Abstract
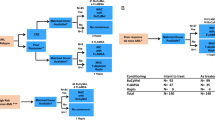
We analyzed the outcome of 243 children with high-risk (HR) AML in first CR1 enrolled in the AIEOP-2002/01 protocol, who were given either allogeneic (ALLO; n=141) or autologous (AUTO; n=102) hematopoietic SCT (HSCT), depending on the availability of a HLA-compatible sibling. Infants, patients with AML-M7, or complex karyotype or those with FLT3-ITD, were eligible to be transplanted also from alternative donors. All patients received a myeloablative regimen combining BU, Cyclophosphamide and Melphalan; AUTO-HSCT patients received BM cells in most cases, while in children given ALLO-HSCT stem cell source was BM in 96, peripheral blood in 19 and cord blood in 26. With a median follow-up of 57 months (range 12–130), the probability of disease-free survival (DFS) was 73% and 63% in patients given either ALLO- or AUTO-HSCT, respectively (P=NS). Although the cumulative incidence (CI) of relapse was lower in ALLO- than in AUTO-HSCT recipients (17% vs 28%, respectively; P=0.043), the CI of TRM was 7% in both groups. Patients transplanted with unrelated donor cord blood had a remarkable 92.3% 8-year DFS probability. Altogether, these data confirm that HSCT is a suitable option for preventing leukemia recurrence in HR children with CR1 AML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
04 February 2015
This article has been corrected since Advance Online Publication and an erratum is also printed in this issue.
References
Pui CH, Carroll WL, Meshinchi S, Arceci RJ . Biology, risk stratification, and therapy of pediatric acute leukemias: an update. J Clin Oncol 2011; 29: 551–565.
Creutzig U, van den Heuvel-Eibrink MM, Gibson B, Dworzak MN, Adachi S, de Bont E et al. Diagnosis and management of acute myeloid leukemia in children and adolescents: recommendations from an international expert panel. Blood 2012; 120: 3187–3205.
Kaspers GJ, Zwaan CM . Pediatric acute myeloid leukemia: towards high-quality cure of all patients. Haematologica 2007; 92: 1519–1532.
Niewerth D, Creutzig U, Bierings MB, Kaspers GJ . A review on allogeneic stem cell transplantation for newly diagnosed pediatric acute myeloid leukemia. Blood 2010; 116: 2205–2214.
Woods WG, Neudorf S, Gold S, Sanders J, Buckley JD, Barnard DR et al. A comparison of allogeneic bone marrow transplantation, autologous bone marrow transplantation, and aggressive chemotherapy in children with acute myeloid leukemia in remission. Blood 2001; 97: 56–62.
Amadori S, Testi AM, Arico M, Comelli A, Giuliano M, Madon E et al. Prospective comparative study of bone marrow transplantation and postremission chemotherapy for childhood acute myelogenous leukemia. The Associazione Italiana Ematologia ed Oncologia Pediatrica Cooperative Group. J Clin Oncol 1993; 11: 1046–1054.
Klusmann JH, Reinhardt D, Zimmermann M, Kremens B, Vormoor J, Dworzak M et al. The role of matched sibling donor allogeneic stem cell transplantation in pediatric high-risk acute myeloid leukemia: results from the AML-BFM 98 study. Haematologica 2012; 97: 21–29.
Horan JT, Alonzo TA, Lyman GH, Gerbing RB, Lange BJ, Ravindranath Y et al. Impact of disease risk on efficacy of matched related bone marrow transplantation for pediatric acute myeloid leukemia: the Children's Oncology Group. J Clin Oncol 2008; 26: 5797–5801.
Ruggeri A, Volt F, Michel G, Diaz de Heredia C, Abecasis M, Zecca M et al. Unrelated cord blood transplantation for infant acute leukemia diagnosed within 1 year of age: outcomes and risk factor analysis on behalf of Eurocord and PDWP-EBMT. Blood 2013; 122: 160-.
Eapen M, Rubinstein P, Zhang MJ, Stevens C, Kurtzberg J, Scaradavou A et al. Outcomes of transplantation of unrelated donor umbilical cord blood and bone marrow in children with acute leukaemia: a comparison study. Lancet 2007; 369: 1947–1954.
Michel G, Rocha V, Chevret S, Arcese W, Chan KW, Filipovich A et al. Unrelated cord blood transplantation for childhood acute myeloid leukemia: a Eurocord Group analysis. Blood 2003; 102: 4290–4297.
Locatelli F, Zecca M, Messina C, Rondelli R, Lanino E, Sacchi N et al. Improvement over time in outcome for children with acute lymphoblastic leukemia in second remission given hematopoietic stem cell transplantation from unrelated donors. Leukemia 2002; 16: 2228–2237.
Dini G, Zecca M, Balduzzi A, Messina C, Masetti R, Fagioli F et al. No difference in outcome between children and adolescents transplanted for acute lymphoblastic leukemia in second remission. Blood 2011; 118: 6683–6690.
Bonetti F, Zecca M, Pession A, Messina C, Montagna D, Lanino E et al. Total-body irradiation and melphalan is a safe and effective conditioning regimen for autologous bone marrow transplantation in children with acute myeloid leukemia in first remission. The Italian Association for Pediatric Hematology and Oncology-Bone Marrow Transplantation Group. J Clin Oncol 1999; 17: 3729–3735.
Zittoun R A, Mandelli F, Willemze R, de Witte T, Labar B, Resegotti L et al. Autologous or allogeneic bone marrow transplantation compared with intensive chemotherapy in acute myelogenous leukemia. European Organization for Research and Treatment of Cancer (EORTC) and the Gruppo Italiano Malattie Ematologiche Maligne dell'Adulto (GIMEMA) Leukemia Cooperative Groups. N Engl J Med 1995; 332: 217–223.
Ravindranath Y, Yeager AM, Chang MN, Steuber CP, Krischer J, Graham-Pole J et al. Autologous bone marrow transplantation versus intensive consolidation chemotherapy for acute myeloid leukemia in childhood. Pediatric Oncology Group. N Engl J Med 1996; 334: 1428–1434.
Pession A, Masetti R, Rizzari C, Putti MC, Casale F, Fagioli F et al. Results of the AIEOP AML 2002/01 multicenter prospective trial for the treatment of children with acute myeloid leukemia. Blood 2013; 122: 170–178.
Locatelli F, Pession A, Bonetti F, Maserati E, Prete L, Pedrazzoli P et al. Busulfan, cyclophosphamide and melphalan as conditioning regimen for bone marrow transplantation in children with myelodysplastic syndromes. Leukemia 1994; 8: 844–849.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 1974; 18: 295–304.
Storb R, Prentice RL, Sullivan KM, Shulman HM, Deeg HJ, Doney KC et al. Predictive factors in chronic graft-versus-host disease in patients with aplastic anemia treated by marrow transplantation from HLA-identical siblings. Ann intern Med 1983; 98: 461–466.
Gooley TA, Leisenring W, Crowley J, Storer BE . Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Zhang X, Zhang MJ, Fine J . A proportional hazards regression model for the subdistribution with right-censored and left-truncated competing risks data. Stat Med 2011; 30: 1933–1951.
Reeves GK, Cox DR, Darby SC, Whitley E . Some aspects of measurement error in explanatory variables for continuous and binary regression models. Stat Med 1998; 17: 2157–2177.
Dignan FL, Wynn RF, Hadzic N, Karani J, Quaglia A, Pagliuca A et al. BCSH/BSBMT guideline: diagnosis and management of veno-occlusive disease (sinusoidal obstruction syndrome) following haematopoietic stem cell transplantation. Brit J Haematol 2013; 163: 444–457.
Kelly MJ, Horan JT, Alonzo TA, Eapen M, Gerbing RB, He W et al. Comparable survival for pediatric acute myeloid leukemia with poor-risk cytogenetics following chemotherapy, matched related donor, or unrelated donor transplantation. Pediatri Blood Cancer 2014; 61: 269–275.
Eapen M, Rubinstein P, Zhang MJ, Camitta BM, Stevens C, Cairo MS et al. Comparable long-term survival after unrelated and HLA-matched sibling donor hematopoietic stem cell transplantations for acute leukemia in children younger than 18 months. J Clin Oncol 2006; 24: 145–151.
Meshinchi S, Arceci RJ, Sanders JE, Smith FO, Woods WB, Radich JP et al. Role of allogeneic stem cell transplantation in FLT3/ITD-positive AML. Blood 2006; 108: 400.
Staffas A, Kanduri M, Hovland R, Rosenquist R, Ommen HB, Abrahamsson J et al. Presence of FLT3-ITD and high BAALC expression are independent prognostic markers in childhood acute myeloid leukemia. Blood 2011; 118: 5905–5913.
Levis M, Small D . FLT3: ITDoes matter in leukemia. Leukemia 2003; 17: 1738–1752.
DeZern A E, Sung A, Kim S, Smith B D, Karp J E, Gore S D et al. Role of allogeneic transplantation for FLT3/ITD acute myeloid leukemia: outcomes from 133 consecutive newly diagnosed patients from a single institution. Biol Blood Marrow Transplant 2011; 17: 1404–1409.
Locatelli F, Nollke P, Zecca M, Korthof E, Lanino E, Peters C et al. Hematopoietic stem cell transplantation (HSCT) in children with juvenile myelomonocytic leukemia (JMML): results of the EWOG-MDS/EBMT trial. Blood 2005; 105: 410–419.
Beier R, Albert MH, Bader P, Borkhardt A, Creutzig U, Eyrich M et al. Allo-SCT using BU, CY and melphalan for children with AML in second CR. Bone Marrow Transplant 2013; 48: 651–656.
Giorgiani G, Bozzola M, Locatelli F, Picco P, Zecca M, Cisternino M et al. Role of busulfan and total body irradiation on growth of prepubertal children receiving bone marrow transplantation and results of treatment with recombinant human growth hormone. Blood 1995; 86: 825–831.
Michel G, Gluckman E, Esperou-Bourdeau H, Reiffers J, Pico JL, Bordigoni P et al. Allogeneic bone marrow transplantation for children with acute myeloblastic leukemia in first complete remission: impact of conditioning regimen without total-body irradiation—a report from the Societe Francaise de Greffe de Moelle. J Clin Oncol 1994; 12: 1217–1222.
Parth P, Dunlap WP, Kennedy RS, Ordy JM, Lane NE . Motor and cognitive testing of bone marrow transplant patients after chemoradiotherapy. Percept Mot Skills 1989; 68: 1227–1241.
Hasle H . A critical review of which children with acute myeloid leukaemia need stem cell procedures. Brit J Haematol 2014; 166: 23–33.
Masetti R, Pigazzi M, Togni M, Astolfi A, Indio V, Manara E et al. CBFA2T3-GLIS2 fusion transcript is a novel common feature in pediatric, cytogenetically normal AML, not restricted to FAB M7 subtype. Blood 2013; 121: 3469–3472.
von Neuhoff C, Reinhardt D, Sander A, Zimmermann M, Bradtke J, Betts DR et al. Prognostic impact of specific chromosomal aberrations in a large group of pediatric patients with acute myeloid leukemia treated uniformly according to trial AML-BFM 98. J Clin Oncol 2010; 28: 2682–2689.
Balgobind BV, Van den Heuvel-Eibrink MM, De Menezes RX, Reinhardt D, Hollink IH, Arentsen-Peters ST et al. Evaluation of gene expression signatures predictive of cytogenetic and molecular subtypes of pediatric acute myeloid leukemia. Haematologica 2011; 96: 221–230.
Rubnitz JE, Inaba H, Dahl G, Ribeiro RC, Bowman WP, Taub J et al. Minimal residual disease-directed therapy for childhood acute myeloid leukaemia: results of the AML02 multicentre trial. Lancet Oncol 2010; 11: 543–552.
Acknowledgements
This study was supported by research grants awarded by Associazione Italiana per la Ricerca sul Cancro (5 x 1000 Special Grant #9962 to FL), by PRIN (Progetti di Rilevante Interesse Nazionale) 2010 to FL and 2012 to SR, by Ospedale Bambino Gesù, Roma, (Progetto di Ricerca Corrente 2012–2013) to AB and FL, and FILAS (Adult Stem Cells) to SR.
Author Contributions
FL designed the study, interpreted data, performed transplantation and wrote the article; RM designed the study, checked data and performed transplantation; RR analyzed data; MZ, FF, AR, CM and EL designed the study, performed transplantation and followed patients; AB, CF, GG, MR, GP, MP and OZ performed transplantation and followed patients; AP designed the study, performed transplantation and interpreted the data; SR interpreted the data and wrote the paper; ArPr contributed to study design, interpreted the data and performed transplantation.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
List of participating centers and investigators
Department of Pediatric Onco-Hematology, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS), Policlinico San Matteo Foundation, Pavia. Franco Locatelli (till January 2010), Marco Zecca, Giovanna Giorgiani [38 HSCT].Department of Pediatric Hematology-Oncology, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS), Bambino Gesù Children's Hospital, Roma. Franco Locatelli (since January 2010), Alice Bertaina, Maurizio Caniglia, Giuseppe Palumbo, Sergio Rutella [30 HSCT]. Department of Pediatric Hematology and Oncology, University of Padova, Padova. Chiara Messina, Marta Pillon [29 HSCT]. Department of Pediatric Hematology, San Gerardo Hospital, Monza. Adriana Balduzzi, Attilio Rovelli [27 HSCT]. BMT Unit, Department of Pediatric Hemato-Oncology, Santobono-Pausilipon Hospital, Napoli. Mimmo Ripaldi [24 HSCT]. Department of Pediatric Hematology Oncology, IRCCS G. Gaslini Institute, Genova. Edoardo Lanino, Giorgio Dini [23 HSCT]. Pediatric Onco-Hematology, Stem Cell Transplantation and Cellular Therapy Division, Regina Margherita Children's Hospital, Torino. Franca Fagioli [15 HSCT]. Pediatric Oncology and Hematology Unit ‘Lalla Seragnoli’, Department of Pediatrics, University of Bologna Sant’Orsola-Malpighi Hospital, Bologna. Riccardo Masetti, Arcangelo Prete, Andrea Pession [12 HSCT]. Department of Pediatrics, University of Pisa, Pisa. Claudio Favre [9 HSCT]. Pediatric Hematology/Oncology, ARNAS Ospedale Civico di Palermo. Ottavio Ziino, Paolo D’Angelo [8 HSCT]. Pediatric Hematology/Oncology, University Hospital, Catania. Luca Lo Nigro [8 HSCT]. BMT Unit, Ospedale di Pescara. Paolo Di Bartolomeo [6 HSCT]. BMT Unit, Ospedale Pediatrico Burlo Garofalo, Trieste. Marco Rabusin [6 HSCT]. BMT Unit, Department of Pediatric Hematology/Oncology, Ospedale Pediatrico Meier, Florence. Desiree Caselli [5 HSCT]. BMT Unit, Pediatric Hematology/Oncology, Ospedale Silvestrini, Perugia. Franco Aversa [3 HSCT].
Rights and permissions
About this article
Cite this article
Locatelli, F., Masetti, R., Rondelli, R. et al. Outcome of children with high-risk acute myeloid leukemia given autologous or allogeneic hematopoietic cell transplantation in the aieop AML-2002/01 study.. Bone Marrow Transplant 50, 181–188 (2015). https://doi.org/10.1038/bmt.2014.246
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.246
This article is cited by
-
Pediatric chronic graft-versus-host disease-related dry eye disease and the diagnostic association of potential clinical findings
Scientific Reports (2023)
-
Response to: meta-analysis on allogeneic transplant for treating pediatric patients with acute myeloid leukemia in first remission: reanalysis of primary data
Annals of Hematology (2023)
-
Allogeneic hematopoietic stem cell transplantation for pediatric acute myeloid leukemia in first complete remission: a meta-analysis
Annals of Hematology (2022)
-
Acute Leukemia in Infants
Current Oncology Reports (2021)
-
Hematopoietic stem cell transplantation for children with acute myeloid leukemia—results of the AML SCT-BFM 2007 trial
Leukemia (2020)