Abstract
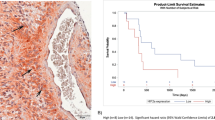
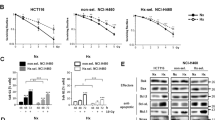
The hypoxic bone marrow (BM) microenvironment confers growth/survival and drug resistance in multiple myeloma (MM) cells. Novel therapies targeting the MM cell in its hypoxic BM milieu may overcome drug resistance. Recent studies led to the development of a novel molecule RRx-001 with hypoxia-selective epigenetic and nitric oxide-donating properties. Here, we demonstrate that RRx-001 decreases the viability of MM cell lines and primary patient cells, as well as overcomes drug resistance. RRx-001 inhibits MM cell growth in the presence of BM stromal cells. RRx-001-induced apoptosis is associated with: (i) activation of caspases; (ii) release of ROS and nitrogen species; (iii) induction of DNA damage via ATM/γ-H2AX; and (iv) decrease in DNA methyltransferase (DNMT) and global methylation. RNA interference study shows a predominant role of DNMT1 in MM cell survival versus DNMT3a or DNMT3b. The deubiquitylating enzyme USP7 stimulates DNMT1 activity, and conversely, USP7-siRNA reduced DNMT1 activity and decreased MM cell viability. RRx-001 plus USP7 inhibitor P5091 triggered synergistic anti-MM activity. MM xenograft studies show that RRx-001 is well tolerated, inhibits tumor growth and enhances survival. Combining RRx-001 with pomalidomide, bortezomib or SAHA induces synergistic anti-MM activity. Our results provide the rationale for translation of RRx-001, either alone or in combination, to clinical evaluation in MM.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Anderson KC . Oncogenomics to target myeloma in the bone marrow microenvironment. Clin Cancer Res 2011; 17: 1225–1233.
Dimopoulos M, Kyle R, Fermand JP, Rajkumar SV, San Miguel J, Chanan-Khan A et al. Consensus recommendations for standard investigative workup: report of the International Myeloma Workshop Consensus Panel 3. Blood 2011; 117: 4701–4705.
Colla S, Storti P, Donofrio G, Todoerti K, Bolzoni M, Lazzaretti M et al. Low bone marrow oxygen tension and hypoxia-inducible factor-1alpha overexpression characterize patients with multiple myeloma: role on the transcriptional and proangiogenic profiles of CD138(+) cells. Leukemia 2010; 24: 1967–1970.
Walker BA, Wardell CP, Chiecchio L, Smith EM, Boyd KD, Neri A et al. Aberrant global methylation patterns affect the molecular pathogenesis and prognosis of multiple myeloma. Blood 2011; 117: 553–562.
Azab AK, Hu J, Quang P, Azab F, Pitsillides C, Awwad R et al. Hypoxia promotes dissemination of multiple myeloma through acquisition of epithelial to mesenchymal transition-like features. Blood 2012; 119: 5782–5794.
Hu J, Van Valckenborgh E, Menu E, De Bruyne E, Vanderkerken K . Understanding the hypoxic niche of multiple myeloma: therapeutic implications and contributions of mouse models. Dis Model Mech 2012; 5: 763–771.
Baylin SB . DNA methylation and gene silencing in cancer. Nat Clin Pract Oncol 2005; 2 (Suppl 1): S4–S11.
Kondo Y . Epigenetic cross-talk between DNA methylation and histone modifications in human cancers. Yonsei Med J 2009; 50: 455–463.
Ng MH, Chung YF, Lo KW, Wickham NW, Lee JC, Huang DP . Frequent hypermethylation of p16 and p15 genes in multiple myeloma. Blood 1997; 89: 2500–2506.
Dimopoulos K, Gimsing P, Gronbaek K . The role of epigenetics in the biology of multiple myeloma. Blood Cancer J 2014; 4: e207.
Heuck CJ, Mehta J, Bhagat T, Gundabolu K, Yu Y, Khan S et al. Myeloma is characterized by stage-specific alterations in DNA methylation that occur early during myelomagenesis. J Immunol 2013; 190: 2966–2975.
Maes K, Menu E, Van Valckenborgh E, Van Riet I, Vanderkerken K, De Bruyne E . Epigenetic modulating agents as a new therapeutic approach in multiple myeloma. Cancers 2013; 5: 430–461.
Chin HG, Esteve PO, Pradhan M, Benner J, Patnaik D, Carey MF et al. Automethylation of G9a and its implication in wider substrate specificity and HP1 binding. Nucleic Acids Res 2007; 35: 7313–7323.
de Carvalho F, Colleoni GW, Almeida MS, Carvalho AL, Vettore AL . TGFbetaR2 aberrant methylation is a potential prognostic marker and therapeutic target in multiple myeloma. Int J Cancer 2009; 125: 1985–1991.
Tiedemann RL, Putiri EL, Lee JH, Hlady RA, Kashiwagi K, Ordog T et al. Acute depletion redefines the division of labor among DNA methyltransferases in methylating the human genome. Cell Rep 2014; 9: 1554–1566.
Oronsky B, Oronsky N, Knox S, Fanger G, Scicinski J . Episensitization: therapeutic tumor resensitization by epigenetic agents: a review and reassessment. Anticancer Agents Med Chem 2014; 14: 1121–1127.
Ning S, Bednarski M, Oronsky B, Scicinski J, Saul G, Knox SJ . Dinitroazetidines are a novel class of anticancer agents and hypoxia-activated radiation sensitizers developed from highly energetic materials. Cancer Res 2012; 72: 2600–2608.
Oronsky B, Oronsky N, Scicinski J, Fanger G, Lybeck M, Reid T . Rewriting the epigenetic code for tumor resensitization: a review. Transl Oncol 2014; 7: 626–631.
Scicinski J, Oronsky B, Ning S, Knox S, Peehl D, Kim MM et al. NO to cancer: The complex and multifaceted role of nitric oxide and the epigenetic nitric oxide donor, RRx-001. Redox Biol 2015; 6: 1–8.
Reid T, Oronsky B, Scicinski J, Scribner CL, Knox SJ, Ning S et al. Safety and activity of RRx-001 in patients with advanced cancer: a first-in-human, open-label, dose-escalation phase 1 study. Lancet Oncol 2015; 16: 1133–1142.
Chauhan D, Singh AV, Brahmandam M, Carrasco R, Bandi M, Hideshima T et al. Functional interaction of plasmacytoid dendritic cells with multiple myeloma cells: a therapeutic target. Cancer Cell 2009; 16: 309–323.
Chauhan D, Tian Z, Nicholson B, Kumar KG, Zhou B, Carrasco R et al. A small molecule inhibitor of ubiquitin-specific protease-7 induces apoptosis in multiple myeloma cells and overcomes bortezomib resistance. Cancer Cell 2012; 22: 345–358.
Das DS, Ray A, Song Y, Richardson P, Trikha M, Chauhan D et al. Synergistic anti-myeloma activity of the proteasome inhibitor marizomib and the IMiD immunomodulatory drug pomalidomide. Br J Haematol 2015; 171: 798–812.
Chauhan D, Singh A, Brahmandam M, Podar K, Hideshima T, Richardson P et al. Combination of proteasome inhibitors bortezomib and NPI-0052 trigger in vivo synergistic cytotoxicity in multiple myeloma. Blood 2008; 111: 1654–1664.
Chauhan D, Singh AV, Ciccarelli B, Richardson PG, Palladino MA, Anderson KC . Combination of novel proteasome inhibitor NPI-0052 and lenalidomide trigger in vitro and in vivo synergistic cytotoxicity in multiple myeloma. Blood 2010; 115: 834–845.
Chou TC, Talalay P . Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 1984; 22: 27–55.
Giuliani N, Storti P, Bolzoni M, Palma BD, Bonomini S . Angiogenesis and multiple myeloma. Cancer Microenviron 2011; 4: 325–337.
Podar K, Tai YT, Davies FE, Lentzsch S, Sattler M, Hideshima T et al. Vascular endothelial growth factor triggers signaling cascades mediating multiple myeloma cell growth and migration. Blood 2001; 98: 428–435.
Chauhan D, Uchiyama H, Akbarali Y, Urashima M, Yamamoto K, Libermann TA et al. Multiple myeloma cell adhesion-induced interleukin-6 expression in bone marrow stromal cells involves activation of NF-kappa B. Blood 1996; 87: 1104–1112.
Anderson KC . Targeted therapy of multiple myeloma based upon tumor-microenvironmental interactions. Exp Hematol 2007; 35 (4 Suppl 1): 155–162.
Ray A, Tian Z, Das DS, Coffman RL, Richardson P, Chauhan D et al. A novel TLR-9 agonist C792 inhibits plasmacytoid dendritic cell-induced myeloma cell growth and enhance cytotoxicity of bortezomib. Leukemia 2014; 28: 1716–1724.
Ray A, Das DS, Song Y, Chauhan PGR, D, K.C A . Targeting PD1-PDL1 immune checkpoint in plasmacytoid dendritic cells interactions with T cells, natural killer cells, and multiple myeloma cells. Leukemia 2015; 29: 1441–1444.
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC . Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE. Nature 1994; 371: 346–347.
Hockenbery DM, Oltvai ZN, Yin XM, Milliman CL, Korsmeyer SJ . Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 1993; 75: 241–251.
Scicinski J, Oronsky B, Taylor M, Luo G, Musick T, Marini J et al. Preclinical evaluation of the metabolism and disposition of RRx-001, a novel investigative anticancer agent. Drug Metab Dispos 2012; 40: 1810–1816.
Scatena R, Bottoni P, Pontoglio A, Giardina B . Pharmacological modulation of nitric oxide release: new pharmacological perspectives, potential benefits and risks. Curr Med Chem 2010; 17: 61–73.
Wang Z . Protein S-nitrosylation and cancer. Cancer Lett 2012; 320: 123–129.
Monteiro HP, Costa PE, Reis AK, Stern A . Nitric oxide: protein tyrosine phosphorylation and protein S-nitrosylation in cancer. Biomed J 2015; 38: 380–388.
Hess DT, Matsumoto A, Kim SO, Marshall HE, Stamler JS . Protein S-nitrosylation: purview and parameters. Nat Rev Mol Cell Biol 2005; 6: 150–166.
Hironaka K, Factor VM, Calvisi DF, Conner EA, Thorgeirsson SS . Dysregulation of DNA repair pathways in a transforming growth factor alpha/c-myc transgenic mouse model of accelerated hepatocarcinogenesis. Lab Invest 2003; 83: 643–654.
Kang MA, So EY, Simons AL, Spitz DR, Ouchi T . DNA damage induces reactive oxygen species generation through the H2AX-Nox1/Rac1 pathway. Cell Death Dis 2012; 3: e249.
Burma S, Chen BP, Murphy M, Kurimasa A, Chen DJ . ATM phosphorylates histone H2AX in response to DNA double-strand breaks. J Biol Chem 2001; 276: 42462–42467.
Cottini F, Hideshima T, Suzuki R, Tai YT, Bianchini G, Richardson PG et al. Synthetic lethal approaches exploiting DNA damage in aggressive myeloma. Cancer Discov 2015; 5: 972–987.
Lee BH, Yegnasubramanian S, Lin X, Nelson WG . Procainamide is a specific inhibitor of DNA methyltransferase 1. J Biol Chem 2005; 280: 40749–40756.
Zhou W, Chen H, Hong X, Niu X, Lu Q . Knockdown of DNA methyltransferase-1 inhibits proliferation and derepresses tumor suppressor genes in myeloma cells. Oncol Lett 2014; 8: 2130–2134.
Kiziltepe T, Hideshima T, Catley L, Raje N, Yasui H, Shiraishi N et al. 5-Azacytidine, a DNA methyltransferase inhibitor, induces ATR-mediated DNA double-strand break responses, apoptosis, and synergistic cytotoxicity with doxorubicin and bortezomib against multiple myeloma cells. Mol Cancer Ther 2007; 6: 1718–1727.
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J et al. STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 2015; 43: D447–D452.
Felle M, Joppien S, Nemeth A, Diermeier S, Thalhammer V, Dobner T et al. The USP7/Dnmt1 complex stimulates the DNA methylation activity of Dnmt1 and regulates the stability of UHRF1. Nucleic Acids Res 2011; 39: 8355–8365.
Bergsagel PL, Kuehl WM . Molecular pathogenesis and a consequent classification of multiple myeloma. J Clin Oncol 2005; 23: 6333–6338.
Bergsagel PL, Chesi M, Nardini E, Brents LA, Kirby SL, Kuehl WM . Promiscuous translocations into immunoglobulin heavy chain switch regions in multiple myeloma. Proc Natl Acad Sci USA 1996; 93: 13931–13936.
Du Z, Song J, Wang Y, Zhao Y, Guda K, Yang S et al. DNMT1 stability is regulated by proteins coordinating deubiquitination and acetylation-driven ubiquitination. Sci Signal 2010; 3: ra80.
Kuhn D, Bjorklund C, Magarotto V, Mathews J, Wang M, Baladandayuthapani V et al. Bortezomib resistance is mediated by increased signaling through the insulin-like growth factor-1/Akt axis. ASH Annual Meeting Abstracts 2009; 114: 2739.
Magesh S, Chen Y, Hu L . Small molecule modulators of Keap1-Nrf2-ARE pathway as potential preventive and therapeutic agents. Med Res Rev 2012; 32: 687–726.
Ning S, Sekar TV, Scicinski J, Oronsky B, Peehl DM, Knox SJ et al. Nrf2 activity as a potential biomarker for the pan-epigenetic anticancer agent, RRx-001. Oncotarget 2015; 6: 21547–21556.
Afanas'ev I . New nucleophilic mechanisms of ros-dependent epigenetic modifications: comparison of aging and cancer. Aging Dis 2014; 5: 52–62.
Bollati V, Fabris S, Pegoraro V, Ronchetti D, Mosca L, Deliliers GL et al. Differential repetitive DNA methylation in multiple myeloma molecular subgroups. Carcinogenesis 2009; 30: 1330–1335.
Kaiser MF, Johnson DC, Wu P, Walker BA, Brioli A, Mirabella F et al. Global methylation analysis identifies prognostically important epigenetically inactivated tumor suppressor genes in multiple myeloma. Blood 2013; 122: 219–226.
Walton EL, Francastel C, Velasco G . Maintenance of DNA methylation: Dnmt3b joins the dance. Epigenetics 2011; 6: 1373–1377.
Wang X, Zhang L, Ding N, Yang X, Zhang J, He J et al. Identification and characterization of DNAzymes targeting DNA methyltransferase I for suppressing bladder cancer proliferation. Biochem Biophys Res Commun 2015; 461: 329–333.
van der Horst A, de Vries-Smits AM, Brenkman AB, van Triest MH, van den Broek N, Colland F et al. FOXO4 transcriptional activity is regulated by monoubiquitination and USP7/HAUSP. Nat Cell Biol 2006; 8: 1064–1073.
Acknowledgements
This investigation was supported by National Institutes of Health Specialized Programs of Research Excellence (SPORE) grant P50100707, PO1-CA078378, and RO1 CA050947. KCA is an American Cancer Society Clinical Research Professor.
Author contributions
DSD designed and performed the experiments, interpreted data and wrote the manuscript; AD helped in acquiring confocal images; AR and YS helped with animal experiments; ZT helped with viability assays, PR contributed clinical samples; BO and JS reviewed the manuscript; DC designed research, analyzed data and wrote the manuscript; KCA analyzed data and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
BO and JS are employees of EpicentRx; KCA is on Advisory board of Celgene, Millenium, Gilead and Sanofi Aventis, and is a Scientific founder of Oncopep and acetylon; DC is consultant to EpicentRx Inc. The remaining authors have no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Das, D., Ray, A., Das, A. et al. A novel hypoxia-selective epigenetic agent RRx-001 triggers apoptosis and overcomes drug resistance in multiple myeloma cells. Leukemia 30, 2187–2197 (2016). https://doi.org/10.1038/leu.2016.96
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.96
This article is cited by
-
Targeting ubiquitin specific proteases (USPs) in cancer immunotherapy: from basic research to preclinical application
Journal of Experimental & Clinical Cancer Research (2023)
-
A Review of RRx-001: A Late-Stage Multi-Indication Inhibitor of NLRP3 Activation and Chronic Inflammation
Drugs (2023)
-
Deubiquitinases in hematological malignancies
Biomarker Research (2021)
-
Targeting aberrant DNA methylation in mesenchymal stromal cells as a treatment for myeloma bone disease
Nature Communications (2021)
-
Ubiquitin-specific protease 7 is a drug-able target that promotes hepatocellular carcinoma and chemoresistance
Cancer Cell International (2020)