Abstract
Background/Objectives:
To investigate the feasibility of Fabuless (previously called Olibra and Reducal) as a food ingredient for food intake and appetite reduction, by assessing the effects of food processing on efficacy.
Subjects/Methods:
In total, 24 healthy volunteers (16 female, 8 male; age: 18–43 years; body mass index: 18–37 kg/m2) took part in a randomized, placebo-controlled, double-blinded, cross-over trial. Yoghurt-based meal replacement drinks (containing processed or unprocessed Fabuless, or a control fat) were followed by an ad libitum lunch and evening meal (dinner). Key outcome measures were energy intake and self-reported appetite ratings.
Results:
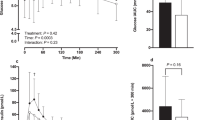
Compared with control, only unprocessed Fabuless reduced subsequent energy intake, although only during dinner (P<0.01; control, processed and unprocessed: 4.3, 3.9 and 4.2 MJ, respectively) and not during lunch (3.6, 3.7 and 3.6 MJ). Self-reported appetite scores did not differ between treatments.
Conclusions:
Although modest effects of unprocessed Fabuless were seen on food intake, but not on appetite, the ingredient was not robust to common food-manufacturing processes (thermal and shear processing). Claims on reduced food intake and appetite relating to this ingredient in food products are, therefore, only valid if functionality has been demonstrated after all relevant processing and storage steps.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Brubaker PL, Drucker DJ, Asa SL, Swallow C, Redston M, Greenberg GR (2002). Prolonged gastrointestinal transit in a patient with a glucagon-like peptide (GLP)-1- and -2-producing neuroendocrine tumor. J Clin Endocrinol Metab 87, 3078–3083.
Burns AA, Livingstone MBE, Welch RW, Dunne A, Reid CA, Rowland IR (2001). The effects of yoghurt containing a novel fat emulsion on energy and macronutrient intakes in non-overweight, overweight and obese subjects. Int J Obes 25, 1487–1496.
Burns AA, Livingstone MBE, Welch RW, Dunne A, Robson PJ, Lindmark L et al. (2000). Short-term effects of yoghurt containing a novel fat emulsion on energy and macronutrient intakes in non-obese subjects. Int J Obes 24, 1419–1425.
Burns AA, Livingstone MBE, Welch RW, Dunne A, Rowland IR (2002). Dose-response effects of a novel fat emulsion (Olibra) on energy and macronutrient intakes up to 36 h post-consumption. Eur J Clin Nutr 56, 368–377.
Delgado-Aros S, Kim D-Y, Burton DD, Thomforde GM, Stephens D, Brinkmann BH et al. (2002). Effect of GLP-1 on gastric volume, emptying, maximum volume ingested, and postprandial symptoms in humans. Am J Physiol 282, G424–G431.
Diepvens K, Steijns J, Zuurendonk P, Westerterp-Plantinga MS (2008). Short-term effects of a novel fat emulsion on appetite and food intake. Physiol Behav 95, 114–117.
Flint A, Raben A, Blundell JE, Astrup A (2000). Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int J Obes 24, 38–48.
Giralt M, Vergara P (1999). Glucagonlike peptide-1 (GLP-1) participation in ileal brake induced by intraluminal peptones in rat. Dig Dis Sci 44, 322–329.
Gregersen NT, Flint A, Bitz A, Blundell JE, Raben A, Astrup A (2008). Reproducibility and power of ad libitum energy intake assessed by repeated single meals. Am J Clin Nutr 87, 1277–1281.
Haenni A, Sundberg B, Yazdanpanah N, Viberg A, Olsson J (2009). Effect of fat emulsion (Fabuless) on orocecal transit time in healthy men. Scand J Gastroenterol 44, 1186–1190.
Lin HC, Zhao X-T, Wang L (1996). Fat absorption is not complete by midgut but is dependent on load of fat. Am J Physiol 271, G62–G67.
Logan CM, McCaffrey TA, Wallace JMW, Robson PJ, Welch RW, Dunne A et al. (2006). Investigation of the medium-term effects of Olibra fat emulsion on food intake in non-obese subjects. Eur J Clin Nutr 60, 1081–1091.
Maljaars PWJ, Peters HPF, Mela DJ, Masclee AAM (2008). Ileal brake: a sensible food target for appetite control. A review. Physiol Behav 95, 271–281.
McClements DJ (2005). Food Emulsions: Principles, Practice and Techniques, 2nd edn. CRC Press: Boca Raton.
Näslund E, Bogefors J, Skogar S, Grybäck P, Jacobsson H, Holst JJ et al. (1999). GLP-1 slows solid gastric emptying and inhibits insulin, glucagon, and PYY release in humans. Am J Physiol 277, R910–R916.
Peh KK, Yuen KH (1996). Indirect gastrointestinal transit monitoring and absorption of theophylline. Int J Pharm 139, 95–103.
Rogers PJ, Blundell JE (1979). Effect of anorexic drugs on food intake and the micro-strucure of eating in human subjects. Psychopharmacology (Berl) 66, 159–165.
Rolls BJ, Shide DJ, Thorwart ML, Ulbrecht JS (1998). Sibutramine reduces food intake in non-dieting women with obesity. Obes Res 6, 1–11.
Staniforth DH (1989). Comparison of orocecal transit times assessed by the lactulose breath hydrogen and the sulfasalazine-sulfapyridine methods. Gut 30, 978–982.
Symersky T, Kee BC, Haddeman E, Peters HP, Masclee AM (2004). Ileal delivery of 3 grams of oil increases satiety, decreases meal intake and increases CCK but not PYY in humans. Obesity Res 12 (Suppl S), A3.
van Strien T, Frijters JER, Bergers GPA, Defares PB (1986a). The Dutch eating behaviour questionnaire for assessment of restrained, emotional and external eating behaviour. Int J Eat Disord 5, 295–315.
van Strien T, Frijters JER, Bergers GPA, Defares PB (1986b). Nederlandse Vragenlijst voor Eetgedrag (NVE), handleiding (Dutch Eating Behavior Questionnaire (DEBQ), manual). Swets & Zeitlinger b.v., Lisse.
Acknowledgements
This work was funded by Unilever R&D, Vlaardingen, The Netherlands.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Smit, Professor Rogers and Mrs Keenan's work was funded by Unilever, Vlaardingen, The Netherlands. They declare no conflict of interest. Dr Mela, Dr Wiseman, Dr Kovacs and Dr Peters represent the funding company.
Rights and permissions
About this article
Cite this article
Smit, H., Keenan, E., Kovacs, E. et al. No efficacy of processed Fabuless (Olibra) in suppressing appetite or food intake. Eur J Clin Nutr 65, 81–86 (2011). https://doi.org/10.1038/ejcn.2010.187
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2010.187
Keywords
This article is cited by
-
Effects of lipid emulsion particle size on satiety and energy intake: a randomised cross-over trial
European Journal of Clinical Nutrition (2018)
-
No appetite efficacy of a commercial structured lipid emulsion in minimally processed drinks
International Journal of Obesity (2012)