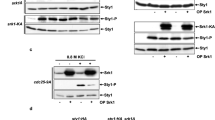
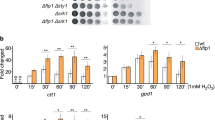
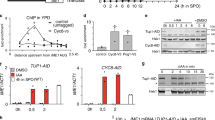
Abstract
Stress-activated mitogen-activated protein kinase cascades instigate a range of changes to enable eukaryotic cells to cope with particular insults. In Schizosaccharomyces pombe these responses include the transcription of specific gene sets and inhibition of entry into mitosis1,2. The S. pombe stress response pathway (SRP) also promotes commitment to mitosis in unperturbed cell cycles to allow cells to match their rate of division with nutrient availability1,3. The nature of this SRP function in cell cycle control is unknown. Entry into mitosis is controlled by mitosis-promoting factor (MPF; Cdc2/cyclin B) activity. Inhibitory phosphorylation of Cdc2 by Wee1 kinase inactivates MPF until Cdc25 removes this phosphate to promote mitosis4. The balance between Wee1 and Cdc25 activities is influenced by the recruitment of polo kinase (Plo1) to the spindle pole body (SPB)5. The SPB component Cut12 mediates this recruitment5,6. Hyper-activating mutations in either cut12 or plo1 enable Cdc25-defective cells to enter mitosis5,7. The hyperactive cut12.s11 mutation suppresses cdc25.22, as it promotes recruitment of active Plo1 to interphase SPBs6,7. Here we show that the SRP promotes phosphorylation of Plo1 on Ser 402. In unperturbed cell cycles, SRP-mediated phosphorylation of Ser 402 promotes Plo1 recruitment to SPBs and thus commitment to mitosis. Ser 402 phosphorylation also ensures efficient reinitiation of cell tip growth and cell division during recovery from particular stresses. Thus, phosphorylation of Plo1 Ser 402 not only enables SRP signalling to modulate the timing of mitotic commitment in response to nutrient status in unperturbed cycles, but also promotes the return to normal cell cycle control after stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Toone, W. M. & Jones, N. in The Molecular Biology of Schizosaccharomyces pombe (ed. Egel, R.) 57–72 (Springer, Berlin, 2004)
Chen, D. et al. Global transcriptional responses of fission yeast to environmental stress. Mol. Biol. Cell 14, 214–229 (2003)
Shiozaki, K. & Russell, P. Cell-cycle control linked to extracellular environment by MAP kinase pathway in fission yeast. Nature 378, 739–743 (1995)
Nurse, P. Universal control mechanism regulating onset of M-phase. Nature 344, 503–508 (1990)
MacIver, F. H., Tanaka, K., Robertson, A. M. & Hagan, I. M. Physical and functional interactions between polo kinase and the spindle pole component Cut12 regulate mitotic commitment in S. pombe. Genes Dev. 17, 1507–1523 (2003)
Mulvihill, D. P., Petersen, J., Ohkura, H., Glover, D. M. & Hagan, I. M. Plo1 kinase recruitment to the spindle pole body and its role in cell division in Schizosaccharomyces pombe. Mol. Biol. Cell 10, 2771–2785 (1999)
Hudson, J. D., Feilotter, H. & Young, P. G. stf1: non wee mutations epistatic to cdc25 in the fission yeast Schizosaccharomyces pombe. Genetics 126, 309–315 (1990)
Soloaga, A. et al. MSK2 and MSK1 mediate the mitogen- and stress-induced phosphorylation of histone H3 and HMG-14. EMBO J. 22, 2788–2797 (2003)
Nowak, S. J. & Corces, V. G. Phosphorylation of histone H3: a balancing act between chromosome condensation and transcriptional activation. Trends Genet. 20, 214–220 (2004)
Nurse, P. Genetic control of cell size at cell division in yeast. Nature 256, 547–551 (1975)
Mitchison, J. M. & Nurse, P. Growth in cell length in the fission yeast Schizosaccharomyces pombe. J. Cell Sci. 75, 357–376 (1985)
Qian, Y. W., Erikson, E. & Maller, J. L. Mitotic effects of a constitutively active mutant of the Xenopus polo-like kinase Plx1. Mol. Cell. Biol. 19, 8625–8632 (1999)
Shiozaki, K., Shiozaki, M. & Russell, P. Heat stress activates fission yeast Spc1/StyI MAPK by a MEKK-independent mechanism. Mol. Biol. Cell 9, 1339–1349 (1998)
Nguyen, A. N. & Shiozaki, K. Heat-shock-induced activation of stress MAP kinase is regulated by threonine- and tyrosine-specific phosphatases. Genes Dev. 13, 1653–1663 (1999)
May, J. & Mitchison, J. M. Length growth in fission yeast cells measured by two novel techniques. Nature 322, 752–754 (1986)
Marks, J. & Hyams, J. S. Localization of F-actin through the cell-division cycle of Schizosaccharomyces pombe. Eur. J. Cell Biol. 39, 27–32 (1985)
Nurse, P., Thuriaux, P. & Nasmyth, K. Genetic control of the cell division cycle in the fission yeast Schizosaccharomyces pombe. Mol. Gen. Genet. 146, 167–178 (1976)
Toczyski, D. P., Galgoczy, D. J. & Hartwell, L. H. CDC5 and CKII control adaptation to the yeast DNA damage checkpoint. Cell 90, 1097–1106 (1997)
Yoo, H. Y., Kumagai, A., Shevchenko, A. & Dunphy, W. G. Adaptation of a DNA replication checkpoint response depends upon inactivation of claspin by the Polo-like kinase. Cell 117, 575–588 (2004)
Van Vugt, M. A., Bras, A. & Medema, R. H. Polo-like kinase-1 controls recovery from a G2 DNA damage-induced arrest in mammalian cells. Mol. Cell 15, 799–811 (2004)
Ogden, J. E. & Fantes, P. A. Isolation of a novel type of mutation in the mitotic control of Schizosaccharomyces pombe whose phenotypic-expression is dependent on the genetic background and nutritional environment. Curr. Genet. 10, 509–514 (1986)
Moreno, S., Klar, A. & Nurse, P. Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol. 194, 795–823 (1991)
Maundrell, K. Thiamine-repressible expression vectors pREP and pRIP for fission yeast. Gene 123, 127–130 (1993)
MacIver, F. H., Glover, D. M. & Hagan, I. M. A ‘marker switch’ approach for targeted mutagenesis of genes in Schizosaccharomyces pombe. Yeast 20, 587–594 (2003)
Caspari, T. et al. Characterization of Schizosaccharomyces pombe Hus1: a PCNA-related protein that associates with Rad1 and Rad9. Mol. Cell. Biol. 20, 1254–1262 (2000)
Elia, A. E. et al. The molecular basis for phosphodependent substrate targeting and regulation of Plks by the Polo-box domain. Cell 115, 83–95 (2003)
Elia, A. E., Cantley, L. C. & Yaffe, M. B. Proteomic screen finds pSer/pThr-binding domain localizing Plk1 to mitotic substrates. Science 299, 1228–1231 (2003)
Simanis, V. & Nurse, P. The cell-cycle control gene cdc2+ of fission yeast encodes a protein-kinase potentially regulated by phosphorylation. Cell 45, 261–268 (1986)
Acknowledgements
We thank A. Grallert for technical assistance in the initial stages of the project; K. Gull, P. Russell, H. Ohkura and N. Jones for reagents; Paul Nurse for support; and N. Jones and P. Nurse for discussions. This work was supported by Cancer Research UK and Rockefeller University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures and Supplementary Table S1
Supplementary Figure S1 shows characterization of the phospho-S402 antibody and phosphorylation of serine 402 under different stress conditions. Supplementary Figure S2 shows the kinetics of S402 phosphorylation after heat shift of cdc25.22 and synchronized wild-type cells along with the kinetics of Spc1/Sty1 activation and the association between Plo1 and Spc1/Sty1 after heat stress. Supplementary Figure S3 shows that centrifugation de-polarizes the F-actin cytoskeleton. Supplementary Figure S4 shows that Plo1 does not dimerise after temperature shift because it is not possible to precipitate untagged Plo1 with epitope tagged Plo1. Supplementary Figure S5 shows a cartoon that summarizes the function of phosphorylation of S402 in cell cycle control and recovery from stress. (PPT 5761 kb)
Supplementary Legends
Full Legends to accompany the above Supplementary Figures. (DOC 61 kb)
Rights and permissions
About this article
Cite this article
Petersen, J., Hagan, I. Polo kinase links the stress pathway to cell cycle control and tip growth in fission yeast. Nature 435, 507–512 (2005). https://doi.org/10.1038/nature03590
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03590
This article is cited by
-
MAPK-dependent control of mitotic progression in S. pombe
BMC Biology (2024)
-
An updated view on the centrosome as a cell cycle regulator
Cell Division (2022)
-
Functional interaction between Cdc42 and the stress MAPK signaling pathway during the regulation of fission yeast polarized growth
International Microbiology (2020)
-
Phospho-mimicking Atf1 mutants bypass the transcription activating function of the MAP kinase Sty1 of fission yeast
Current Genetics (2018)
-
Plk1 regulates contraction of postmitotic smooth muscle cells and is required for vascular homeostasis
Nature Medicine (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.