Abstract
Introduction:
Gastric bypass surgery (GBP) leads to sustained weight loss and significant improvement in type 2 diabetes (T2DM). Bile acids (BAs), signaling molecules which influence glucose metabolism, are a potential mediator for the improvement in T2DM after GBP. This study sought to investigate the effect of GBP on BA levels and composition in individuals with T2DM.
Methods:
Plasma BA levels and composition and fibroblast growth factor (FGF)-19 levels were measured during fasting and in response to an oral glucose load before and at 1 month and 2 years post GBP in 13 severely obese women with T2DM.
Results:
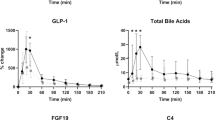
A striking temporal change in BA levels and composition was observed after GBP. During the fasted state, BA concentrations were generally reduced at 1 month, but increased 2 years post GBP. Postprandial BA levels were unchanged 1 month post GBP, but an exaggerated postprandial peak was observed 2 years after the surgery. A significant increase in the 12α-hydroxylated/non12α-hydroxylated BA ratio during fasting and postprandially at 2 years, but not 1 month, post GBP was observed. Significant correlations between BAs vs FGF-19, body weight, the incretin effect and peptide YY (PYY) were also found.
Conclusions:
This study provides evidence that GBP temporally modifies the concentration and composition of circulating BAs in individuals with T2DM, and suggests that BAs may be linked to the improvement in T2DM after GBP.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schauer PR, Bhatt DL, Kirwan JP, Wolski K, Brethauer SA, Navaneethan SD et al. Bariatric surgery versus intensive medical therapy for diabetes—3-year outcomes. N Engl J Med 2014; 370: 2002–2013.
Arterburn DE, Bogart A, Sherwood NE, Sidney S, Coleman KJ, Haneuse S et al. A multisite study of long-term remission and relapse of type 2 diabetes mellitus following gastric bypass. Obes Surg 2013; 23: 93–102.
Zhang Y, Lee FY, Barrera G, Lee H, Vales C, Gonzalez FJ et al. Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc Natl Acad Sci USA 2006; 103: 1006–1011.
Smushkin G, Sathananthan M, Piccinini F, Dalla Man C, Law JH, Cobelli C et al. The effect of a bile acid sequestrant on glucose metabolism in subjects with type 2 diabetes. Diabetes 2013; 62: 1094–1101.
Watanabe M, Houten SM, Mataki C, Christoffolete MA, Kim BW, Sato H et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature 2006; 439: 484–489.
Patti ME, Houten SM, Bianco AC, Bernier R, Larsen PR, Holst JJ et al. Serum bile acids are higher in humans with prior gastric bypass: potential contribution to improved glucose and lipid metabolism. Obesity 2009; 17: 1671–1677.
Pournaras DJ, Glicksman C, Vincent RP, Kuganolipava S, Alaghband-Zadeh J, Mahon D et al. The role of bile after Roux-en-Y gastric bypass in promoting weight loss and improving glycaemic control. Endocrinology 2012; 153: 3613–3619.
Simonen M, Dali-Youcef N, Kaminska D, Venesmaa S, Kakela P, Paakkonen M et al. Conjugated bile acids associate with altered rates of glucose and lipid oxidation after Roux-en-Y gastric bypass. Obes Surg 2012; 22: 1473–1480.
Gerhard GS, Styer AM, Wood GC, Roesch SL, Petrick AT, Gabrielsen J et al. A role for fibroblast growth factor 19 and bile acids in diabetes remission after Roux-en-Y gastric bypass. Diabetes Care 2013; 36: 1859–1864.
Werling M, Vincent RP, Cross GF, Marschall HU, Fandriks L, Lonroth H et al. Enhanced fasting and post-prandial plasma bile acid responses after Roux-en-Y gastric bypass surgery. Scand J Gastroenterol 2013; 48: 1257–1264.
Scholtz S, Miras AD, Chhina N, Prechtl CG, Sleeth ML, Daud NM et al. Obese patients after gastric bypass surgery have lower brain-hedonic responses to food than after gastric banding. Gut 2014; 63: 891–902.
Kohli R, Bradley D, Setchell KD, Eagon JC, Abumrad N, Klein S . Weight loss induced by Roux-en-Y gastric bypass but not laparoscopic adjustable gastric banding increases circulating bile acids. J Clin Endocrinol Metab 2013; 98: E708–E712.
Steinert RE, Peterli R, Keller S, Meyer-Gerspach AC, Drewe J, Peters T et al. Bile acids and gut peptide secretion after bariatric surgery: a 1-year prospective randomized pilot trial. Obesity 2013; 21: E660–E668.
Ahmad NN, Pfalzer A, Kaplan LM . Roux-en-Y gastric bypass normalizes the blunted postprandial bile acid excursion associated with obesity. Int J Obes (Lond) 2013; 37: 1553–1559.
Jansen PL, van Werven J, Aarts E, Berends F, Janssen I, Stoker J et al. Alterations of hormonally active fibroblast growth factors after Roux-en-Y gastric bypass surgery. Dig Dis 2011; 29: 48–51.
Prawitt J, Caron S, Staels B . Bile acid metabolism and the pathogenesis of type 2 diabetes. Curr Diab Rep 2011; 11: 160–166.
Porez G, Prawitt J, Gross B, Staels B . Bile acid receptors as targets for the treatment of dyslipidemia and cardiovascular disease. J Lipid Res 2012; 53: 1723–1737.
Thomas C, Gioiello A, Noriega L, Strehle A, Oury J, Rizzo G et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metab 2009; 10: 167–177.
Sato H, Genet C, Strehle A, Thomas C, Lobstein A, Wagner A et al. Anti-hyperglycemic activity of a TGR5 agonist isolated from Olea europaea. Biochem Biophys Res Commun 2007; 362: 793–798.
Chiang JY . Bile acids: regulation of synthesis. J Lipid Res 2009; 50: 1955–1966.
Song KH, Li T, Owsley E, Strom S, Chiang JY . Bile acids activate fibroblast growth factor 19 signaling in human hepatocytes to inhibit cholesterol 7alpha-hydroxylase gene expression. Hepatology 2009; 49: 297–305.
Sinal CJ, Tohkin M, Miyata M, Ward JM, Lambert G, Gonzalez FJ . Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell 2000; 102: 731–744.
Cariou B, van Harmelen K, Duran-Sandoval D, van Dijk TH, Grefhorst A, Abdelkarim M et al. The farnesoid X receptor modulates adiposity and peripheral insulin sensitivity in mice. J Biol Chem 2006; 281: 11039–11049.
Ma K, Saha PK, Chan L, Moore DD . Farnesoid X receptor is essential for normal glucose homeostasis. J Clin Invest 2006; 116: 1102–1109.
Haeusler RA, Astiarraga B, Camastra S, Accili D, Ferrannini E . Human insulin resistance is associated with increased plasma levels of 12alpha-hydroxylated bile acids. Diabetes 2013; 62: 4184–4191.
Brufau G, Stellaard F, Prado K, Bloks VW, Jonkers E, Boverhof R et al. Improved glycemic control with colesevelam treatment in patients with type 2 diabetes is not directly associated with changes in bile acid metabolism. Hepatology 2010; 52: 1455–1464.
Haeusler RA, Pratt-Hyatt M, Welch CL, Klaassen CD, Accili D . Impaired generation of 12-hydroxylated bile acids links hepatic insulin signaling with dyslipidemia. Cell Metab 2012; 15: 65–74.
Uchida K, Makino S, Akiyoshi T . Altered bile acid metabolism in nonobese, spontaneously diabetic (NOD) mice. Diabetes 1985; 34: 79–83.
Laferrère B, Heshka S, Wang K, Khan Y, McGinty J, Teixeira J et al. Incretin levels and effect are markedly enhanced 1 month after Roux-en-Y gastric bypass surgery in obese patients with type 2 diabetes. Diabetes Care 2007; 30: 1709–1716.
Tagliacozzi D, Mozzi AF, Casetta B, Bertucci P, Bernardini S, Di Ilio C et al. Quantitative analysis of bile acids in human plasma by liquid chromatography-electrospray tandem mass spectrometry: a simple and rapid one-step method. Clin Chem Lab Med 2003; 41: 1633–1641.
Magee L . R2 measures based on Wald and likelihood ratio joint significance tests. Am Stat 1990; 44: 250–253.
Kong LC, Tap J, Aron-Wisnewsky J, Pelloux V, Basdevant A, Bouillot JL et al. Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. Am J Clin Nutr 2013; 98: 16–24.
Liou AP, Paziuk M, Luevano Jr JM, Machineni S, Turnbaugh PJ, Kaplan LM . Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med 2013; 5: 178ra41.
Furet JP, Kong LC, Tap J, Poitou C, Basdevant A, Bouillot JL et al. Differential adaptation of human gut microbiota to bariatric surgery-induced weight loss: links with metabolic and low-grade inflammation markers. Diabetes 2010; 59: 3049–3057.
Zhang H, DiBaise JK, Zuccolo A, Kudrna D, Braidotti M, Yu Y et al. Human gut microbiota in obesity and after gastric bypass. Proc Natl Acad Sci USA 2009; 106: 2365–2370.
Kaur A, Patankar JV, de Haan W, Ruddle P, Wijesekara N, Groen AK et al. Loss of Cyp8b1 improves glucose homeostasis by increasing GLP-1. Diabetes 2014. e-pub ahead of print 22 October 2014.
Fu ZD, Klaassen CD . Increased bile acids in enterohepatic circulation by short-term calorie restriction in male mice. Toxicol Appl Pharmacol 2013; 273: 680–690.
Glicksman C, Pournaras DJ, Wright M, Roberts R, Mahon D, Welbourn R et al. Postprandial plasma bile acid responses in normal weight and obese subjects. Ann Clin Biochem 2010; 47: 482–484.
Vincent RP, Omar S, Ghozlan S, Taylor DR, Cross G, Sherwood RA et al. Higher circulating bile acid concentrations in obese patients with type 2 diabetes. Ann Clin Biochem 2013; 50: 360–364.
Wewalka M, Patti ME, Barbato C, Houten SM, Goldfine AB . Fasting serum taurine-conjugated bile acids are elevated in type 2 diabetes and do not change with intensification of insulin. J Clin Endocrinol Metab 2014; 99: 1442–1451.
Cariou B, Chetiveaux M, Zair Y, Pouteau E, Disse E, Guyomarc'h-Delasalle B et al. Fasting plasma chenodeoxycholic acid and cholic acid concentrations are inversely correlated with insulin sensitivity in adults. Nutr Metab 2011; 8: 48.
Taylor DR, Alaghband-Zadeh J, Cross GF, Omar S, le Roux CW, Vincent RP . Urine bile acids relate to glucose control in patients with type 2 diabetes mellitus and a body mass index below 30kg/m2. PLoS One 2014; 9: e93540.
Lundasen T, Galman C, Angelin B, Rudling M . Circulating intestinal fibroblast growth factor 19 has a pronounced diurnal variation and modulates hepatic bile acid synthesis in man. J Int Med 2006; 260: 530–536.
Tomlinson E, Fu L, John L, Hultgren B, Huang X, Renz M et al. Transgenic mice expressing human fibroblast growth factor-19 display increased metabolic rate and decreased adiposity. Endocrinology 2002; 143: 1741–1747.
Fu L, John LM, Adams SH, Yu XX, Tomlinson E, Renz M et al. Fibroblast growth factor 19 increases metabolic rate and reverses dietary and leptin-deficient diabetes. Endocrinology 2004; 145: 2594–2603.
Ryan KK, Kohli R, Gutierrez-Aguilar R, Gaitonde SG, Woods SC, Seeley RJ . Fibroblast growth factor-19 action in the brain reduces food intake and body weight and improves glucose tolerance in male rats. Endocrinology 2013; 154: 9–15.
Ryan KK, Tremaroli V, Clemmensen C, Kovatcheva-Datchary P, Myronovych A, Karns R et al. FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature 2014; 509: 183–188.
Adrian TE, Gariballa S, Parekh KA, Thomas SA, Saadi H, Al Kaabi J et al. Rectal taurocholate increases L cell and insulin secretion, and decreases blood glucose and food intake in obese type 2 diabetic volunteers. Diabetologia 2012; 55: 2343–2347.
Katsuma S, Hirasawa A, Tsujimoto G . Bile acids promote glucagon-like peptide-1 secretion through TGR5 in a murine enteroendocrine cell line STC-1. Biochem Biophys Res Commun 2005; 329: 386–390.
Ullmer C, Alvarez Sanchez R, Sprecher U, Raab S, Mattei P, Dehmlow H et al. Systemic bile acid sensing by G protein-coupled bile acid receptor 1 (GPBAR1) promotes PYY and GLP-1 release. Br J Pharmacol 2013; 169: 671–684.
Wu T, Bound MJ, Standfield SD, Gedulin B, Jones KL, Horowitz M et al. Effects of rectal administration of taurocholic acid on glucagon-like peptide-1 and peptide YY secretion in healthy humans. Diabetes Obes Metab 2013; 15: 474–477.
Reimann F, Habib AM, Tolhurst G, Parker HE, Rogers GJ, Gribble FM . Glucose sensing in L cells: a primary cell study. Cell Metab 2008; 8: 532–539.
Roberts RE, Glicksman C, Alaghband-Zadeh J, Sherwood RA, Akuji N, le Roux CW . The relationship between postprandial bile acid concentration, GLP-1, PYY and ghrelin. Clin Endocrinol 2011; 74: 67–72.
Morinigo R, Moize V, Musri M, Lacy AM, Navarro S, Marin JL et al. Glucagon-like peptide-1, peptide YY, hunger, and satiety after gastric bypass surgery in morbidly obese subjects. J Clin Endocrinol Metab 2006; 91: 1735–1740.
Nguyen NQ, Debreceni TL, Bambrick JE, Bellon M, Wishart J, Standfield S et al. Rapid gastric and intestinal transit is a major determinant of changes in blood glucose, intestinal hormones, glucose absorption and postprandial symptoms after gastric bypass. Obesity 2014; 22: 2003–2009.
Wang G, Agenor K, Pizot J, Kotler DP, Harel Y, Van Der Schueren BJ et al. Accelerated gastric emptying but no carbohydrate malabsorption 1 year after gastric bypass surgery (GBP). Obes Surg 2012; 22: 1263–1267.
Dirksen C, Damgaard M, Bojsen-Moller KN, Jorgensen NB, Kielgast U, Jacobsen SH et al. Fast pouch emptying, delayed small intestinal transit, and exaggerated gut hormone responses after Roux-en-Y gastric bypass. Neurogastroenterol Motil 2013; 25: 346–e255.
le Roux CW, Borg C, Wallis K, Vincent RP, Bueter M, Goodlad R et al. Gut hypertrophy after gastric bypass is associated with increased glucagon-like peptide 2 and intestinal crypt cell proliferation. Ann Surg 2010; 252: 50–56.
Ridlon JM, Kang DJ, Hylemon PB . Bile salt biotransformations by human intestinal bacteria. J Lipid Res 2006; 47: 241–259.
Galman C, Angelin B, Rudling M . Pronounced variation in bile acid synthesis in humans is related to gender, hypertriglyceridaemia and circulating levels of fibroblast growth factor 19. J Int Med 2011; 270: 580–588.
Steiner C, Othman A, Saely CH, Rein P, Drexel H, von Eckardstein A et al. Bile acid metabolites in serum: intraindividual variation and associations with coronary heart disease, metabolic syndrome and diabetes mellitus. PLoS One 2011; 6: e25006.
Trottier J, Caron P, Straka RJ, Barbier O . Profile of serum bile acids in noncholestatic volunteers: gender-related differences in response to fenofibrate. Clin Pharmacol Ther 2011; 90: 279–286.
Kimball A, Pertsemlidis D, Panveliwalla D . Composition of biliary lipids and kinetics of bile acids after cholecystectomy in man. Am J Dig Dis 1976; 21: 776–781.
Almond HR, Vlahcevic ZR, Bell Jr CC, Gregory DH, Swell L . Bile acid pools, kinetics and biliary lipid composition before and after cholecystectomy. N Engl J Med 1973; 289: 1213–1216.
Berr F, Stellaard F, Pratschke E, Paumgartner G . Effects of cholecystectomy on the kinetics of primary and secondary bile acids. J Clin Invest 1989; 83: 1541–1550.
Kullak-Ublick GA, Paumgartner G, Berr F . Long-term effects of cholecystectomy on bile acid metabolism. Hepatology 1995; 21: 41–45.
Angelin B, Bjorkhem I, Einarsson K, Ewerth S . Hepatic uptake of bile acids in man. Fasting and postprandial concentrations of individual bile acids in portal venous and systemic blood serum. J Clin Invest 1982; 70: 724–731.
Acknowledgements
We thank our participants, Yim Dam and Ping Zhou (technicians from the NYONRC Hormonal Core laboratory), and the entire GCRC-CTSA staff. This work was funded by grants from the American Diabetes Association (CR-7-05 CR-18) and NIH (R01-DK067561, P30-DK26687, P30-DK063608). This publication was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through grant number UL1 TR000040, formerly the National Center for Research Resources, grant number UL1 RR024156. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. RD was supported by the NYONRC training grant 5T32DK007559-22. ClR received funding from Science Foundation Ireland 12/YI/B2480.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Rights and permissions
About this article
Cite this article
Dutia, R., Embrey, M., O'Brien, S. et al. Temporal changes in bile acid levels and 12α-hydroxylation after Roux-en-Y gastric bypass surgery in type 2 diabetes. Int J Obes 39, 806–813 (2015). https://doi.org/10.1038/ijo.2015.1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2015.1
This article is cited by
-
Research progress on the relationship between bile acid metabolism and type 2 diabetes mellitus
Diabetology & Metabolic Syndrome (2023)
-
Reversal of NAFLD After VSG Is Independent of Weight-Loss but RYGB Offers More Efficacy When Maintained on a High-Fat Diet
Obesity Surgery (2022)
-
High-throughput mediation analysis of human proteome and metabolome identifies mediators of post-bariatric surgical diabetes control
Nature Communications (2021)
-
Metabolites and diabetes remission after weight loss
Nutrition & Diabetes (2021)
-
Bile acid, glucose, lipid profile, and liver enzyme changes in prediabetic patients 1 year after sleeve gastrectomy
BMC Surgery (2020)