Abstract
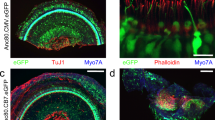
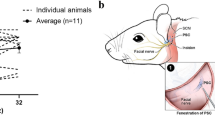
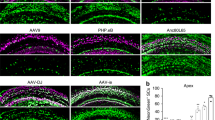
Balance dysfunction is closely associated with loss of vestibular hair cells (HCs). Gene therapy shows promise when used to protect or regenerate vestibular HCs to preserve or restore adequate vestibular function. Adeno-associated virus (AAV) vectors allow long-term gene expression in the absence of toxicity. To noninvasively define an AAV serotype exhibiting favorable tropism toward the vestibular sensory epithelium, we characterized the transgene expression potential of AAV vectors (serotypes 1, 2, 5, 6 and 8) inoculated into adult mouse utricle via canalostomy. We found that AAV8 was the most effective AAV vector in utricular gene transfer. Swim tests and measurements of auditory brainstem response revealed minimal loss of vestibular function and hearing after canalostomy. In the normal utricle after AAV8 infusion, transduction efficiency peaked at 7 days, and was maintained thereafter, in vestibular HCs, and at 3 days in supporting cells (SCs). In the streptomycin-lesioned utricle, the SC transduction efficiency peaked at 7 days and decreased at 30 days. In conclusion, AAV8-mediated gene transfer via canalostomy facilitates efficient and safe transduction in mouse vestibular sensory epithelium, and may in the future become clinically relevant for human vestibular gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Tsuji K, Velazquez-Villasenor L, Rauch SD, Glynn RJ, Wall 3rd C, Merchant SN . Temporal bone studies of the human peripheral vestibular system. Meniere's disease. Ann Otol Rhinol Laryngol Suppl 2000; 181: 26–31.
Rauch SD, Velazquez-Villasenor L, Dimitri PS, Merchant SN . Decreasing hair cell counts in aging humans. Ann NY Acad Sci 2001; 942: 220–227.
Tsuji K, Velazquez-Villasenor L, Rauch SD, Glynn RJ, Wall 3rd C, Merchant SN . Temporal bone studies of the human peripheral vestibular system. Aminoglycoside ototoxicity. Ann Otol Rhinol Laryngol Suppl 2000; 181: 20–25.
Forge A, Li L, Corwin JT, Nevill G . Ultrastructural evidence for hair cell regeneration in the mammalian inner ear. Science 1993; 259: 1616–1619.
Warchol ME, Lambert PR, Goldstein BJ, Forge A, Corwin JT . Regenerative proliferation in inner ear sensory epithelia from adult guinea pigs and humans. Science 1993; 259: 1619–1622.
Kawamoto K, Izumikawa M, Beyer LA, Atkin GM, Raphael Y . Spontaneous hair cell regeneration in the mouse utricle following gentamicin ototoxicity. Hear Res 2009; 247: 17–26.
Wang GP, Chatterjee I, Batts SA, Wong HT, Gong TW, Gong SS et al. Notch signaling and Atoh1 expression during hair cell regeneration in the mouse utricle. Hear Res 2010; 267: 61–70.
Rubel EW, Dew LA, Roberson DW . Mammalian vestibular hair cell regeneration. Science 1995; 267: 701–707.
Shou J, Zheng JL, Gao WQ . Robust generation of new hair cells in the mature mammalian inner ear by adenoviral expression of Hath1. Mol Cell Neurosci 2003; 23: 169–179.
Staecker H, Praetorius M, Baker K, Brough DE . Vestibular hair cell regeneration and restoration of balance function induced by math1 gene transfer. Otol Neurotol 2007; 28: 223–231.
Schlecker C, Praetorius M, Brough DE, Presler Jr RG, Hsu C, Plinkert PK et al. Selective atonal gene delivery improves balance function in a mouse model of vestibular disease. Gene Therapy 2011; 18: 884–890.
Kesser BW, Hashisaki GT, Fletcher K, Eppard H, Holt JR . An in vitro model system to study gene therapy in the human inner ear. Gene Therapy 2007; 14: 1121–1131.
Izumikawa M, Minoda R, Kawamoto K, Abrashkin KA, Swiderski DL, Dolan DF et al. Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat Med 2005; 11: 271–276.
Akil O, Seal RP, Burke K, Wang C, Alemi A, During M et al. Restoration of hearing in the VGLUT3 knockout mouse using virally mediated gene therapy. Neuron 2012; 75: 283–293.
Suzuki M, Yagi M, Brown JN, Miller AL, Miller JM, Raphael Y . Effect of transgenic GDNF expression on gentamicin-induced cochlear and vestibular toxicity. Gene Therapy 2000; 7: 1046–1054.
Pfannenstiel SC, Praetorius M, Plinkert PK, Brough DE, Staecker H . Bcl-2 gene therapy prevents aminoglycoside-induced degeneration of auditory and vestibular hair cells. Audiol Neurootol 2009; 14: 254–266.
Ishimoto S, Kawamoto K, Stover T, Kanzaki S, Yamasoba T, Raphael Y . A glucocorticoid reduces adverse effects of adenovirus vectors in the cochlea. Audiol Neurootol 2003; 8: 70–79.
Liu Y, Okada T, Sheykholeslami K, Shimazaki K, Nomoto T, Muramatsu S et al. Specific and efficient transduction of Cochlear inner hair cells with recombinant adeno-associated virus type 3 vector. Mol Ther 2005; 12: 725–733.
Ballana E, Wang J, Venail F, Estivill X, Puel JL, Arbones ML et al. Efficient and specific transduction of cochlear supporting cells by adeno-associated virus serotype 5. Neurosci Lett 2008; 442: 134–139.
Kilpatrick LA, Li Q, Yang J, Goddard JC, Fekete DM, Lang H . Adeno-associated virus-mediated gene delivery into the scala media of the normal and deafened adult mouse ear. Gene Therapy 2011; 18: 569–578.
Ishimoto S, Kawamoto K, Kanzaki S, Raphael Y . Gene transfer into supporting cells of the organ of Corti. Hear Res 2002; 173: 187–197.
Kawamoto K, Oh SH, Kanzaki S, Brown N, Raphael Y . The functional and structural outcome of inner ear gene transfer via the vestibular and cochlear fluids in mice. Mol Ther 2001; 4: 575–585.
Okada H, Iizuka T, Mochizuki H, Nihira T, Kamiya K, Inoshita A et al. Gene transfer targeting mouse vestibule using adenovirus and adeno-associated virus vectors. Otol Neurotol 2012; 33: 655–659.
Beyea JA, Agrawal SK, Parnes LS . Transmastoid semicircular canal occlusion: a safe and highly effective treatment for benign paroxysmal positional vertigo and superior canal dehiscence. Laryngoscope 2012; 122: 1862–1866.
Hardisty-Hughes RE, Parker A, Brown SD . A hearing and vestibular phenotyping pipeline to identify mouse mutants with hearing impairment. Nat Protoc 2010; 5: 177–190.
Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM . Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci USA 2002; 99: 11854–11859.
Nakai H, Fuess S, Storm TA, Muramatsu S, Nara Y, Kay MA . Unrestricted hepatocyte transduction with adeno-associated virus serotype 8 vectors in mice. J Virol 2005; 79: 214–224.
Wang Z, Zhu T, Qiao C, Zhou L, Wang B, Zhang J et al. Adeno-associated virus serotype 8 efficiently delivers genes to muscle and heart. Nat Biotechnol 2005; 23: 321–328.
Bedrosian JC, Gratton MA, Brigande JV, Tang W, Landau J, Bennett J . In vivo delivery of recombinant viruses to the fetal murine cochlea: transduction characteristics and long-term effects on auditory function. Mol Ther 2006; 14: 328–335.
Blacklow NR, Hoggan MD, Rowe WP . Serologic evidence for human infection with adenovirus-associated viruses. J Natl Cancer Inst 1968; 40: 319–327.
Erles K, Sebokova P, Schlehofer JR . Update on the prevalence of serum antibodies (IgG and IgM) to adeno-associated virus (AAV). J Med Virol 1999; 59: 406–411.
Akache B, Grimm D, Pandey K, Yant SR, Xu H, Kay MA . The 37/67-kilodalton laminin receptor is a receptor for adeno-associated virus serotypes 8, 2, 3, and 9. J Virol 2006; 80: 9831–9836.
Sabatino DE, Mingozzi F, Hui DJ, Chen H, Colosi P, Ertl HC et al. Identification of mouse AAV capsid-specific CD8+ T cell epitopes. Mol Ther 2005; 12: 1023–1033.
Stone IM, Lurie DI, Kelley MW, Poulsen DJ . Adeno-associated virus-mediated gene transfer to hair cells and support cells of the murine cochlea. Mol Ther 2005; 11: 843–848.
Boeda B, Weil D, Petit C . A specific promoter of the sensory cells of the inner ear defined by transgenesis. Hum Mol Genet 2001; 10: 1581–1589.
Liu Y, Okada T, Nomoto T, Ke X, Kume A, Ozawa K et al. Promoter effects of adeno-associated viral vector for transgene expression in the cochlea in vivo. Exp Mol Med 2007; 39: 170–175.
Okada T, Nomoto T, Shimazaki K, Lijun W, Lu Y, Matsushita T et al. Adeno-associated virus vectors for gene transfer to the brain. Methods 2002; 28: 237–247.
Golub JS, Tong L, Ngyuen TB, Hume CR, Palmiter RD, Rubel EW et al. Hair cell replacement in adult mouse utricles after targeted ablation of hair cells with diphtheria toxin. J Neurosci 2012; 32: 15093–15105.
Acknowledgements
This work was supported by the Major State Basic Research Development Program of China (973 Program) (No. 2011CB504503), the National Natural Science Foundation of China (No. 81100717, 81271089 and 81300831), the Technology Development Project of the Beijing Municipal Education Commission (No. KZ201210025026) and the National Science & Technology Pillar Program during the twelfth five-year Plan Period of China (No. 2012BA112B05). We thank Dr Yehoash Raphael for critical comments regarding the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Rights and permissions
About this article
Cite this article
Wang, GP., Guo, JY., Peng, Z. et al. Adeno-associated virus-mediated gene transfer targeting normal and traumatized mouse utricle. Gene Ther 21, 958–966 (2014). https://doi.org/10.1038/gt.2014.73
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2014.73