Abstract
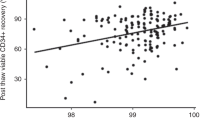
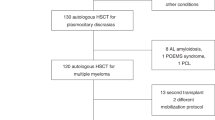
Hematopoietic precursor cells (HPC) are able to restore hematopoiesis after high-dose chemotherapy and their cryopreservation is routinely employed prior to the autologous hematopoietic cell transplantation (AHCT). Although previous studies showed feasibility of long-term HPC storage, concerns remain about possible negative effects on their potency. To study the effects of long-term cryopreservation, we compared time to neutrophil and platelet recovery in 50 patients receiving two AHCT for multiple myeloma at least 2 years apart between 2006 and 2016, using HPC obtained from one mobilization and collection attempt before the first transplant. This product was divided into equivalent fractions allowing a minimum of 2 × 106 CD34+ cells/kg recipient’s weight. One fraction was used for the first transplant after median storage of 60 days (range, 17–165) and another fraction was used after median storage of 1448 days (range, 849–3510) at the second AHCT. Neutrophil recovery occurred at 14 days (median; range, 11–21) after the first and 13 days (10–20) after the second AHCT. Platelets recovered at a median of 16 days after both procedures. Considering other factors, such as disease status, conditioning and HPC dose, this single institution data demonstrated no reduction in the potency of HPC after long-term storage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Park B, Yoo KH, Kim C . Hematopoietic stem cell expansion and generation: the ways to make a breakthrough. Blood Res 2015; 50: 194–203.
Cook G, Liakopoulou E, Pearce R, Cavet J, Morgan GJ, Kirkland K et al. Factors influencing the outcome of a second autologous stem cell transplant (ASCT) in relapsed multiple myeloma: a study from the British Society of Blood and Marrow Transplantation Registry. Biol Blood Marrow Transplant 2011; 17: 1638–1645.
FACT-JACIE International Standards for hematopoietic cellular therapy product collection, processing, and administration. http://www.jacie.org/standards/6th-edition-2015.
Palumbo A, Cavallo F, Gay F, Di Raimondo F, Ben Yehuda D, Petrucci MT et al. Autologous transplantation and maintenance therapy in multiple myeloma. N Engl J Med 2014; 371: 895–905.
Moreau P, Attal M, Facon T . Frontline therapy of multiple myeloma. Blood 2015; 125: 3076–3084.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med 2003; 348: 1875–1883.
Morgenstern DA, Ahsan G, Brocklesby M, Ings S, Balsa C, Veys P et al. Post-thaw viability of cryopreserved peripheral blood stem cells (PBSC) does not guarantee functional activity: important implications for quality assurance of stem cell transplant programmes. Br J Haematol 2016; 174: 942–951.
Aird W, Labopin M, Gorin NC, Antin JH . Long-term cryopreservation of human stem cells. Bone Marrow Transplant 1992; 9: 487–490.
Berz D, McCormack EM, Winer ES, Colvin GA, Quesenberry PJ . Cryopreservation of hematopoietic stem cells. Am J Hematol 2007; 82: 463–472.
Acknowledgements
This research was not directly funded. All authors are supported by the National Institute for Health Research Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London. Authors are grateful to all the clinical, laboratory and administrative staff, and also to all the patients.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Pavlů, J., Auner, H., Szydlo, R. et al. Analysis of hematopoietic recovery after autologous transplantation as method of quality control for long-term progenitor cell cryopreservation. Bone Marrow Transplant 52, 1599–1601 (2017). https://doi.org/10.1038/bmt.2017.113
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2017.113