergoing autologous hematopoietic stem cell transplantation
BMC Oral Health (2023)
-
Viable Bifidobacterium tablets for the prevention of chemotherapy-/radiation-induced mucositis in patients undergoing haematopoietic stem cell transplantation
Supportive Care in Cancer (2023)
-
Prospective longitudinal analysis of clinical and immunological risk factors associated with oral and gastrointestinal mucositis following autologous stem cell transplant in adults
Supportive Care in Cancer (2023)
-
Hangeshashinto for prevention of oral mucositis in patients undergoing hematopoietic stem cell transplantation: a randomized phase II study
Supportive Care in Cancer (2023)
-
Efficacy and safety of a colostrum- and Aloe vera-based oral care protocol to prevent and treat severe oral mucositis in patients undergoing hematopoietic stem cell transplantation: a single-arm phase II study
Annals of Hematology (2022)