Abstract
BACKGROUND:
Protein infusion in the small intestine results in intestinal brake activation: a negative feedback mechanism that may be mediated by the release of gastrointestinal peptides resulting in a reduction in food intake. It has been proposed that duodenum, jejunum and ileum may respond differently to infused proteins.
OBJECTIVE:
To investigate differences in ad libitum food intake, feelings of hunger and satiety and the systemic levels of cholecystokinin (CCK), glucagon-like peptide-1 (GLP-1), peptide YY (PYY), glucose and insulin after intraduodenal, intrajejunal and intraileal protein infusion.
METHODS:
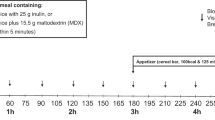
Fourteen subjects (four male, mean age: 23±2.1 years, mean body mass index: 21.6±1.8 kg m−2) were intubated with a naso-ileal catheter in this double-blind, randomized, placebo-controlled crossover study. Test days (four in total, executed on consecutive days) started with the ingestion of a standardized breakfast, followed by the infusion of 15 g of protein in the duodenum, jejunum or ileum over a period of 60 min. Food intake was measured by offering an ad libitum meal and Visual Analogue Scale (VAS) scores were used to assess feelings of hunger and satiety. Blood samples were drawn at regular intervals for CCK, GLP-1, PYY, glucose and insulin analyses.
RESULTS:
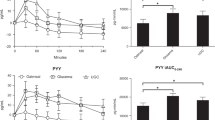
Intraileal protein infusion decreased ad libitum food intake compared with both intraduodenal and placebo infusion (ileum: 628.5±63 kcal vs duodenum: 733.6±50 kcal, P<0.01 and placebo: 712.2±53 kcal, P<0.05). GLP-1 concentrations were increased after ileal infusion compared with jejunal and placebo infusion, whereas CCK concentrations were only increased after intraileal protein infusion compared with placebo. None of the treatments affected VAS scores for hunger and satiety nor plasma concentrations of PYY and glucose.
CONCLUSIONS:
Protein infusion into the ileum decreases food intake during the next meal compared with intraduodenal infusion, whereas it increases systemic levels of GLP-1 compared with protein infusion into the jejunum and placebo respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Astrup A . The satiating power of protein—a key to obesity prevention? Am J Clin Nutr 2005; 82: 1–2.
Paddon-Jones D, Westman E, Mattes RD, Wolfe RR, Astrup A, Westerterp-Plantenga M . Protein, weight management, and satiety. Am J Clin Nutr 2008; 87: 1558S–1561SS.
Weigle DS, Breen PA, Matthys CC, Callahan HS, Meeuws KE, Burden VR et al. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am J Clin Nutr 2005; 82: 41–48.
Cummings DE, Overduin J . Gastrointestinal regulation of food intake. J Clin Invest 2007; 117: 13–23.
Mars M, Stafleu A, de Graaf C . Use of satiety peptides in assessing the satiating capacity of foods. Physiol Behav 2012; 105: 483–488.
Geraedts MC, Troost FJ, Munsters MJ, Stegen JH, de Ridder RJ, Conchillo JM et al. Intraduodenal administration of intact pea protein effectively reduces food intake in both lean and obese male subjects. PloS ONE 2011; 6: e24878.
Shin HS, Ingram JR, McGill AT, Poppitt SD . Lipids, CHOs, proteins: can all macronutrients put a 'brake' on eating? Physiol Behav 2013; 120: 114–123.
Ryan AT, Feinle-Bisset C, Kallas A, Wishart JM, Clifton PM, Horowitz M et al. Intraduodenal protein modulates antropyloroduodenal motility, hormone release, glycemia, appetite, and energy intake in lean men. Am J Clin Nutr 2012; 96: 474–482.
Ryan AT, Luscombe-Marsh ND, Saies AA, Little TJ, Standfield S, Horowitz M et al. Effects of intraduodenal lipid and protein on gut motility and hormone release, glycemia, appetite, and energy intake in lean men. Am J Clin Nutr 2013; 98: 300–311.
Soenen S, Giezenaar C, Hutchison AT, Horowitz M, Chapman I, Luscombe-Marsh ND . Effects of intraduodenal protein on appetite, energy intake, and antropyloroduodenal motility in healthy older compared with young men in a randomized trial. Am J Clin Nutr 2014; 100: 1108–1115.
Maljaars J, Haddeman E, Peters H, Masclee AA . Comparison of ileal and duodenal brake mechanisms on satiety and gastrointestinal transport. Gastroenterology 2007; 132: A207–A208.
Maljaars PW, Peters HP, Mela DJ, Masclee AA . Ileal brake: a sensible food target for appetite control. A review. Physiol Behav 2008; 95: 271–281.
Atkinson RL, Whipple JH, Atkinson SH, Stewart CC . Role of the small bowel in regulating food intake in rats. Am J Physiol 1982; 242: R429–R433.
le Roux CW, Welbourn R, Werling M, Osborne A, Kokkinos A, Laurenius A et al. Gut hormones as mediators of appetite and weight loss after Roux-en-Y gastric bypass. Ann Surg 2007; 246: 780–785.
DeMaria EJ . Bariatric surgery for morbid obesity. N Engl J Med 2007; 356: 2176–2183.
van Avesaat M, Troost FJ, Ripken D, Hendriks HF, Masclee AAM . Ileal brake activation: macronutrient-specific effects on eating behavior? Int J Obesity 2015; 39: 235–243.
Blundell J, de Graaf C, Hulshof T, Jebb S, Livingstone B, Lluch A et al. Appetite control: methodological aspects of the evaluation of foods. Obes Rev 2010; 11: 251–270.
Bendtsen LQ, Lorenzen JK, Bendsen NT, Rasmussen C, Astrup A . Effect of dairy proteins on appetite, energy expenditure, body weight, and composition: a review of the evidence from controlled clinical trials. Adv Nutr 2013; 4: 418–438.
Calbet JA, Holst JJ . Gastric emptying, gastric secretion and enterogastrone response after administration of milk proteins or their peptide hydrolysates in humans. Eur J Nutr 2004; 43: 127–139.
Lang V, Bellisle F, Oppert JM, Craplet C, Bornet FR, Slama G et al. Satiating effect of proteins in healthy subjects: a comparison of egg albumin, casein, gelatin, soy protein, pea protein, and wheat gluten. Am J Clin Nutr 1998; 67: 1197–1204.
Veldhorst MA, Nieuwenhuizen AG, Hochstenbach-Waelen A, van Vught AJ, Westerterp KR, Engelen MP et al. Dose-dependent satiating effect of whey relative to casein or soy. Physiol Behav 2009; 96: 675–682.
Veldhorst MA, Nieuwenhuizen AG, Hochstenbach-Waelen A, Westerterp KR, Engelen MP, Brummer RJ et al. A breakfast with alpha-lactalbumin, gelatin, or gelatin+TRP lowers energy intake at lunch compared with a breakfast with casein, soy, whey, or whey-GMP. Clin Nutr 2009; 28: 147–155.
Powley TL, Phillips RJ . Gastric satiation is volumetric, intestinal satiation is nutritive. Physiol Behav 2004; 82: 69–74.
Feinle C, Christen M, Grundy D, Faas H, Meier O, Otto B et al. Effects of duodenal fat, protein or mixed-nutrient infusions on epigastric sensations during sustained gastric distension in healthy humans. Neurogastroenterol Motil 2002; 14: 205–213.
Janssen P, Vanden Berghe P, Verschueren S, Lehmann A, Depoortere I, Tack J . Review article: the role of gastric motility in the control of food intake. Aliment Pharmacol Ther 2011; 33: 880–894.
Korner J, Bessler M, Cirilo LJ, Conwell IM, Daud A, Restuccia NL et al. Effects of Roux-en-Y gastric bypass surgery on fasting and postprandial concentrations of plasma ghrelin, peptide YY, and insulin. J Clin Endocrinol Metab 2005; 90: 359–365.
le Roux CW, Aylwin SJ, Batterham RL, Borg CM, Coyle F, Prasad V et al. Gut hormone profiles following bariatric surgery favor an anorectic state, facilitate weight loss, and improve metabolic parameters. Ann Surg 2006; 243: 108–114.
Strader AD, Vahl TP, Jandacek RJ, Woods SC, D'Alessio DA, Seeley RJ . Weight loss through ileal transposition is accompanied by increased ileal hormone secretion and synthesis in rats. Am J Physiol Endocrinol Metab 2005; 288: E447–E453.
Lutz TA, Bueter M . Physiological mechanisms behind Roux-en-Y gastric bypass surgery. Dig Surg 2014; 31: 13–24.
Salinari S, Carr RD, Guidone C, Bertuzzi A, Cercone S, Riccioni ME et al. Nutrient infusion bypassing duodenum-jejunum improves insulin sensitivity in glucose-tolerant and diabetic obese subjects. Am J Physiol Endocrinol Metab 2013; 305: E59–E66.
Nauck MA, Homberger E, Siegel EG, Allen RC, Eaton RP, Ebert R et al. Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J Clin Endocrinol Metab 1986; 63: 492–498.
Sachdeva P, Kantor S, Knight LC, Maurer AH, Fisher RS, Parkman HP . Use of a high caloric liquid meal as an alternative to a solid meal for gastric emptying scintigraphy. Dig Dis Sci 2013; 58: 2001–2006.
Windey K, De Preter V, Verbeke K . Relevance of protein fermentation to gut health. Mol Nutr Food Res 2012; 56: 184–196.
Bojsen-Moller KN, Jacobsen SH, Dirksen C, Jorgensen NB, Reitelseder S, Jensen JB et al. Accelerated protein digestion and amino acid absorption after Roux-en-Y gastric bypass. Am J Clin Nutr 2015; 102: 600–607.
Acknowledgements
The research was funded by TI Food and Nutrition, a public-private partnership on pre-competitive research in food and nutrition. All funders had an input in the study design, whereas study conduct, data collection and analysis, as well as manuscript writing were the sole responsibility of the academic partners. Registered at clinicaltrials.gov: NCT02500069. Names for PubMed indexing: van Avesaat, Ripken, Hendriks, Masclee, Troost.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
van Avesaat, M., Ripken, D., Hendriks, H. et al. Small intestinal protein infusion in humans: evidence for a location-specific gradient in intestinal feedback on food intake and GI peptide release. Int J Obes 41, 217–224 (2017). https://doi.org/10.1038/ijo.2016.196
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2016.196
This article is cited by
-
Small intestinal CaSR-dependent and CaSR-independent protein sensing regulates feeding and glucose tolerance in rats
Nature Metabolism (2024)
-
The metabolic impact of small intestinal nutrient sensing
Nature Communications (2021)