Abstract
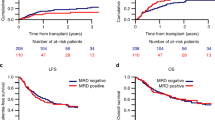
The prognosis for children with high-risk relapsed acute lymphoblastic leukemia (ALL) is poor. Here, we assessed the prognostic importance of response during induction and consolidation treatment prior to hematopoietic stem cell transplantation (HSCT) aiming to evaluate the best time to assess minimal residual disease (MRD) for intervention strategies and in future trials in high-risk ALL relapse patients. Included patients (n=125) were treated uniformly according to the ALL-REZ BFM (Berlin-Frankfurt-Münster) 2002 relapse trial (median follow-up time=4.8 years). Patients with MRD ⩾10−3 after induction treatment (76/119, 64%) or immediately preceding HSCT (19/71, 27%) had a significantly worse probability of disease-free survival 10 years after relapse treatment begin, with 26% (±6%) or 23% (±7%), respectively, compared with 58% (±8%) or 48% (±7%) for patients with MRD <10−3. Conventional intensive consolidation treatment reduced MRD to <10−3 before HSCT in 63% of patients, whereas MRD remained high or increased in the rest of this patient group. Our data support that MRD after induction treatment can be used to quantify the activity of different induction treatment strategies in phase II trials. MRD persistence at ⩾10−3 before HSCT reflects a disease highly resistant to conventional intensive chemotherapy and requiring prospective controlled investigation of new treatment strategies and drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Borgmann A, von Stackelberg A, Hartmann R, Ebell W, Klingebiel T, Peters C et al. Unrelated donor stem cell transplantation compared with chemotherapy for children with acute lymphoblastic leukemia in a second remission: a matched-pair analysis. Blood 2003; 101: 3835–3839.
Gaynon PS . Childhood acute lymphoblastic leukaemia and relapse. Br J Haematol 2005; 131: 579–587.
Nguyen K, Devidas M, Cheng SC, La M, Raetz EA, Carroll WL et al. Factors influencing survival after relapse from acute lymphoblastic leukemia: a Children's Oncology Group study. Leukemia 2008; 22: 2142–2150.
Parker C, Waters R, Leighton C, Hancock J, Sutton R, Moorman AV et al. Effect of mitoxantrone on outcome of children with first relapse of acute lymphoblastic leukaemia (ALL R3): an open-label randomised trial. Lancet 2010; 376: 2009–2017.
Bailey LC, Lange BJ, Rheingold SR, Bunin NJ . Bone-marrow relapse in paediatric acute lymphoblastic leukaemia. Lancet Oncol 2008; 9: 873–883.
Henze G, v Stackelberg A, Eckert C . ALL-REZ BFM—the consecutive trials for children with relapsed acute lymphoblastic leukemia. Klinische Padiatrie 2013; 225: S73–S78.
Mehta PA, Davies SM . Allogeneic transplantation for childhood ALL. Bone Marrow Transplant 2008; 41: 133–139.
Einsiedel HG, von Stackelberg A, Hartmann R, Fengler R, Schrappe M, Janka-Schaub G et al. Long-term outcome in children with relapsed ALL by risk-stratified salvage therapy: results of trial acute lymphoblastic leukemia-relapse study of the Berlin-Frankfurt-Munster Group 87. J Clin Oncol 2005; 23: 7942–7950.
Leung W, Campana D, Yang J, Pei D, Coustan-Smith E, Gan K et al. High success rate of hematopoietic cell transplantation regardless of donor source in children with very high-risk leukemia. Blood 2011; 118: 223–230.
Tallen G, Ratei R, Mann G, Kaspers G, Niggli F, Karachunsky A et al. Long-term outcome in children with relapsed acute lymphoblastic leukemia after time-point and site-of-relapse stratification and intensified short-course multidrug chemotherapy: results of trial ALL-REZ BFM 90. J Clin Oncol 2010; 28: 2339–2347.
Bader P, Kreyenberg H, Henze GH, Eckert C, Reising M, Willasch A et al. Prognostic value of minimal residual disease quantification before allogeneic stem-cell transplantation in relapsed childhood acute lymphoblastic leukemia: The ALL-REZ BFM Study Group. J Clin Oncol 2009; 27: 377–384.
Leung W, Pui CH, Coustan-Smith E, Yang J, Pei D, Gan K et al. Detectable minimal residual disease before hematopoietic cell transplantation is prognostic but does not preclude cure for children with very-high-risk leukemia. Blood 2012; 120: 468–472.
Paganin M, Zecca M, Fabbri G, Polato K, Biondi A, Rizzari C et al. Minimal residual disease is an important predictive factor of outcome in children with relapsed 'high-risk' acute lymphoblastic leukemia. Leukemia 2008; 22: 2193–2200.
Raetz EA, Borowitz MJ, Devidas M, Linda SB, Hunger SP, Winick NJ et al. Reinduction platform for children with first marrow relapse of acute lymphoblastic Leukemia: A Children's Oncology Group Study. J Clin Oncol 2008; 26: 3971–3978.
Bene MC, Bernier M, Casasnovas RO, Castoldi G, Knapp W, Lanza F et al. The reliability and specificity of c-kit for the diagnosis of acute myeloid leukemias and undifferentiated leukemias. The European Group for the Immunological Classification of Leukemias (EGIL). Blood 1998; 92: 596–599.
Bene MC, Castoldi G, Knapp W, Ludwig WD, Matutes E, Orfao A et al. Proposals for the immunological classification of acute leukemias. European Group for the Immunological Characterization of Leukemias (EGIL). Leukemia 1995; 9: 1783–1786.
Borowitz M, Bene M-C, Harris N, Porwit A, Matutes E . Acute leukemias of ambiguous lineage. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. Lyon: IARC 2008, 149–155.
Henze G Charite University Berlin, Germany. ALL-REZ BFM 2002: Multi-Center Study for Children With Relapsed Acute Lymphoblastic Leukemia. In. ClinicalTrialsgov [Internet] Bethesda (MD): National Library of Medicine (US) 2000 (cited 18 July 2009) available from http://clinicaltrialsgov/ct2/show/NCT00114348, NLM identifier: NCT00114348 2005.
Schrauder A, von Stackelberg A, Schrappe M, Cornish J, Peters C . Allogeneic hematopoietic SCT in children with ALL: current concepts of ongoing prospective SCT trials. Bone Marrow Transplant 2008; 41: S71–S74.
Eckert C, von Stackelberg A, Seeger K, Groeneveld TW, Peters C, Klingebiel T et al. Minimal residual disease after induction is the strongest predictor of prognosis in intermediate risk relapsed acute lymphoblastic leukaemia - long-term results of trial ALL-REZ BFM P95/96. Eur J Cancer 2013; 49: 1346–1355.
Langerak AW, Szczepanski T, van der Burg M, Wolvers-Tettero IL, van Dongen JJ . Heteroduplex PCR analysis of rearranged T cell receptor genes for clonality assessment in suspect T cell proliferations. Leukemia 1997; 11: 2192–2199.
Pongers-Willemse MJ, Seriu T, Stolz F, d'Aniello E, Gameiro P, Pisa P et al. Primers and protocols for standardized detection of minimal residual disease in acute lymphoblastic leukemia using immunoglobulin and T cell receptor gene rearrangements and TAL1 deletions as PCR targets: report of the BIOMED-1 CONCERTED ACTION: investigation of minimal residual disease in acute leukemia. Leukemia 1999; 13: 110–118.
Szczepanski T, Pongers-Willemse MJ, Langerak AW, Harts WA, Wijkhuijs AJ, van Wering ER et al. Ig heavy chain gene rearrangements in T-cell acute lymphoblastic leukemia exhibit predominant DH6-19 and DH7-27 gene usage, can result in complete V-D-J rearrangements, and are rare in T-cell receptor alpha beta lineage. Blood 1999; 93: 4079–4085.
Szczepanski T, van der Velden VH, Hoogeveen PG, de Bie M, Jacobs DC, van Wering ER et al. Vdelta2-Jalpha rearrangements are frequent in precursor-B-acute lymphoblastic leukemia but rare in normal lymphoid cells. Blood 2004; 103: 3798–3804.
Eckert C, Landt O . Real-time PCR to detect minimal residual disease in childhood ALL. Methods Mol Med 2004; 91: 175–182.
van der Velden VH, Cazzaniga G, Schrauder A, Hancock J, Bader P, Panzer-Grumayer ER et al. Analysis of minimal residual disease by Ig/TCR gene rearrangements: guidelines for interpretation of real-time quantitative PCR data. Leukemia 2007; 21: 604–611.
Pocock SJ, Clayton TC, Altman DG . Survival plots of time-to-event outcomes in clinical trials: good practice and pitfalls. Lancet 2002; 359: 1686–1689.
Gray RJ . A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat 1988; 16: 1141–1154.
Kalbfleisch JD, Prentice RL . The Statistical Analysis of Failure Time Data. Wiley: New York, NY, USA, 1980, 168–189.
Goulden N, Bader P, Van Der Velden V, Moppett J, Schilham M, Masden HO et al. Minimal residual disease prior to stem cell transplant for childhood acute lymphoblastic leukaemia. Br J Haematol 2003; 122: 24–29.
Knechtli CJ, Goulden NJ, Hancock JP, Grandage VL, Harris EL, Garland RJ et al. Minimal residual disease status before allogeneic bone marrow transplantation is an important determinant of successful outcome for children and adolescents with acute lymphoblastic leukemia. Blood 1998; 92: 4072–4079.
Krejci O, van der Velden VH, Bader P, Kreyenberg H, Goulden N, Hancock J et al. Level of minimal residual disease prior to haematopoietic stem cell transplantation predicts prognosis in paediatric patients with acute lymphoblastic leukaemia: a report of the Pre-BMT MRD Study Group. Bone Marrow Transplant 2003; 32: 849–851.
Sramkova L, Muzikova K, Fronkova E, Krejci O, Sedlacek P, Formankova R et al. Detectable minimal residual disease before allogeneic hematopoietic stem cell transplantation predicts extremely poor prognosis in children with acute lymphoblastic leukemia. Pediatr Blood Cancer 2007; 48: 93–100.
Balduzzi A, Di Maio L, Silvestri D, Songia S, Bonanomi S, Rovelli A et al. Minimal residual disease before and after transplantation for childhood acute lymphoblastic leukaemia: is there any room for intervention? Br J Haematol 2014; 164: 396–408.
Brentjens RJ, Riviere I, Park JH, Davila ML, Wang X, Stefanski J et al. Safety and persistence of adoptively transferred autologous CD19-targeted T cells in patients with relapsed or chemotherapy refractory B-cell leukemias. Blood 2011; 118: 4817–4828.
Handgretinger R, Zugmaier G, Henze G, Kreyenberg H, Lang P, von Stackelberg A . Complete remission after blinatumomab-induced donor T-cell activation in three pediatric patients with post-transplant relapsed acute lymphoblastic leukemia. Leukemia 2011; 25: 181–184.
Klinger M, Brandl C, Zugmaier G, Hijazi Y, Bargou RC, Topp MS et al. Immunopharmacologic response of patients with B-lineage acute lymphoblastic leukemia to continuous infusion of T cell-engaging CD19/CD3-bispecific BiTE antibody blinatumomab. Blood 2012; 119: 6226–6233.
Acknowledgements
The authors heartily thank all patients, their families and treating physicians who participated in this study. We thank our molecular genetics laboratory technicians for their excellent work, PD Dr Richard Ratei and his laboratory for immunophenotyping and Andrea Kretschmann in the ALL-REZ BFM trial center for reliable comprehensive data management. We thank the Deutsche Kinderkrebsstiftung for financing the ALL-REZ BFM 2002 clinical trial and MRD studies in Germany, the Pediatric Oncology Competence Network for collaboration on frontline MRD studies (supported by the German Federal Ministry of Education and Research), the Deutsche José Carreras Leukämie-Stiftung e.V. for supporting the international principal investigator, and the GACR Centre of Excellence (#P302/12/G101) and MH CZ – DRO, University Hospital Motol (Prague, #00064203) for supporting the study in the Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Eckert, C., Hagedorn, N., Sramkova, L. et al. Monitoring minimal residual disease in children with high-risk relapses of acute lymphoblastic leukemia: prognostic relevance of early and late assessment. Leukemia 29, 1648–1655 (2015). https://doi.org/10.1038/leu.2015.59
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2015.59
This article is cited by
-
Measurable residual disease (MRD)-testing in haematological and solid cancers
Leukemia (2024)
-
Nelarabine-containing salvage therapy and conditioning regimen in transplants for pediatric T-cell acute lymphoblastic leukemia and lymphoma
International Journal of Hematology (2024)
-
Blinatumomab therapy for B cell acute lymphoblastic leukemia accompanied by persistent or relapsed low-level MRD prior to hematopoietic stem cell transplantation in Chinese children: a case series
Bulletin of the National Research Centre (2023)
-
Phase 2 study of combination chemotherapy with bortezomib in children with relapsed and refractory acute lymphoblastic leukemia
International Journal of Hematology (2023)
-
Blinatumomab to improve the outcome of children with relapsed B-cell acute lymphoblastic leukemia
Clinical and Translational Oncology (2021)