Abstract
Objective:
The purpose of this pilot trial was to determine whether rates of contact dermatitis following cutaneous antisepsis for central catheter placement were similar among neonates treated with chlorhexidine gluconate and povidone-iodine. Chlorhexidine gluconate absorption was also evaluated.
Study Design:
Infants weighing ⩾1500 g and ⩾7 days of age were randomized to a 10% povidone-iodine or 2% chlorhexidine gluconate site scrub before catheter placement. Primary outcomes evaluated included dermatitis, catheter colonization and chlorhexidine gluconate absorption.
Result:
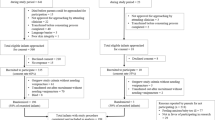
A total of 48 neonates were enrolled. Colonization rates were similar among treatment groups (P<0.6). Dermatitis did not occur at chlorhexidine gluconate (central catheters, n=24; peripheral catheters, n=29) sites. Seven neonates had measurable chlorhexidine gluconate concentrations (range 13 to 100 ng ml−1) during catheterization.
Conclusion:
In this small trial chlorhexidine gluconate antisepsis was tolerated by study neonates. Chlorhexidine gluconate was cutaneously absorbed. Larger trials are needed to determine efficacy and tolerance of chlorhexidine gluconate in neonates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Stoll BJ, Gordon T, Korones SB, Shankaran S, Tyson JE, Bauer CR et al. Late-onset sepsis in very low birth weight neonates: a report from the National Institute of Child Health and Human Development Neonatal Research Network. J Pediatr 1996; 129: 63–71.
Gray JE, Richardson DK, McCormick MC, Goldmann DA . Coagulase-negative staphylococcal bacteremia among very low birth weight infants: relation to admission illness severity, resource use, and outcome. Pediatrics 1995; 95: 225–230.
Fanaroff AA, Korones SB, Wright LL, Verter J, Poland RL, Bauer CR et al. Incidence, presenting features, risk factors, and significance of late onset septicemia in very low birth weight infants. The National Institute of Child Health and Human Development Neonatal Research Network. Pediatr Infect Dis J 1998; 17: 593–598.
Sohn AG, Garrett DO, Sinkowitz-Cochran RL, Grohskopf LA, Levine GL, Stover BH et al. Prevalence of nosocomial infections in neonatal intensive care unit patients: results from the first national point-prevalence survey. J Pediatr 2001; 139: 821–827.
Stoll BJ, Hansen NI, Adams-Chapman I, Fanaroff AA, Hintz SR, Vohr B et al. Neurodevelopmental and growth impairment among extremely low-birth-weight infants with neonatal infection. JAMA 2004; 292: 2357–2365.
Smerdely P, Lim A, Boyages SC, Waite K, Wu D, Roberts V et al. Topical iodine-containing antiseptics and neonatal hypothyroidism in very-low-birthweight infants. Lancet 1989; 2 (8664): 661–664.
Parravicini E, Fontana C, Paterlini G, Tagliabue P, Rovelli F, Leung K et al. Iodine, thyroid function, and very low birth weight infants. Pediatrics 1996; 98: 730–734.
Gordan CM, Rowitch DH, Mitchell ML, Kohane IS . Topical iodine and neonatal hypothyroidism. Arch Pediatr Adolesc Med 1995; 149: 1336–1339.
Brown RS, Bloomfield S, Bednarek FJ, Mitchell ML, Braverman L . Routine skin cleansing with povidone-iodine is not a common cause of transient neonatal hypothyroidism in North America: a prospective controlled study. Thyroid 1997; 7: 395–400.
Mimoz O, Pieroni L, Lawrence C, Edouard A, Costa Y, Samii K et al. Prospective, randomized trial of two antiseptic solutions for prevention of central venous or arterial catheter colonization and infection in intensive care unit patients. Crit Care Med 1996; 24: 1818–1823.
Maki DG, Ringer M, Alvarado CJ . Prospective randomized trial of povidone-iodine, alcohol and chlorhexidine for prevention of infection associated with central venous and arterial catheters. Lancet 1991; 338: 339–343.
Garland JS, Buck RK, Maloney P, Durkin BM, Toth-Lloyd S, Duffy M et al. A comparison of 10% povidone-iodine and 0.5% chlorhexidine gluconate for the prevention of peripheral intravenous catheter colonization in neonates: a prospective trial. Pediatr Infect Dis J 1995; 14: 510–516.
Chaiyakunapruk N, Veenstra DL, Lipsky B, Saint S . Chlorhexidine compared with povidone-iodine solution for vascular catheter-site care: a meta-analysis. Ann Intern Med 2002; 136: 792–801.
Maki DG, Weise CE, Sarafin HW . A semiquantitative culture method for identifying intravenous catheter-related infection. N Engl J Med 1997; 296: 1305–1309.
Brun-Buisson C, Abrouk F, Legrand P, Huet Y, Larabi S, Rapin M . Diagnosis of central venous catheter-related sepsis. Critical level of quantitative tip cultures. Arch Intern Med 1987; 147: 873–877.
Collignon PJ, Soni N, Pearson IY . Is semiquantitative culture of central vein catheter tips useful in the diagnosis of catheter-associated bacteremia? J Clin Microbiol 1986; 24: 532–535.
Cooper GL, Hopkins CC . Rapid diagnosis of intravascular catheter-associated infection by direct Gram staining of catheter segments. N Engl J Med 1985; 18: 1142–1147.
Garland JS, Alex C, Mueller CP, Cisler-Kahill LA . Local reactions to a chlorhexidine gluconate-impregnated antimicrobial dressing in very low birth weight infants. Pediatr Infect Dis J 1996; 15: 912–914.
Richardson DK, Gray JE, McCormick MC, Workman K, Goldmann DA . Score for Neonatal Acute Physiology: a physiologic severity index for neonatal intensive care. Pediatrics 1993; 91: 617–623.
Aly R, Mailbach HI . Effect of antimicrobial soap containing chlorhexidine on the microbial flora of skin. Appl Environ Microbiol 1976; 31: 931–935.
Shelton DM . A comparison of the effects of two antiseptic agents on staphylococcus epidermis colony forming units at the peritoneal dialysis catheter exit site. Adv Perit Dial 1991; 7: 120–124.
Andersen C, Hart J, Vemgal P, Harrison C . Prospective evaluation of a multi-factorial prevention strategy on the impact of nosocomial infection in very-low-birthweight infants. J Hosp Infect 2005; 61: 162–167.
Okano M, Nomura M, Hata S, Okada N, Sato K, Kitano Y et al. Anaphylactic symptoms due to chlorhexidine gluconate. Arch Dermatol 1989; 125: 50–52.
Oda T, Hamasaki J, Kanda N, Mikami K . Anaphylactic shock induced by an antiseptic-coated central venous catheter. Anesthesiology 1997; 87: 1242–1244.
Garland JS, Alex CP, Sevallius JM, Murphy DM, Good MJ, Volberding AM et al. Cohort study of the pathogenesis and molecular epidemiology of catheter-related bloodstream infection in neonates with peripherally inserted central venous catheters. Infect Control Hosp Epidemiol 2008; 29: 243–249.
Garland JS, Alex CP, Mueller CD, Otten D, Shivpuri C, Harris MC et al. A randomized trial comparing povidone-iodine to a chlorhexidine gluconate-impregnated dressing for prevention of central venous catheter infections in neonates. Pediatrics 2001; 107: 1431–1437.
Obrien CA, Blummer JL, Speck WT, Carr H . Effect of bathing with a 4% chlorhexidine gluconate solution on neonatal bacterial colonization. J Hosp Infect 1984; 5 (Suppl A): 141.
Cowen J, Ellis SH, McAinsh J . Absorption of chlorhexidine from intact skin of newborn infants. Arch Dis Child 1979; 54: 379–383.
Agget PJ, Copper LV, Ellis SH, McAinsh J . Percutaneous absorption of chlorhexidine in neonatal cord care. Arch Dis Child 1981; 56: 878–891.
Case DE, McAinsh J, Rushton A, Winrow MJ . Chlorhexidine: attempts to detect percutaneous absorption in man. Chemotherapy 1976; 3: 367–374.
Acknowledgements
We acknowledge the research coordinator assistance received from the various centers—Wheaton Franciscan Healthcare, St Joseph: Elizabeth Bongert, Melinda P Bushong, Leslie L Hofer, Mary Koehnke, Heidi May, Lori A McLain, Dawn M Murphy, Tiffany N Perine, Jackie M Sevallius, Rae Toman, Amy WernerWoida. Aurora Sinai Medical Center: Joan Becker; Children's Hospital of Wisconsin: Laura Lane; Children's Hospital of Philadelphia: Linda Corcoran, Jane Fricko, Sharon Zirin; Children's Hospital (Boston): Hannah Pawlzendo. Financial support was provided by Enturia (formerly Medi-Flex). The sponsor reviewed the study design developed by the primary investigator. It was not involved with the data collection, implementation, analysis or interpretation of data. They had an opportunity to review the paper before publication. The project described was supported by Grant Number UL1-RR-024134 from the National Center for Research Resources. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garland, J., Alex, C., Uhing, M. et al. Pilot trial to compare tolerance of chlorhexidine gluconate to povidone-iodine antisepsis for central venous catheter placement in neonates. J Perinatol 29, 808–813 (2009). https://doi.org/10.1038/jp.2009.161
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2009.161
Keywords
This article is cited by
-
Myelin toxicity of chlorhexidine in zebrafish larvae
Pediatric Research (2023)
-
Global variation in skin injures and skincare practices in extremely preterm infants
World Journal of Pediatrics (2023)
-
2% aqueous vs alcohol-based chlorhexidine for skin antisepsis in VLBW neonates undergoing peripheral venipuncture: a non-inferiority trial
Journal of Perinatology (2022)
-
Chlorhexidine baths in preterm infants — are we there yet?
Journal of Perinatology (2019)
-
Prävention von Gefäßkatheter-assoziierten Infektionen bei Früh- und Neugeborenen
Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (2018)