Abstract
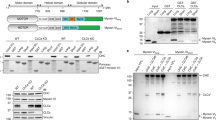
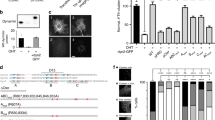
Vesicle transport is essential for the movement of proteins, lipids and other molecules between membrane compartments within the cell. The role of the class VI myosins in vesicular transport is particularly intriguing because they are the only class that has been shown to move 'backwards' towards the minus end of actin filaments1. Myosin VI is found in distinct intracellular locations and implicated in processes such as endocytosis2,3, exocytosis, maintenance of Golgi morphology4,5 and cell movement6. We have shown that the carboxy-terminal tail is the key targeting region and have identified three binding sites: a WWY motif for Disabled-2 (Dab2) binding, a RRL motif for glucose-transporter binding protein (GIPC) and optineurin binding and a site that binds specifically and with high affinity (Kd = 0.3 μM) to PtdIns(4,5)P2-containing liposomes. This is the first demonstration that myosin VI binds lipid membranes. Lipid binding induces a large structural change in the myosin VI tail (31% increase in helicity) and when associated with lipid vesicles, it can dimerize. In vivo targeting and recruitment of myosin VI to clathrin-coated structures (CCSs) at the plasma membrane is mediated by Dab2 and PtdIns(4,5)P2 binding.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wells, A. L. et al. Myosin VI is an actin-based motor that moves backwards. Nature 401, 505–508 (1999).
Buss, F., Arden, S. D., Lindsay, M., Luzio, J. P. & Kendrick-Jones, J. Myosin VI isoform localized to clathrin-coated vesicles with a role in clathrin-mediated endocytosis. EMBO J. 20, 3676–3684 (2001).
Aschenbrenner, L., Lee, T. & Hasson, T. Myo6 facilitates the translocation of endocytic vesicles from cell peripheries. Mol. Biol. Cell 14, 2728–2743 (2003).
Sahlender, D. A. et al. Optineurin links myosin VI to the Golgi complex and is involved in Golgi organization and exocytosis. J. Cell Biol. 169, 285–295 (2005).
Warner, C. L. et al. Loss of myosin VI reduces secretion and the size of the Golgi in fibroblasts from Snell's waltzer mice. EMBO J. 22, 569–579 (2003).
Geisbrecht, E. R. & Montell, D. J. Myosin VI is required for E-cadherin-mediated border cell migration. Nature Cell Biol. 4, 616–620 (2002).
Morris, S. M. et al. Myosin VI binds to and localises with Dab2, potentially linking receptor-mediated endocytosis and the actin cytoskeleton. Traffic 3, 331–341 (2002).
Dance, A. L. et al. Regulation of myosin-VI targeting to endocytic compartments. Traffic 5, 798–813 (2004).
Bunn, R. C., Jensen, M. A. & Reed, B. C. Protein interactions with the glucose transporter binding protein GLUT1CBP that provide a link between GLUT1 and the cytoskeleton. Mol. Biol. Cell 10, 819–832 (1999).
Lou, X., McQuistan, T., Orlando, R. A. & Farquhar, M. G. GAIP, GIPC and Gαi3 are concentrated in endocytic compartments of proximal tubule cells: putative role in regulating megalin's function. J. Am. Soc. Nephrol. 13, 918–927 (2002).
Pashkova, N., Jin, Y., Ramaswamy, S. & Weisman, L. S. Structural basis for myosin V discrimination between distinct cargoes. EMBO J. 25, 693–700 (2006).
Klopfenstein, D. R., Tomishige, M., Stuurman, N. & Vale, R. D. Role of phosphatidylinositol(4,5)bisphosphate organization in membrane transport by the Unc104 kinesin motor. Cell 109, 347–358 (2002).
Lister, I. et al. A monomeric myosin VI with a large working stroke. EMBO J. 23, 1729–1738 (2004).
Evans, P. R. & Owen, D. J. Endocytosis and vesicle trafficking. Curr. Opin. Struct. Biol. 12, 814–821 (2002).
Dell'Angelica, E. C. Clathrin-binding proteins: got a motif? Join the network! Trends Cell Biol. 11, 315–318 (2001).
Combet, C., Blanchet, C., Geourjon, C. & Deleage, G. NPS@: network protein sequence analysis. Trends Biochem. Sci. 25, 147–150 (2000).
Brett, T. J., Traub, L. M. & Fremont, D. H. Accessory protein recruitment motifs in clathrin-mediated endocytosis. Structure (Camb) 10, 797–809 (2002).
Kalthoff, C., Alves, J., Urbanke, C., Knorr, R. & Ungewickell, E. J. Unusual structural organization of the endocytic proteins AP180 and epsin 1. J. Biol. Chem. 277, 8209–8216 (2002).
Sakisaka, T., Itoh, T., Miura, K. & Takenawa, T. Phosphatidylinositol 4,5-bisphosphate phosphatase regulates the rearrangement of actin filaments. Mol. Cell Biol. 17, 3841–3849 (1997).
Czech, M. P. PIP2 and PIP3: complex roles at the cell surface. Cell 100, 603–606 (2000).
Cremona, O. & De Camilli, P. Phosphoinositides in membrane traffic at the synapse. J. Cell. Sci. 114, 1041–1052 (2001).
Itoh, T. & Takenawa, T. Regulation of endocytosis by phosphatidylinositol 4,5-bisphosphate and ENTH proteins. Curr. Top. Microbiol. Immunol. 282, 31–47 (2004).
Janmey, P. A., Xian, W. & Flanagan, L. A. Controlling cytoskeleton structure by phosphoinositide-protein interactions: phosphoinositide binding protein domains and effects of lipid packing. Chem. Phys. Lipids 101, 93–107 (1999).
Klopfenstein, D. R. & Vale, R. D. The lipid binding pleckstrin homology domain in UNC-104 kinesin is necessary for synaptic vesicle transport in Caenorhabditis elegans. Mol. Biol. Cell. 15, 3729–3739 (2004).
Park, H. et al. Full-length myosin VI dimerizes and moves processively along actin filaments upon monomer clustering. Mol. Cell 21, 331–336 (2006).
Iwaki, M. et al. Cargo binding makes a wild-type single-headed myosin-VI move processively. Biophys J. 90, 3643–3652 (2006).
Buss, F. et al. The localization of myosin VI at the Golgi complex and leading edge of fibroblasts and its phosphorylation and recruitment into membrane ruffles of A431 cells after growth factor stimulation. J. Cell Biol. 143, 1535–1545 (1998).
Peter, B. J. et al. BAR domains as sensors of membrane curvature: the amphiphysin BAR structure. Science 303, 495–9 (2003).
Acknowledgements
We thank: B. Peter and H. McMahon for help with lipid binding assays and discussion; R. Williams, D. Veprintsev and O. Perisic for programmes and help with the FRET assay; S. Peak-Chew and F. Begum for N-terminal sequencing and mass spectrometry; and D. Owen for critical reading of the manuscript. The work was funded by a USA Royal Society Postdoctoral Fellowship (G.S.), a Croucher Foundation (Hong Kong) Student Scholarship (J.S.A.), a Wellcome Trust Senior Fellowship (F.B.) and was supported by the Medical Research Council. The Cambridge Institute for Medical Research is in receipt of a strategic award from the Wellcome Trust.
Author information
Authors and Affiliations
Contributions
Each of the authors made a significant contribution to the experimental work described.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1 and S2 (PDF 407 kb)
Rights and permissions
About this article
Cite this article
Spudich, G., Chibalina, M., Au, JY. et al. Myosin VI targeting to clathrin-coated structures and dimerization is mediated by binding to Disabled-2 and PtdIns(4,5)P2. Nat Cell Biol 9, 176–183 (2007). https://doi.org/10.1038/ncb1531
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1531
This article is cited by
-
Nuclear myosin VI maintains replication fork stability
Nature Communications (2023)
-
MYL3 protects chondrocytes from senescence by inhibiting clathrin-mediated endocytosis and activating of Notch signaling
Nature Communications (2023)
-
How myosin VI traps its off-state, is activated and dimerizes
Nature Communications (2023)
-
The cross-talk of energy sensing and mitochondrial anchoring sustains synaptic efficacy by maintaining presynaptic metabolism
Nature Metabolism (2020)
-
Structure of Myosin VI/Tom1 complex reveals a cargo recognition mode of Myosin VI for tethering
Nature Communications (2019)