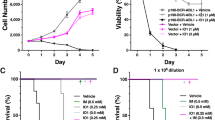
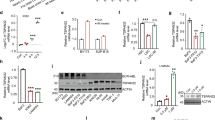
Abstract
Chronic myelogenous leukemia (CML) is a malignant disease characterized by expression of p210-BCR-ABL, the product of the Philadelphia chromosome. Survival of CML patients has been significantly improved with the introduction of tyrosine kinase inhibitors that induce long-term hematologic remissions. However, mounting evidence indicates that the use of a single tyrosine kinase inhibitor does not cure this disease due to the persistence of p210-BCR-ABL at the molecular level or the acquired resistance in the stem cell compartment to individual inhibitors. We have recently shown in a murine model that deficiency of the Rho GTPases Rac1 and Rac2 significantly reduces p210-BCR-ABL-mediated proliferation in vitro and myeloproliferative disease in vivo, suggesting Rac as a potential therapeutic target in p210-BCR-ABL-induced disease. This target has been further validated using a first-generation Rac-specific small molecule inhibitor. In this review we describe the role of Rac GTPases in p210-BCR-ABL-induced leukemogenesis and explore the possibility of combinatorial therapies that include tyrosine kinase inhibitor(s) and Rac GTPase inhibitors in the treatment of CML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Daley GQ, Van Etten RA, Baltimore D . Induction of chronic myelogenous leukemia in mice by the P210bcr/abl gene of the Philadelphia chromosome. Science 1990; 247: 824–830.
Michor F, Iwasa Y, Nowak MA . The age incidence of chronic myeloid leukemia can be explained by a one-mutation model. Proc Natl Acad Sci USA 2006; 103: 14931–14934.
Faderl S, Talpaz M, Estrov Z, O’Brien S, Kurzrock R, Kantarjian HM . The biology of chronic myeloid leukemia. N Engl J Med 1999; 341: 164–172.
Goldman JM . Treatment of chronic myeloid leukaemia: some topical questions. Baillieres Clin Haematol 1997; 10: 405–421.
Fausel C . Targeted chronic myeloid leukemia therapy: seeking a cure. J Manag Care Pharm 2007; 13 (8 Suppl A): 8–12.
Savona M, Talpaz M . Chronic myeloid leukemia: changing the treatment paradigms. Oncology (Williston Park) 2006; 20: 707–711; discussion 712–4, 719, 724.
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med 1996; 2: 561–566.
Druker BJ, O’Brien SG, Cortes J, Radich J . Chronic myelogenous leukemia. Hematology (Am Soc Hematol Educ Program) 2002; 1: 111–135.
Deininger MW, Goldman JM, Lydon N, Melo JV . The tyrosine kinase inhibitor CGP57148B selectively inhibits the growth of BCR-ABL-positive cells. Blood 1997; 90: 3691–3698.
Gorre ME, Ellwood-Yen K, Chiosis G, Rosen N, Sawyers CL . BCR-ABL point mutants isolated from patients with imatinib mesylate-resistant chronic myeloid leukemia remain sensitive to inhibitors of the BCR-ABL chaperone heat shock protein 90. Blood 2002; 100: 3041–3044.
Lowenberg B . Minimal residual disease in chronic myeloid leukemia. N Engl J Med 2003; 349: 1399–1401.
Shah NP, Skaggs BJ, Branford S, Hughes TP, Nicoll JM, Paquette RL et al. Sequential ABL kinase inhibitor therapy selects for compound drug-resistant BCR-ABL mutations with altered oncogenic potency. J Clin Invest 2007; 117: 2562–2569.
Casalou C, Fragoso R, Nunes JF, Dias S . VEGF/PLGF induces leukemia cell migration via P38/ERK1/2 kinase pathway, resulting in Rho GTPases activation and caveolae formation. Leukemia 2007; 21: 1590–1594.
Sini P, Cannas A, Koleske AJ, Di Fiore PP, Scita G . Abl-dependent tyrosine phosphorylation of Sos-1 mediates growth-factor-induced Rac activation. Nat Cell Biol 2004; 6: 268–274.
Renshaw MW, Lea-Chou E, Wang JY . Rac is required for v-Abl tyrosine kinase to activate mitogenesis. Curr Biol 1996; 6: 76–83.
Skorski T, Wlodarski P, Daheron L, Salomoni P, Nieborowska-Skorska M, Majewski M et al. BCR/ABL-mediated leukemogenesis requires the activity of the small GTP- binding protein Rac. Proc Natl Acad Sci USA 1998; 95: 11858–11862.
Harnois T, Constantin B, Rioux A, Grenioux E, Kitzis A, Bourmeyster N . Differential interaction and activation of Rho family GTPases by p210bcr-abl and p190bcr-abl. Oncogene 2003; 22: 6445–6454.
Burridge K, Wennerberg K . Rho and Rac take center stage. Cell 2004; 116: 167–179.
Schwartz M . Rho signalling at a glance. J Cell Sci 2004; 117 (Part 23): 5457–5458.
Thomas EK, Cancelas JA, Chae HD, Cox AD, Keller PJ, Perrotti D et al. Rac guanosine triphosphatases represent integrating molecular therapeutic targets for BCR-ABL-induced myeloproliferative disease. Cancer Cell 2007; 12: 467–478.
Blanchard JM . Small GTPases, adhesion, cell cycle control and proliferation. Pathol Biol (Paris) 2000; 48: 318–327.
Gu Y, Filippi MD, Cancelas JA, Siefring JE, Williams EP, Jasti AC et al. Hematopoietic cell regulation by Rac1 and Rac2 guanosine triphosphatases. Science 2003; 302: 445–449.
Turner M, Billadeau DD . VAV proteins as signal integrators for multi-subunit immune-recognition receptors. Nat Rev Immunol 2002; 2: 476–486.
Diaz-Blanco E, Bruns I, Neumann F, Fischer JC, Graef T, Rosskopf M et al. Molecular signature of CD34(+) hematopoietic stem and progenitor cells of patients with CML in chronic phase. Leukemia 2007; 21: 494–504.
Cancelas JA, Lee AW, Prabhakar R, Stringer KF, Zheng Y, Williams DA . Rac GTPases differentially integrate signals regulating hematopoietic stem cell localization. Nat Med 2005; 11: 886–891.
Haataja L, Groffen J, Heisterkamp N . Characterization of RAC3, a novel member of the Rho family. J Biol Chem 1997; 272: 20384–20388.
Cho YJ, Zhang B, Kaartinen V, Haataja L, de Curtis I, Groffen J et al. Generation of rac3 null mutant mice: role of Rac3 in Bcr/Abl-caused lymphoblastic leukemia. Mol Cell Biol 2005; 25: 5777–5785.
Mira JP, Benard V, Groffen J, Sanders LC, Knaus UG . Endogenous, hyperactive Rac3 controls proliferation of breast cancer cells by a p21-activated kinase-dependent pathway. Proc Natl Acad Sci USA 2000; 97: 185–189.
Chan AY, Coniglio SJ, Chuang YY, Michaelson D, Knaus UG, Philips MR et al. Roles of the Rac1 and Rac3 GTPases in human tumor cell invasion. Oncogene 2005; 24: 7821–7829.
Zhang X, Ren R . Bcr-Abl efficiently induces a myeloproliferative disease and production of excess interleukin-3 and granulocyte-macrophage colony-stimulating factor in mice: a novel model for chronic myelogenous leukemia. Blood 1998; 92: 3829–3840.
Pendergast AM, Quilliam LA, Cripe LD, Bassing CH, Dai Z, Li N et al. BCR-ABL-induced oncogenesis is mediated by direct interaction with the SH2 domain of the GRB-2 adaptor protein. Cell 1993; 75: 175–185.
Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG et al. Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway. EMBO J 1994; 13: 764–773.
Sattler M, Mohi MG, Pride YB, Quinnan LR, Malouf NA, Podar K et al. Critical role for Gab2 in transformation by BCR/ABL. Cancer Cell 2002; 1: 479–492.
Oda T, Heaney C, Hagopian JR, Okuda K, Griffin JD, Druker BJ . Crkl is the major tyrosine-phosphorylated protein in neutrophils from patients with chronic myelogenous leukemia. J Biol Chem 1994; 269: 22925–22928.
Pelicci G, Lanfrancone L, Salcini AE, Romano A, Mele S, Grazia Borrello M et al. Constitutive phosphorylation of Shc proteins in human tumors. Oncogene 1995; 11: 899–907.
Wertheim JA, Perera SA, Hammer DA, Ren R, Boettiger D, Pear WS . Localization of BCR-ABL to F-actin regulates cell adhesion but does not attenuate CML development. Blood 2003; 102: 2220–2228.
Klejman A, Schreiner SJ, Nieborowska-Skorska M, Slupianek A, Wilson M, Smithgall TE et al. The Src family kinase Hck couples BCR/ABL to STAT5 activation in myeloid leukemia cells. EMBO J 2002; 21: 5766–5774.
Nishihara H, Maeda M, Oda A, Tsuda M, Sawa H, Nagashima K et al. DOCK2 associates with CrkL and regulates Rac1 in human leukemia cell lines. Blood 2002; 100: 3968–3974.
Sattler M, Verma S, Pride YB, Salgia R, Rohrschneider LR, Griffin JD . SHIP1, an SH2 domain containing polyinositol-5-phosphatase, regulates migration through two critical tyrosine residues and forms a novel signaling complex with DOK1 and CRKL. J Biol Chem 2001; 276: 2451–2458.
Miranda MB, Johnson DE . Signal transduction pathways that contribute to myeloid differentiation. Leukemia 2007; 21: 1363–1377.
Carlesso N, Frank DA, Griffin JD . Tyrosyl phosphorylation and DNA binding activity of signal transducers and activators of transcription (STAT) proteins in hematopoietic cell lines transformed by Bcr/Abl. J Exp Med 1996; 183: 811–820.
Ilaria Jr RL, Van Etten RA . P210 and P190(BCR/ABL) induce the tyrosine phosphorylation and DNA binding activity of multiple specific STAT family members. J Biol Chem 1996; 271: 31704–31710.
Lin TS, Mahajan S, Frank DA . STAT signaling in the pathogenesis and treatment of leukemias. Oncogene 2000; 19: 2496–2504.
Hoover RR, Gerlach MJ, Koh EY, Daley GQ . Cooperative and redundant effects of STAT5 and Ras signaling in BCR/ABL transformed hematopoietic cells. Oncogene 2001; 20: 5826–5835.
Spiekermann K, Pau M, Schwab R, Schmieja K, Franzrahe S, Hiddemann W . Constitutive activation of STAT3 and STAT5 is induced by leukemic fusion proteins with protein tyrosine kinase activity and is sufficient for transformation of hematopoietic precursor cells. Exp Hematol 2002; 30: 262–271.
Nieborowska-Skorska M, Wasik MA, Slupianek A, Salomoni P, Kitamura T, Calabretta B et al. Signal transducer and activator of transcription (STAT)5 activation by BCR/ABL is dependent on intact Src homology (SH)3 and SH2 domains of BCR/ABL and is required for leukemogenesis. J Exp Med 1999; 189: 1229–1242.
Sillaber C, Gesbert F, Frank DA, Sattler M, Griffin JD . STAT5 activation contributes to growth and viability in Bcr/Abl-transformed cells. Blood 2000; 95: 2118–2125.
Wang Y, Cai D, Brendel C, Barett C, Erben P, Manley PW et al. Adaptive secretion of granulocyte-macrophage colony-stimulating factor (GM-CSF) mediates imatinib and nilotinib resistance in BCR/ABL+ progenitors via JAK-2/STAT-5 pathway activation. Blood 2007; 109: 2147–2155.
Sexl V, Piekorz R, Moriggl R, Rohrer J, Brown MP, Bunting KD et al. Stat5a/b contribute to interleukin 7-induced B-cell precursor expansion, but abl- and bcr/abl-induced transformation are independent of stat5. Blood 2000; 96: 2277–2283.
Hoelbl A, Kovacic B, Kerenyi MA, Simma O, Warsch W, Cui Y et al. Clarifying the role of Stat5 in lymphoid development and Abelson-induced transformation. Blood 2006; 107: 4898–4906.
Gao Y, Dickerson JB, Guo F, Zheng J, Zheng Y . Rational design and characterization of a Rac GTPase-specific small molecule inhibitor. Proc Natl Acad Sci USA 2004; 101: 7618–7623.
Yang FC, Atkinson SJ, Gu Y, Borneo JB, Roberts AW, Zheng Y et al. Rac and Cdc42 GTPases control hematopoietic stem cell shape, adhesion, migration, and mobilization. Proc Natl Acad Sci USA 2001; 98: 5614–5618.
Reya T, Morrison SJ, Clarke MF, Weissman IL . Stem cells, cancer, and cancer stem cells. Nature 2001; 414: 105–111.
Williams DA, Cancelas JA . Leukaemia: niche retreats for stem cells. Nature 2006; 444: 827–828.
Krause DS, Lazarides K, von Andrian UH, Van Etten RA . Requirement for CD44 in homing and engraftment of BCR-ABL-expressing leukemic stem cells. Nat Med 2006; 12: 1175–1180.
Jin L, Hope KJ, Zhai Q, Smadja-Joffe F, Dick JE . Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 2006; 12: 1167–1174.
Gaiger A, Henn T, Horth E, Geissler K, Mitterbauer G, Maier-Dobersberger T et al. Increase of bcr-abl chimeric mRNA expression in tumor cells of patients with chronic myeloid leukemia precedes disease progression. Blood 1995; 86: 2371–2378.
Lin CS, Lim SK, D’Agati V, Costantini F . Differential effects of an erythropoietin receptor gene disruption on primitive and definitive erythropoiesis. Genes Dev 1996; 10: 154–164.
Elmaagacli AH, Beelen DW, Opalka B, Seeber S, Schaefer UW . The amount of BCR-ABL fusion transcripts detected by the real-time quantitative polymerase chain reaction method in patients with Philadelphia chromosome positive chronic myeloid leukemia correlates with the disease stage. Ann Hematol 2000; 79: 424–431.
Guo JQ, Wang JY, Arlinghaus RB . Detection of BCR-ABL proteins in blood cells of benign phase chronic myelogenous leukemia patients. Cancer Res 1991; 51: 3048–3051.
Schultheis B, Szydlo R, Mahon FX, Apperley JF, Melo JV . Analysis of total phosphotyrosine levels in CD34+ cells from CML patients to predict the response to imatinib mesylate treatment. Blood 2005; 105: 4893–4894.
Bhatia R, Holtz M, Niu N, Gray R, Snyder DS, Sawyers CL et al. Persistence of malignant hematopoietic progenitors in chronic myelogenous leukemia patients in complete cytogenetic remission following imatinib mesylate treatment. Blood 2003; 101: 4701–4707.
Copland M, Hamilton A, Elrick LJ, Baird JW, Allan EK, Jordanides N et al. Dasatinib (BMS-354825) targets an earlier progenitor population than imatinib in primary CML but does not eliminate the quiescent fraction. Blood 2006; 107: 4532–4539.
Jamieson CH, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL et al. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N Engl J Med 2004; 351: 657–667.
Deininger MW . Optimizing therapy of chronic myeloid leukemia. Exp Hematol 2007; 35 (4 Suppl 1): 144–154.
Melo JV, Barnes DJ . Chronic myeloid leukaemia as a model of disease evolution in human cancer. Nat Rev Cancer 2007; 7: 441–453.
Chu S, Xu H, Shah NP, Snyder DS, Forman SJ, Sawyers CL et al. Detection of BCR-ABL kinase mutations in CD34+ cells from chronic myelogenous leukemia patients in complete cytogenetic remission on imatinib mesylate treatment. Blood 2005; 105: 2093–2098.
Acknowledgements
This work was supported by the National Institute of Health grant numbers HL69974 and DK62757 (DAW), Leukemia Lymphoma Society grant 6152-06 (DAW), T32 HD046387 (EKT) and the Department of Defense New Investigator Award CM064050 (JAC). DAW and YZ may obtain royalties based on milestones set forth in a licensing agreement between Cincinnati Children’s Hospital Medical Center and Amgen related to the development of drug inhibitors of Rac GTPases.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thomas, E., Cancelas, J., Zheng, Y. et al. Rac GTPases as key regulators of p210-BCR-ABL-dependent leukemogenesis. Leukemia 22, 898–904 (2008). https://doi.org/10.1038/leu.2008.71
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.71
Keywords
This article is cited by
-
Inhibition of the RacGEF VAV3 by the small molecule IODVA1 impedes RAC signaling and overcomes resistance to tyrosine kinase inhibition in acute lymphoblastic leukemia
Leukemia (2022)
-
Phosphorylation of SOS1 on tyrosine 1196 promotes its RAC GEF activity and contributes to BCR-ABL leukemogenesis
Leukemia (2018)
-
Mtss1 is a critical epigenetically regulated tumor suppressor in CML
Leukemia (2016)
-
Helicobacter pylori-induced gastric mucosal TGF-α ectodomain shedding and EGFR transactivation involves Rac1/p38 MAPK-dependent TACE activation
Inflammopharmacology (2016)
-
PAK-dependent STAT5 serine phosphorylation is required for BCR-ABL-induced leukemogenesis
Leukemia (2014)