Abstract
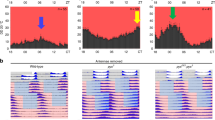
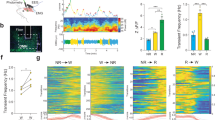
In Drosophila, a ‘clock’ situated in the brain controls circadian rhythms of locomotor activity. This clock relies on several groups of neurons that express the Period (PER) protein, including the ventral lateral neurons (LNvs), which express the Pigment-dispersing factor (PDF) neuropeptide, and the PDF-negative dorsal lateral neurons (LNds)1. In normal cycles of day and night, adult flies exhibit morning and evening peaks of activity1,2; however, the contribution of the different clock neurons to the rest–activity pattern remains unknown. Here, we have used targeted expression of PER to restore the clock function of specific subsets of lateral neurons in arrhythmic per0 mutant flies. We show that PER expression restricted to the LNvs only restores the morning activity, whereas expression of PER in both the LNvs and LNds also restores the evening activity. This provides the first neuronal bases for ‘morning’ and ‘evening’ oscillators in the Drosophila brain. Furthermore, we show that the LNvs alone can generate 24 h activity rhythms in constant darkness, indicating that the morning oscillator is sufficient to drive the circadian system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hall, J. C. Genetics and molecular biology of rhythms in Drosophila and other insects. Adv. Genet. 48, 1–280 (2003)
Helfrich-Förster, C. Differential control of morning and evening components in the activity rhythm of Drosophila melanogaster—sex-specific differences suggest a different quality of activity. J. Biol. Rhythms 15, 135–154 (2000)
Dunlap, J., Loros, J. & DeCoursey, P. J. (eds) Chronobiology. Biological Timekeeping (Sinauer Associates, Sunderland, Massachusetts, 2004)
Pittendrigh, C. & Daan, S. A Functional analysis of circadian pacemakers in nocturnal rodents. V. Pacemaker structure: a clock for all seasons. J. Comp. Physiol. A 106, 333–335 (1976)
de la Iglesia, H. O., Meyer, J., Carpino, A. & Schwartz, W. J. Antiphase oscillation of the left and right suprachiasmatic nuclei. Science 290, 799–801 (2000)
Yoshii, T. et al. Drosophila cry(b) mutation reveals two circadian clocks that drive locomotor rhythm and have different responsiveness to light. J. Insect Physiol. 50, 479–488 (2004)
Jagota, A., de la Iglesia, H. O. & Schwartz, W. J. Morning and evening circadian oscillations in the suprachiasmatic nucleus in vitro. Nature Neurosci. 3, 372–376 (2000)
Kaneko, M. & Hall, J. C. Neuroanatomy of cells expressing clock genes in Drosophila: transgenic manipulation of the period and timeless genes to mark the perikarya of circadian pacemaker neurons and their projections. J. Comp. Neurol. 422, 66–94 (2000)
Frisch, B., Hardin, P. E., Hamblen-Coyle, M. J., Rosbash, M. & Hall, J. C. A promoterless period gene mediates behavioral rhythmicity and cyclical per expression in a restricted subset of the Drosophila nervous system. Neuron 12, 555–570 (1994)
Kaneko, M., Park, J. H., Cheng, Y., Hardin, P. E. & Hall, J. C. Disruption of synaptic transmission or clock-gene-product oscillations in circadian pacemaker cells of Drosophila cause abnormal behavioral rhythms. J. Neurobiol. 43, 207–233 (2000)
Helfrich-Förster, C. The period clock gene is expressed in central nervous system neurons which also produce a neuropeptide that reveals the projections of circadian pacemaker cells within the brain of Drosophila melanogaster. Proc. Natl Acad. Sci. USA 92, 612–616 (1995)
Renn, S. C., Park, J. H., Rosbash, M., Hall, J. C. & Taghert, P. H. A pdf neuropeptide gene mutation and ablation of PDF neurons each cause severe abnormalities of behavioral circadian rhythms in Drosophila. Cell 99, 791–802 (1999)
Blanchardon, E. et al. Defining the role of Drosophila lateral neurons in the control of circadian activity and eclosion rhythms by targeted genetic ablation and PERIOD protein overexpression. Eur. J. Neurosci. 13, 871–888 (2001)
Cheng, Y. Z. & Hardin, P. E. Drosophila photoreceptors contain an autonomous circadian oscillator that can function without period mRNA cycling. J. Neurosci. 18, 741–750 (1998)
Yang, Z. & Sehgal, A. Role of molecular oscillations in generating behavioral rhythms in Drosophila. Neuron 29, 453–467 (2001)
Klarsfeld, A. et al. Novel features of cryptochrome-mediated photoreception in the brain circadian clock of Drosophila. J. Neurosci. 24, 1468–1477 (2004)
Taghert, P. H. et al. Multiple amidated neuropeptides are required for normal circadian locomotor rhythms in Drosophila. J. Neurosci. 21, 6673–6686 (2001)
Siegmund, T. & Korge, G. Innervation of the ring gland of Drosophila melanogaster. J. Comp. Neurol. 431, 481–491 (2001)
Stoleru, D., Peng, P., Agosto, J. & Rosbash, M. Coupled oscillators control morning and evening locomotor behaviour of Drosophila. Nature doi:10.1038/nature02926 (this issue)
Ito, K., Urban, J. & Technau, G. Distribution, classification, and development of Drosophila glial cells in the late embryonic and early larval ventral nerve cord. Roux Arch. Dev. Biol. 204, 284–307 (1995)
Klarsfeld, A., Leloup, J. C. & Rouyer, F. Circadian rhythms of locomotor activity in Drosophila. Behav. Processes 64, 161–175 (2003)
Malpel, S., Klarsfeld, A. & Rouyer, F. Larval optic nerve and adult extra-retinal photoreceptors sequentially associate with the clock neurons during Drosophila brain development. Development 129, 1443–1453 (2002)
Acknowledgements
This work was supported by ACI ‘Biologie du développement et physiologie intégrative’ from the Ministère de la Recherche. F.R. is supported by INSERM, and R.X. is supported by Fondation des Treilles and previously by CNRS and Fondation Electricité de France. We thank M. Boudinot for the continuously improving Faas software, L. Collet for artwork, A. Klarsfeld and A. Lamouroux for critical reading of the manuscript, and J. Champagnat for his support. We thank J. Hall, P. Taghert and T. Siegmund for providing pdf-Gal4, C929-Gal4 and Mai179-Gal4 lines, respectively, as well as R. Stanewsky for anti-PER serum. We are grateful to H. Otsuna and K. Ito for sharing unpublished observations with the Mz520-Gal4 line, and to M. Rosbash and colleagues for communicating their unpublished manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Table 1
Gal4-driven Per expression in the lateral neurons in LD conditions. (DOC 28 kb)
Rights and permissions
About this article
Cite this article
Grima, B., Chélot, E., Xia, R. et al. Morning and evening peaks of activity rely on different clock neurons of the Drosophila brain. Nature 431, 869–873 (2004). https://doi.org/10.1038/nature02935
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02935
This article is cited by
-
A single photoreceptor splits perception and entrainment by cotransmission
Nature (2023)
-
Circadian clock disruption promotes the degeneration of dopaminergic neurons in male Drosophila
Nature Communications (2023)
-
A four-oscillator model of seasonally adapted morning and evening activities in Drosophila melanogaster
Journal of Comparative Physiology A (2023)
-
Direct and correlated responses to artificial selection on foraging in Drosophila
Behavioral Ecology and Sociobiology (2023)
-
Peculiar sleep features in sympatric species may contribute to the temporal segregation
Journal of Comparative Physiology B (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.