Abstract
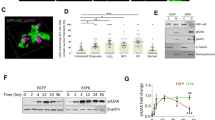
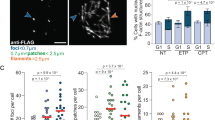
The presence of actin and nuclear myosin I (NMI) in the nucleus suggests a role for these motor proteins in nuclear functions. We have investigated the role of actin and nuclear myosin I (NMI) in the transcription of ribosomal RNA genes (rDNA). Both proteins are associated with rDNA and are required for RNA polymerase I (Pol I) transcription. Microinjection of antibodies against actin or NMI, as well as short interfering RNA-mediated depletion of NMI, decreased Pol I transcription in vivo, whereas overexpression of NMI augmented pre-rRNA synthesis. In vitro, recombinant NMI activated Pol I transcription, and antibodies to NMI or actin inhibited Pol I transcription both on naked DNA and pre-assembled chromatin templates. Whereas actin associated with Pol I, NMI bound to Pol I through the transcription-initiation factor TIF-IA. The association with Pol I requires phosphorylation of TIF-IA at Ser 649 by RSK kinase, indicating a role for NMI in the growth-dependent regulation of rRNA synthesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rando, O. J., Zhao, K. & Crabtree, G. R. Searching for a function for nuclear actin. Trends Cell Biol. 10, 92–97 (2000).
Pederson, T. Half a century of “the nuclear matrix”. Mol. Biol. Cell 11, 799–805 (2000).
Pederson, T. & Aebi, U. Actin in the nucleus: what form and what for? J. Struct. Biol. 140, 3–9 (2002).
Olave, I. A., Reck-Peterson, S. L. & Crabtree, G. R. Nuclear actin and actin-related proteins in chromatin remodeling. Annu. Rev. Biochem. 71, 755–781 (2002).
Bettinger, B., Gilbert, D. & Amberg, D. C. Actin up in the nucleus. Nature Rev. Mol. Cell Biol. 5, 410–415 (2004).
Muratani, M. et al. Metabolic-energy-dependent movement of PML bodies within the mammalian cell nucleus. Nature Cell Biol. 4, 106–110 (2002).
Nowak, G. et al. Evidence for the presence of myosin I in the nucleus. J. Biol. Chem. 272, 17176–17181 (1997).
Pestic-Dragovich, L. et al. A myosin I isoform in the nucleus. Science 290, 337–341 (2000).
Funaki, K., Katsumoto, T. & Iino, A. Immunocytochemical localization of actin in the nucleolus of rat oocytes. Biol. Cell. 84, 139–146 (1995).
Soyer-Gobillard, M. O., Ausseil, J. & Geraud, M. L. Nuclear and cytoplasmic actin in dinoflagellates. Biol. Cell. 87, 17–35 (1996).
Fomproix, N. & Percipalle, P. An actin-myosin complex on actively transcribing genes. Exp. Cell Res. 294, 140–148 (2004).
Egly, J. M., Miyamoto, N. G., Moncollin, V. & Chambon, P. Is actin a transcription initiation factor for RNA polymerase B? EMBO J. 3, 2363–2371 (1984).
Scheer, U., Hinssen, H., Franke, W. W. & Jockusch, B. M. Microinjection of actin-binding proteins and actin antibodies demonstrates involvement of nuclear actin in transcription of lampbrush chromosomes. Cell 39, 111–122 (1984).
Hofmann, W. A. et al. Actin is part of pre-initiation complexes and necessary for transcription by RNA polymerase II. Nature Cell Biol. 6, 1094–1101 (2004).
Percipalle, P. et al. An actin-ribonucleoprotein interaction is involved in transcription by RNA polymerase II. Proc. Natl Acad. Sci. USA 100, 6475–6480 (2003).
Tower, J. & Sollner-Webb, B. Transcription of mouse rDNA is regulated by an activated subform of RNA polymerase I. Cell 50, 873–883 (1987).
Schnapp, A., Pfleiderer, C., Rosenbauer, H. & Grummt, I. A growth-dependent transcription initiation factor (TIF-IA) interacting with RNA polymerase I regulates mouse ribosomal RNA synthesis. EMBO J. 9, 2857–2863 (1990).
Miller, G. et al. hRRN3 is essential in the SL1-mediated recruitment of RNA polymerase I to RNA gene promoters. EMBO J. 20, 1373–1382 (2001).
Grummt, I. Life on a planet of its own: regulation of RNA polymerase I transcription in the nucleolus. Genes Dev. 17, 1691–1702 (2003).
Yuan, X., Zhao, J., Zentgraf, H., Hoffmann-Rohrer, U. & Grummt, I. Multiple interactions between RNA polymerase I, TIF-IA and TAFI subunits regulate preinitiation complex assembly at the ribosomal gene promoter. EMBO Rep. 3, 1082–1087 (2002).
Bodem, J. et al. TIF–IA, the factor mediating growth-dependent control of ribosomal RNA synthesis, is the mammalian homolog of yeast Rrn3p. EMBO Rep. 1, 171–175 (2000).
Perry, R. P. & Kelley, D. E. Inhibition of RNA synthesis by actinomycin D: characteristic dose-response of different RNA species. J. Cell Physiol. 76, 127–139 (1970).
Zhao, J., Yuan, Y., Frödin, M. & Grummt, I. The activity of TIF-IA, a basal RNA polymerase I transcription factor, is regulated by MAP kinase-mediated signaling Mol. Cell 11, 405–413 (2003).
Zhao, K. et al. Rapid and phosphoinositol-dependent binding of the SWI/SNF-like BAF complex to chromatin after T lymphocyte receptor signaling. Cell 95, 625–636 (1998).
Smith, S. S., Kelly, K. H. & Jockusch, B. M. Actin co-purifies with RNA polymerase II. Biochem. Biophys. Res. Comm. 86, 161–166 (1979).
Nakayasu, H. & Ueda, K. Ultrastructural localization of actin in nuclear matrices from mouse leukemia L5178Y cells. Cell. Struct. Funct. 10, 305–309 (1985).
Schröder, H. C. et al. Cytochalasin B selectively releases ovalbumin mRNA precursors but not the mature ovalbumin mRNA from hen oviduct nuclear matrix. Eur. J. Biochem. 167, 239–245 (1987).
Percipalle, P. et al. Nuclear actin is associated with a specific subset of hnRNP A/B-type proteins. Nucleic Acids Res. 30, 1725–1734 (2002).
Nguyen, E., Besombes, D. & Debey, P. Immunofluorescent localization of actin in relation to transcription sites in mouse pronuclei. Mol. Reprod. Dev. 50, 263–272 (1998).
Percipalle, P. et al. Actin bound to the heterogeneous nuclear ribonucleoprotein hrp36 is associated with Balbiani ring mRNA from the gene to polysomes. J. Cell Biol. 153, 229–236 (2001).
Cavanaugh, A. H. et al. Rrn3 phosphorylation is a regulatory checkpoint for ribosome biogenesis. J. Biol. Chem. 277, 27423–27432 (2002).
Brun, R. P., Ryan, K. & Sollner-Webb, B. Factor C*, the specific initiation component of the mouse RNA polymerase I holoenzyme, is inactivated early in the transcription process. Mol. Cell. Biol. 14, 5010–5020 (1994).
Hirschler-Laszkiewicz, I. et al. Rrn3 becomes inactivated in the process of ribosomal DNA transcription. J. Biol. Chem. 278, 18953–18959 (2003).
Seither, P. & Grummt, I. Molecular cloning of RPA2, the gene encoding the second largest subunit of mouse RNA polymerase I. Genomics 37, 135–139 (1996).
Schnapp, A. & Grummt, I. Purification, assay, and properties of RNA polymerase I and class I-specific transcription factors in mouse. Methods Enzymol. 273, 233–248 (1996).
Budde, A. & Grummt I. p53 represses ribosomal gene transcription. Oncogene 18, 1119–1124 (1999).
Längst, G., Becker, P. B. & Grummt, I. TTF-I determines the chromatin architecture of the active rDNA promoter. EMBO J. 17, 3135–3145 (1998).
Acknowledgements
We are grateful to J. Lessard for the C4 monoclonal anti-actin antibody and R. Santoro for providing pre-assembled chromatin templates. P.H. was supported by the Grant Agency of the Czech Republic (Reg. No. 304/01/0661 and 2004/04/0108), by the Grant Agency of the Academy of Sciences of the Czech Republic (Reg. No. IAA5039202), by NSF/MŠMT ME 470 and by the Institutional Grant No. AV0Z5039906. V.P. was supported by the Grant Agency of the Czech Republic (Reg. No. 304/03/P118). P.de L. was supported in part by grants from the US Public Health Service (NIH GM 59648) and the US National Science Foundation (INT 9724168). I.G. was supported by the Deutsche Forschungsgemeinschaft, the EU-network “Epigenome” and the Fonds der Chemischen Industrie.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Philimonenko, V., Zhao, J., Iben, S. et al. Nuclear actin and myosin I are required for RNA polymerase I transcription. Nat Cell Biol 6, 1165–1172 (2004). https://doi.org/10.1038/ncb1190
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1190
This article is cited by
-
Beyond the cytoplasm: nuclear α-actin influences differentiation
Nature Cardiovascular Research (2023)
-
Nuclear myosin VI maintains replication fork stability
Nature Communications (2023)
-
Novel role of CAP1 in regulation RNA polymerase II-mediated transcription elongation depends on its actin-depolymerization activity in nucleoplasm
Oncogene (2021)
-
Nuclear actin filaments in DNA repair dynamics
Nature Cell Biology (2019)
-
Targeting ROCK/LIMK/cofilin signaling pathway in cancer
Archives of Pharmacal Research (2019)