Abstract
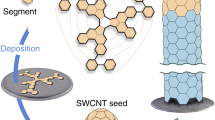
Over the past two decades, single-walled carbon nanotubes (SWCNTs) have received much attention because their extraordinary properties are promising for numerous applications1,2. Many of these properties depend sensitively on SWCNT structure, which is characterized by the chiral index (n,m) that denotes the length and orientation of the circumferential vector in the hexagonal carbon lattice. Electronic properties are particularly strongly affected, with subtle structural changes switching tubes from metallic to semiconducting with various bandgaps. Monodisperse ‘single-chirality’ (that is, with a single (n,m) index) SWCNTs are thus needed to fully exploit their technological potential1,2. Controlled synthesis through catalyst engineering3,4,5,6, end-cap engineering7 or cloning strategies8,9, and also tube sorting based on chromatography10,11, density-gradient centrifugation, electrophoresis and other techniques12, have delivered SWCNT samples with narrow distributions of tube diameter and a large fraction of a predetermined tube type. But an effective pathway to truly monodisperse SWCNTs remains elusive. The use of template molecules to unambiguously dictate the diameter and chirality of the resulting nanotube8,13,14,15,16 holds great promise in this regard, but has hitherto had only limited practical success7,17,18. Here we show that this bottom-up strategy can produce targeted nanotubes: we convert molecular precursors into ultrashort singly capped (6,6) ‘armchair’ nanotube seeds using surface-catalysed cyclodehydrogenation on a platinum (111) surface, and then elongate these during a subsequent growth phase to produce single-chirality and essentially defect-free SWCNTs with lengths up to a few hundred nanometres. We expect that our on-surface synthesis approach will provide a route to nanotube-based materials with highly optimized properties for applications such as light detectors, photovoltaics, field-effect transistors and sensors2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jorio, A., Dresselhaus, G. & Dresselhaus, M. S. Carbon Nanotubes: Advanced Topics in the Synthesis, Structure, Properties, and Applications (Springer, 2008)
Jariwala, D., Sangwan, V. K., Lauhon, L. J., Marks, T. J. & Hersam, M. C. Carbon nanomaterials for electronics, optoelectronics, photovoltaics, and sensing. Chem. Soc. Rev. 42, 2824–2860 (2013)
Wang, H. et al. Selective synthesis of (9,8) single walled carbon nanotubes on cobalt incorporated TUD-1 catalysts. J. Am. Chem. Soc. 132, 16747–16749 (2010)
He, M. et al. Selective growth of SWNTs on partially reduced monometallic cobalt catalyst. Chem. Commun. 47, 1219–1221 (2011)
Chiang, W.-H. & Mohan Sankaran, R. Linking catalyst composition to chirality distributions of as-grown single-walled carbon nanotubes by tuning NixFe1−x nanoparticles. Nature Mater. 8, 882–886 (2009)
Hong, G., Chen, Y., Li, P. & Zhang, J. Controlling the growth of single-walled carbon nanotubes on surfaces using metal and non-metal catalysts. Carbon 50, 2067–2082 (2012)
Yu, X. et al. Cap formation engineering: from opened C60 to single-walled carbon nanotubes. Nano Lett. 10, 3343–3349 (2010)
Smalley, R. E. et al. Single wall carbon nanotube amplification: en route to a type-specific growth mechanism. J. Am. Chem. Soc. 128, 15824–15829 (2006)
Yao, Y., Feng, C., Zhang, J. & Liu, Z. ‘Cloning’ of single-walled carbon nanotubes via open-end growth mechanism. Nano Lett. 9, 1673–1677 (2009)
Tu, X., Manohar, S., Jagota, A. & Zheng, M. DNA sequence motifs for structure-specific recognition and separation of carbon nanotubes. Nature 460, 250–253 (2009)
Liu, H., Nishide, D., Tanaka, T. & Kataura, H. Large-scale single-chirality separation of single-wall carbon nanotubes by simple gel chromatography. Nature Commun. 2, 309 (2011)
Hersam, M. C. Progress towards monodisperse single-walled carbon nanotubes. Nature Nanotechnol. 3, 387–394 (2008)
Mueller, A., Amsharov, K. Y. & Jansen, M. Synthesis of end-cap precursor molecules for (6, 6) armchair and (9, 0) zig-zag single-walled carbon nanotubes. Tetrahedr. Lett. 51, 3221–3225 (2010)
Bunz, U. H. F., Menning, S. & Martín, N. para-Connected cyclophenylenes and hemispherical polyarenes: building blocks for single-walled carbon nanotubes? Angew. Chem. Int. Edn Engl. 51, 7094–7101 (2012)
Mueller, A. & Amsharov, K. Y. Synthesis of precursors for large-diameter hemispherical buckybowls and precursors for short carbon nanotubes. Eur. J. Org. Chem. 2012, 6155–6164 (2012)
Omachi, H., Segawa, Y. & Itami, K. Synthesis of cycloparaphenylenes and related carbon nanorings: a step toward the controlled synthesis of carbon nanotubes. Acc. Chem. Res. 45, 1378–1389 (2012)
Mueller, A., Amsharov, K. Y. & Jansen, M. End-cap precursor molecules for the controlled growth of single-walled carbon nanotubes. Fullerenes Nanotubes Carbon Nanostruct. 20, 401–404 (2012)
Omachi, H., Nakayama, T., Takahashi, E., Segawa, Y. & Itami, K. Initiation of carbon nanotube growth by well-defined carbon nanorings. Nature Chem. 5, 572–576 (2013)
Otero, G. et al. Fullerenes from aromatic precursors by surface-catalysed cyclodehydrogenation. Nature 454, 865–868 (2008)
Amsharov, K. et al. Towards the isomer-specific synthesis of higher fullerenes and buckybowls by the surface-catalyzed cyclodehydrogenation of aromatic precursors. Angew. Chem. Int. Edn Engl. 49, 9392–9396 (2010)
Rim, K. T. et al. Forming aromatic hemispheres on transition-metal surfaces. Angew. Chem. Int. Edn Engl. 46, 7891–7895 (2007)
Gavillet, J. et al. Root-growth mechanism for single-wall carbon nanotubes. Phys. Rev. Lett. 87, 275504 (2001)
Dresselhaus, M. S., Dresselhaus, G., Saito, R. & Jorio, A. Raman spectroscopy of carbon nanotubes. Phys. Rep. 409, 47–99 (2005)
Jorio, A. et al. Quantifying carbon-nanotube species with resonance Raman scattering. Phys. Rev. B 72, 075207 (2005)
Soares, J. S. & Jorio, A. Study of carbon nanotube-substrate interaction. J. Nanotechnol. 2012, 1–10 (2012)
Jorio, A. et al. Linewidth of the Raman features of individual single-wall carbon nanotubes. Phys. Rev. B 66, 115411 (2002)
Telg, H. et al. Chiral index dependence of the G+ and G– Raman modes in semiconducting carbon nanotubes. ACS Nano 6, 904–911 (2012)
Piscanec, S., Lazzeri, M., Robertson, J., Ferrari, A. & Mauri, F. Optical phonons in carbon nanotubes: Kohn anomalies, Peierls distortions, and dynamic effects. Phys. Rev. B 75, 035427 (2007)
Rao, A. M. et al. Diameter-selective Raman scattering from vibrational modes in carbon nanotubes. Science 275, 187–191 (1997)
Xu, Y.-Q. et al. Vertical array growth of small diameter single-walled carbon nanotubes. J. Am. Chem. Soc. 128, 6560–6561 (2006)
Acknowledgements
This research was supported in part by the Swiss National Science Foundation and by the State Secretariat for Education, Research and Innovation via the COST Action MP0901 ‘NanoTP’. K.A. acknowledges financial support from Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Contributions
K.A., M.J. and R.F. initiated and conceived this work. K.A. designed the precursor molecules and the corresponding synthetic routes, K.A. and A.M. synthesised precursor molecules and performed HPLC, NMR and MS analyses. J.R.S.-V. carried out on-surface synthesis work. J.R.S.-V. and T.D. performed STM and Raman measurements, I.S. did the He ion microscopy analysis. O.G. performed the calculations. All authors participated in analysis and interpretation of the results. J.R.S.-V. drafted the manuscript, with contributions from P.R. and O.G. R.F. and K.A. edited the manuscript and coordinated the efforts of the research teams.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Conformations of precursor P1 adsorbed on Pt(111).
a, Sketch of the molecular conformations due to the axial chirality of the benzo[c]phenanthrene moieties. b, c, Simulations of the extended HOMO (LUMO shows a similar structure) with the outer biphenyls at a lower (b) or higher (c) elevation (left), and with the corresponding molecular structure superposed (right). d–h, Conformational analysis of P1 (top) and STM images of observed conformations of the P1 molecules on Pt(111) (bottom). i, Statistical analysis of the different molecular conformations shows that at least 50% of the molecules adopt a configuration suitable for a correct dehydrogenation pathway (red square).
Extended Data Figure 2 Dehydrogenation pathways for ‘right’ and ‘wrong’ conformations of P1.
a–d, Molecular dehydrogenation pathways for a ‘right’ conformation (a), and for ‘wrong’ conformations with 1 (b), 2 (c) and 3 (d) biphenyl arms rotated. Cases b, c, and d do not lead to a completed singly capped SWCNT and do not act as seeds for nanotube growth on epitaxial elongation.
Extended Data Figure 3 Scanning helium ion microscopy images.
SHIM images of epitaxially elongated SWCNTs obtained by exposing seeds S1 to 1 × 10−7 mbar of ethanol for 1 h (270 L) at 770 K. Long carbon nanotubes can be observed to lie on the surface, and in some cases to shake under the ion beam (indicated by light green arrows, bottom). Top left, lower-magnification view of surface: numbered coloured boxes are shown at higher magnification to the right. The unnumbered grey-scale images show different surface locations at higher magnifications. The rightmost panel gives a higher magnification image of the long SWCNT seen in panel 1.
Extended Data Figure 4 Synthesis of SWCNT precursor P1.
Details are given in Methods: here we describe reaction steps a to j. a, PPh3, toluene, reflux, 95%; b, BrPh3PCH2PhBr, KOtBu, EtOH, reflux, 81%; c, I2, hv, propylene oxide, cyclohexane, 72%; d, Pd(PPh3)4, Cs2CO3, toluene/MeOH, 110°C, 79%; e, NBS, DBPO, CCl4, reflux, 70%; f, NaCN, DMSO, RT, 40%; g, H2SO4, H2O, HOAc, reflux, 98%; h, SOCl2, 65 °C; i, AlCl3, CH2Cl2, RT, 57%; j, propanoic acid, TsOH, o-DCB, 180 °C, 65%.
Extended Data Figure 5 LDI mass spectra of precursor P1 (C96H54).
a, b, LDI mass spectra of P1 before (a) and after (b) sublimation. Computed and experimentally observed isotope distribution patterns for C96H54 are given in the inset of b.
Rights and permissions
About this article
Cite this article
Sanchez-Valencia, J., Dienel, T., Gröning, O. et al. Controlled synthesis of single-chirality carbon nanotubes. Nature 512, 61–64 (2014). https://doi.org/10.1038/nature13607
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13607
This article is cited by
-
Investigating valley-dependent current generation due to asymmetric energy dispersion for charge-transfer from a quantum dot to single-walled carbon nanotube
Scientific Reports (2023)
-
Trichalcogenasupersumanenes and its concave-convex supramolecular assembly with fullerenes
Nature Communications (2023)
-
Carbon Nanotube (10, 0) and Silicon Nanotube (7, 0) as a Novel Material for Drug Delivery of Substituted Eugenols as Antioxidant Drugs
Silicon (2023)
-
Synthesis of an all-carbon conjugated polymeric segment of carbon nanotubes and its application for lithium-ion batteries
Nano Research (2023)
-
Emerging Internet of Things driven carbon nanotubes-based devices
Nano Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.