Abstract
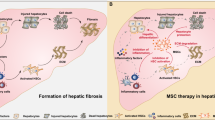
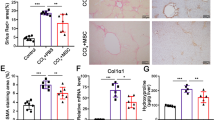
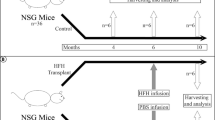
Transplantation of mesenchymal stem cells (MSCs) has been recently studied in animal models, and in clinical trials of patients with fulminant hepatic failure, end-stage liver diseases and inherited metabolic disorders. Modulatory cytokines produced by MSCs can inhibit immunocyte proliferation and migration to the liver, thereby attenuating inflammatory injury and reducing hepatocyte apoptosis. In addition, MSCs play an important role in regressing liver fibrosis and in supporting the function, proliferation and differentiation of endogenous hepatocytes under appropriate conditions. Although remarkable progress has been achieved in basic and clinical MSC studies, optimal therapeutic regimens for the clinical application of MSCs, such as optimal doses, transplantation routine and interval period for transplantation, need to be elucidated in detail. Furthermore, the long-term safety and therapeutic efficacy of MSC transplantation should be evaluated in future clinical trials. This review summarizes our current understanding of the immunomodulatory effects of MSC therapies on human liver diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR et al. Pluripotency of mesenchymal stem cells drived from adult marrow. Nature 2002; 418: 41–49.
Deng X, Chen YX, Zhang X, Zhang JP, Yin C, Yue HY et al. Hepatic stellate cells modulate the differentiation of bone marrow mesenchymal stem cells into hepatocyte-like cells. J Cell Physiol 2008; 217: 138–144.
Okuyama H, Krishnamachary B, Zhou YF, Nagasawa H, Bosch-Marce M, Semenza GL . Expression of vascular endothelial growth factor receptor 1 in bone marrow-derived mesenchymal cells is dependent on hypoxia-inducible factor. J Biol Chem 2006; 281: 15554–15563.
Aurich H, Sgodda M, Kaltwasser P, Vetter M, Weise A, Liehr T et al. Hepatocyte differentiation of mesenchymal stem cells from human adipose tissue in vitro promotes hepatic integration in vivo. Gut 2009; 58: 570–581.
Bonora-Centelles A, Jover R, Mirabet V, Lahoz A, Bonora-Centelles A, Carbonell F et al. Sequential hepatogenic trans-differentiation of adipose tissue-derived stem cells: relevance of different extracellular signaling molecules, transcription factors involved and expression of new key marker genes. Cell Transplant 2009; 18: 1319–1340.
Campard D, Lysy PA, Najimi M, Sokal EM . Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology 2008; 134: 833–848.
Lange C, Bassler P, Lioznov MV, Bruns H, Kluth D, Zander AR et al. Liver-specific gene expression in mesenchymal stem cells is induced by liver cells. World J Gastroenterol 2005; 11: 4497–4504.
van Poll D, Parekkadan B, Cho CH, Berthiaume F, Nahmias Y, Tilles AW et al. Mesenchymal stem cell-derived molecules directly modulate hepatocellular death and regeneration in vitro and in vivo. Hepatology 2008; 47: 1634–1643.
Zhou P, Hohm S, Olusanya Y, Hess DA, Nolta J . Human progenitor cells with high aldehyde dehydrogenase activity efficiently engraft into damaged liver in a novel model. Hepatology 2009; 49: 1992–2000.
Ortiz LA, Dutreil M, Fattman C, Pandey AC, Torres G, Go K et al. Interleukin 1 receptor antagonist mediates the antiinflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. Proc Natl Acad Sci USA 2007; 104: 11002–11007.
Hematti P . Role of mesenchymal stromal cells in solid organ transplantation. Transplant Rev 2008; 22: 262–273.
Parekkadan B, van Poll D, Suganuma K, Carter EA, Berthiaume F, Tilles AW et al. Mesenchymal stem cell-derived molecules reverse fulminant hepatic failure. PLoS One 2007; 2: e941.
Kiss J, Urbán VS, Dudics V, Vas V, Uher F . Mesenchymal stem cells and the immune system–immunosuppression without drugs? Orv Hetil 2008; 149: 339–346.
Yagi K, Kojima M, Oyagi S, Ikeda E, Hirose M, Isoda K et al. Application of mesenchymal stem cells to liver regenerative medicine. Yakugaku Zasshi 2008; 128: 3–9.
Banas A, Teratani T, Yamamoto Y, Tokuhara M, Takeshita F, Osaki M et al. IFATS collection: in vivo therapeutic potential of human adipose tissue mesenchymal stem cells after transplantation into mice with liver injury. Stem Cells 2008; 26: 2705–2712.
Mohamadnejad M, Alimoghaddam K, Mohyeddin-Bonab M, Mohyeddin-Bonab M, Bagheri M, Bashtar M et al. Phase I trial of autologous bone marrow mesenchymal stem cell transplantation in patients with decompensated cirrhosis. Arch Iran Med 2007; 10: 459–466.
Tsai PC, Fu TW, Chen YM, Ko TL, Chen TH, Shih YH et al. The therapeutic potential of human umbilical mesenchymal stem cells from Wharton's jelly in the treatment of rat liver fibrosis. Liver Transpl 2009; 15: 484–495.
Higashiyama R, Inagaki Y, Hong YY, Kushida M, Nakao S, Niioka M et al. Bone marrow-derived cells express matrix metalloproteinases and contribute to regression of liver fibrosis in mice. Hepatology 2007; 45: 213–222.
Parekkadan B, van Poll D, Megeed Z, Kobayashi N, Tilles AW, Berthiaume F et al. Immunomodulation of activated hepatic stellate cells by mesenchymal stem cells. Biochem. Biophys Res Commun 2007; 363: 247–252.
Carvalho AB, Quintanilha LF, Dias JV, Paredes BD, Mannheimer EG, Carvalho FG et al. Bone marrow multipotent mesenchymal stromal cells do not reduce fibrosis or improve function in a rat model of severe chronic liver injury. Stem Cells 2008; 26: 1307–1314.
di Bonzo LV, Ferrero I, Cravanzola C, Mareschi K, Rustichell D, Novo E et al. Human MSCs as a two-edged sword in hepatic regenerative medicine-engraftment and hepatocytes differentiation versus profibrogenic potential. Gut 2008; 57: 223–231.
Agnieszka B, Takumi T, Yusuke Y, Tokuhara M, Takeshita F, Osaki M et al. Rapid hepatic fate specification of adipose-derived stem cells and their therapeutic potential for liver failure. J Gastroenterol Hepatol 2009; 24: 70–77.
Kharaziha P, Hellström PM, Noorinayer B, Farzaneh F, Aghajani K, Jafari F et al. Improvement of liver function in liver cirrhosis patients after autologous mesenchymal stem cell injection: a phase I–II clinical trial. Eur J Gastroenterol Hepatol 2009; 21: 1199–1205.
Kuo TK, Hung SP, Chuang CH, Chen CT, Shih YR, Fang SC et al. Stem cell therapy for liver disease: parameters governing the success of using bone marrow mesenchymal stem cells. Gastroenterology 2008; 134: 2111–2121.
Yagi H, Parekkadan B, Suganuma K, Soto-Gutierrez A, Tompkins R, Tilles A et al. Long term superior performance of a stem cell/hepatocyte device for the treatment of acute liver failure. Tissue Eng Part A 2009; 15: 3377–3388.
Khan AA, Parveen N, Mahaboob VS, Rajendraprasad A, Ravindraprakash HR, Venkateswarlu J et al. Treatment of Crigler–Najjar Syndrome type 1 by hepatic progenitor cell transplantation: a simple procedure for management of hyperbilirubinemia. Transplant Proc 2008; 40: 1148–1150.
Frossard V, Ketterer N, Rosselet A, Meier P, Cairoli A, Duchosal MA et al. Early intensification and autologous stem cell transplantation in patients with systemic AL amyloidosis: a single-centre experience. Ann Hematol 2009; 88: 681–685.
Miranda PS, Bosma PJ . Towards liver-directed gene therapy for Crigler–Najjar syndrome. Curr Gene Ther 2009; 9: 72–82.
Rubio D, Garcia-Castro J, Martín MC, de la Fuente R, Cigudosa JC, Lloyd AC et al. Spontaneous human adult stem cell transformation. Cancer Res 2005; 65: 3035–3039.
McBride C, Gaupp D, Phinney DG . Quantifying levels of transplanted murine and human mesenchymal stem cells in vivo by real-time PCR. Cytotherapy 2003; 5: 7–18.
Takeda M, Yamamoto M, Isoda K, Higashiyama S, Hirose M, Ohgushi H et al. Availability of bone marrow stromal cells in three-dimensional coculture with hepatocytes and transplantation into liver-damaged mice. J Biosci Bioeng 2005; 100: 77–81.
Schrepfer S, Deuse T, Reichenspurner H, Fischbein MP, Robbins RC, Pelletier MP . Stem cell transplantation: the lung barrier. Transplant Proc 2007; 39: 573–576.
Mohamadnejad M, Namiri M, Bagheri M, Hashemi SM, Ghanaati H, Zare Mehrjardi N et al. Phase 1 human trial of autologous bone marrow-hematopoietic stem cell transplantation in patients with decompensated cirrhosis. World J Gastroenterol 2007; 13: 3359–3363.
Xiang GA, Zhang GQ, Fang CH, Gao P, Chen KY . A preliminary study of the homing capacity of allograft mesenchymal stem cells to rat liver. Di Yi Jun Yi Da Xue Xue Bao 2005; 25: 994–997.
Hashemi S, Ghods S, Kolodgie FD, Parcham-Azad K, Keane M, Hamamdzic D et al. A placebo controlled, dose-ranging, safety study of allogenic mesenchymal stem cells injected by endomyocardial delivery after an acute myocardial infarction. Eur Heart J 2008; 29: 251–259.
Kuhn NZ, Tuan RS . Regulation of stemness and stem cell niche of mesenchymal stem cells: implications in tumorigenesis and metastasis. J Cell Physiol 2010; 222: 268–277.
Lazennec G, Jorgensen C . Concise review: adult multipotent stromal cells and cancer: risk or benefit? Stem cells 2008; 26: 1387–1394.
Ren G, Su J, Zhang L, Zhao X, Ling W, L'huillie A et al. Species variation in the mechanisms of mesenchymal stem cell-mediated immunosuppression. Stem Cells 2009; 27: 1954–1962.
Bernardo ME, Locatelli F, Fibbe WE . Mesenchymal stromal cells. Ann NY Acad Sci 2009; 1176: 101–117.
Sundin M, Orvell C, Rasmusson I, Sundberg B, Ringdén O, Le Blanc K . Mesenchymal stem cells are susceptible to human herpesviruses, but viral DNA cannot be detected in the healthy seropositive individual. Bone Marrow Transplant 2006; 37: 1051–1059.
Acknowledgements
This work was supported by grants from the National Grand Program on Key Infectious Disease (No. 2009ZX10004-309, No. 2008ZX10002-007 and No. 2008ZX10002-005-6) and the National Key Basic Research Program of China (No. 2007CB512805 and No. 2007CB512804).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, H., Xu, R., Zhang, Z. et al. Implications of the immunoregulatory functions of mesenchymal stem cells in the treatment of human liver diseases. Cell Mol Immunol 8, 19–22 (2011). https://doi.org/10.1038/cmi.2010.57
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2010.57
Keywords
This article is cited by
-
Mesenchymal stromal/stem cells and their extracellular vesicles in liver diseases: insights on their immunomodulatory roles and clinical applications
Cell & Bioscience (2023)
-
Multiple Dimensions of using Mesenchymal Stem Cells for Treating Liver Diseases: From Bench to Beside
Stem Cell Reviews and Reports (2023)
-
Prednisolone and mesenchymal stem cell preloading protect liver cell migration and mitigate extracellular matrix modification in transplanted decellularized rat liver
Stem Cell Research & Therapy (2022)
-
The impact of recipient age on the effects of umbilical cord mesenchymal stem cells on HBV-related acute-on-chronic liver failure and liver cirrhosis
Stem Cell Research & Therapy (2021)
-
In vitro efficacy of liver microenvironment in bone marrow mesenchymal stem cell differentiation
In Vitro Cellular & Developmental Biology - Animal (2020)