Abstract
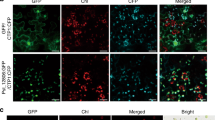
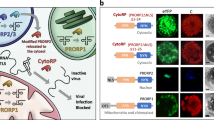
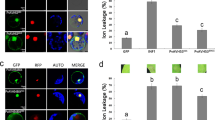
Systemic movement is central to plant viral infection. Exposure of tobacco plants to low levels of cadmium ions blocks the systemic spread of turnip vein-clearing tobamovirus (TVCV). We identified a tobacco glycine-rich protein, cdiGRP, specifically induced by low concentrations of cadmium and expressed in the cell walls of plant vascular tissues. Constitutive cdiGRP expression inhibited systemic transport of TVCV, whereas suppression of cdiGRP production allowed TVCV movement in the presence of cadmium. cdiGRP exerted its inhibitory effect on TVCV transport by enhancing callose deposits in the vasculature. So cdiGRP may function to control plant viral systemic movement.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Leisner, S. M. & Howell, S. H. Long-distance movement of viruses in plants. Trends Microbiol. 1, 314–317 (1993).
Rhee, Y., Tzfira, T., Chen, M.-H., Waigmann, E. & Citovsky, V. Cell-to-cell movement of tobacco mosaic virus: enigmas and explanations. Mol. Plant Pathol. 1, 33–39 (2000).
Oparka, K. J. & Santa Cruz, S. The great escape: phloem transport and unloading of macromoleculaes. Annu. Rev. Plant Physiol. Plant Mol. Biol. 51, 323–347 (2000).
Creager, A. N. H., Scholthof, K.-B., Citovsky, V. & Scholthof, H. B. Tobacco mosaic virus: pioneering research for a century. Plant Cell 11, 301–308 (1999).
Deom, C. M., Shaw, M. J. & Beachy, R. N. The 30-kilodalton gene product of tobacco mosaic virus potentiates virus movement. Science 237, 389–394 (1987).
Hilf, M. E. & Dawson, W. O. The tobamovirus capsid protein functions as a host-specific determinant of long-distance movement. Virology 193, 106–114 (1993).
Derrick, P. M., Carter, S. A. & Nelson, R. S. Mutation of the tobacco mosaic tobamovirus 126- and 183-kDa proteins: effects on phloem-dependent virus accumulation and synthesis of viral proteins. Mol. Plant-Microbe Interact. 10, 589–596 (1997).
Boyko, V., Ferralli, J., Ashby, J., Schellenbaum, P. & Heinlein, M. Function of microtubules in intercellular transport of plant virus RNA. Nature Cell Biol. 2, 826–832 (2000).
Heinlein, M., Epel, B. L., Padgett, H. S. & Beachy, R. N. Interaction of tobamovirus movement proteins with the plant cytoskeleton. Science 270, 1983–1985 (1995).
Waigmann, E., Chen, M.-H., Bachmaier, R., Ghoshroy, S. & Citovsky, V. Phosphorylation of tobacco mosaic virus cell-to-cell movement protein regulates viral movement in a host-specific fashion. EMBO J. 19, 4875–4884 (2000).
Kawakami, S. et al. Phosphorylation and/or presence of serine 37 in the movement protein of tomato mosaic tobamovirus is essential for intracellular localization and stability in vivo. J. Virol. 73, 6831–6840 (1999).
Chen, M.-H., Sheng, J., Hind, G., Handa, A. & Citovsky, V. Interaction between the tobacco mosaic virus movement protein and host cell pectin methylesterases is required for viral cell-to-cell movement. EMBO J. 19, 913–920 (2000).
Dorokhov, Y. L. et al. A novel function for a ubiquitous plant enzyme pectin methylesterase: the host-cell receptor for the tobacco mosaic virus movement protein. FEBS Lett. 461, 223–228 (1999).
Ghoshroy, S., Freedman, K., Lartey, R. & Citovsky, V. Inhibition of plant viral systemic infection by non-toxic concentrations of cadmium. Plant J. 13, 591–602 (1998).
Citovsky, V., Ghoshroy, S., Tsui, F. & Klessig, D. F. Non-toxic concentrations of cadmium inhibit tobamoviral systemic movement by a salicylic acid-independent mechanism. Plant J. 16, 13–20 (1998).
Kang, D. C., LaFrance, R., Su, Z. Z. & Fisher, P. B. Reciprocal subtraction differential RNA display: an efficient and rapid procedure for isolating differentially expressed gene sequences. Proc. Natl Acad. Sci. USA 95, 13788–13793 (1998).
van Kan, J. A., Cornelissen, B. J. & Bol, J. F. A virus-inducible tobacco gene encoding a glycine-rich protein shares putative regulatory elements with the ribulose bisphosphate carboxylase small subunit gene. Mol. Plant-Microbe Interact. 1, 107–112 (1988).
Domingo, C., Sauri, A., Mansilla, E., Conejero, V. & Vera, P. Identification of a novel peptide motif that mediates cross-linking of proteins to cell walls. Plant J. 20, 563–570 (1999).
Sachetto-Martins, G., Franco, L. O. & de Oliveira, D. E. Plant glycine-rich proteins: a family or just proteins with a common motif? Biochim. Biophys. Acta 1492, 1–14 (2000).
Cheng, N. H., Su, C. L., Carter, S. A. & Nelson, R. S. Vascular invasion routes and systemic accumulation patterns of tobacco mosaic virus in Nicotiana benthamiana. Plant J. 23, 349–362 (2000).
Roberts, A. G. et al. Phloem unloading in sink leaves of Nicotiana benthamiana: comparison of a fluorescent solute with a fluorescent virus. Plant Cell 9, 1381–1396 (1997).
Lartey, R., Ghoshroy, S., Ho, J. & Citovsky, V. Movement and subcellular localization of a tobamovirus in Arabidopsis. Plant J. 12, 537–545 (1997).
Lartey, R. T., Voss, T. C. & Melcher, U. Completion of a cDNA sequence from a tobamovirus pathogenic to crucifers. Gene 166, 331–332 (1995).
Delmer, D. P. et al. A monoclonal antibody recognizes a 65 kDa higher plant membrane polypeptide which undergoes cation-dependent association with callose deposition in vivo. Protoplasma 176, 33–42 (1993).
Iglesias, V. A. & Meins Jr., F. Movement of plant viruses is delayed in a beta-1,3-glucanase-deficient mutant showing a reduced plasmodesmatal size exclusion limit and enhanced callose deposition. Plant J. 21, 157–166 (2000).
Bucher, G. L. et al. Local expression of enzymatically active class I beta-1,3-glucanase enhances symptoms of TMV infection in tobacco. Plant J. 28, 361–369 (2001).
Beffa, R. S., Hofer, R.-M., Thomas, M. & Meins Jr., F. Decreased susceptibility to virus disease of beta-1,3-glucanase-deficient plants generated by antisense transformation. Plant Cell 8, 1001–1011 (1996).
Jackson, D. Opening up the communication channels: recent insights into plasmodesmal function. Curr. Opin. Plant Biol. 3, 394–399 (2000).
Tzfira, T., Rhee, Y., Chen, M.-H. & Citovsky, V. Nucleic acid transport in plant–microbe interactions: the molecules that walk through the walls. Annu. Rev. Microbiol. 54, 187–219 (2000).
Vaucheret, H. et al. Transgene-induced gene silencing in plants. Plant J. 16, 651–659 (1998).
Linthorst, H. J., Meuwissen, R. L., Kauffmann, S. & Bol, J. F. Constitutive expression of pathogenesis-related proteins PR-1, GRP, and PR-S in tobacco has no effect on virus infection. Plant Cell 1, 285–291 (1989).
Stone, B. A. & Clarke, A. E. Chemistry and Biology of 1→3-beta-glucans (La Trobe University Press, Victoria, Australia, 1992).
Xoconostle-Cázares, B. et al. Plant paralog to viral movement protein that potentiates transport of mRNA into the phloem. Science 283, 94–98 (1999).
Hamilton, A. J. & Baulcombe, D. C. A species of small antisense RNA in posttranscriptional gene silencing in plants. Science 286, 950–952 (1999).
Mourrain, P. et al. Arabidopsis SGS2 and SGS3 genes are required for posttranscriptional gene silencing and natural virus resistance. Cell 101, 533–542 (2000).
Fagard, M. & Vaucheret, H. (Trans)gene silncing in plants: How many mechanisms? Annu. Rev. Plant Physiol. Plant Mol. Biol. 51, 167–194 (2000).
Ueki, S. & Citovsky, V. Inhibition of post transcriptional gene silencing by non-toxic concentrations of cadmium. Plant J. 28, 283–291 (2001).
Kunik, T. et al. Transgenic tomato plants expressing the tomato yellow leaf curl virus capsid protein are resistant to the virus. Bio/Technology 12, 500–504 (1994).
Abel, P. P. et al. Delay of disease development in transgenic plants that express the tobacco mosaic virus coat protein gene. Science 232, 738–743 (1986).
Golemboski, D. B., Lomonossoff, G. P. & Zaitlin, M. Plants transformed with a tobacco mosaic virus nonstructural gene sequence are resistant to the virus. Proc. Natl Acad. Sci. USA 87, 6311–6315 (1990).
Pawlowski, K., Kunze, R., de Vries, S. & Bisseling, T. in Plant Molecular Biology Manual (eds Gelvin, S. B., Schilperoort, R. & Verma, D. P. S.) D5:1–13 (Kluwer Acad., 1994).
Ausubel, F. M. et al. Current Protocols in Molecular Biology (Greene Publishing-Wiley Interscience, New York, 1987).
Salt, D. E., Prince, R. C., Pickering, I. J. & Raskin, I. Mechanisms of cadmium mobility and accumulation in indian mustard. Plant Physiol. 109, 1427–1433 (1995).
Ghoshroy, S. & Citovsky, V. Preservation of plant cell ultrastructure during immunolocalization of virus particles. J. Virol. Methods 74, 223–229 (1998).
Gallie, D. R., Lucas, W. J. & Walbot, V. Visualizing mRNA expression in plant protoplasts: factors influencing efficient mRNA uptake and translation. Plant Cell 1, 303–311 (1989).
Horsch, R. B. et al. A simple and general method for transferring genes into plants. Science 227, 1229–1231 (1985).
Murashige, T. & Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiolog. Plant. 15, 473–497 (1962).
Kraft, R., Tardiff, J., Kranter, K. S. & Leinwand, L. A. Using miniprep plasmid DNA for sequencing double stranded templates with Sequenase. BioTechniques 6, 544–547 (1988).
Lartey, R. T., Hartson, S. D., Pennington, R. E., Sherwood, J. L. & Melcher, U. Occurrence of a vein-clearing tobamovirus in turnip. Plant Disease 77, 21–24 (1993).
Esau, K. in Anatomy of Seed Plants (ed. Esau, K.) 157–180 (John Wiley & Sons, Inc., New York, 1977).
Acknowledgements
We thank H. Scholthof for helpful discussions, and P. Vera and N. Carpita for critical reading of the paper. We are also grateful to F. Meins Jr. for his kind gift of TAG4.4 plants. We would like to express our gratitude to the University Microscopy Imaging Center (UMIC) at the State University of New York at Stony Brook for their technical help. This work was supported by grants from National Institutes of Health, National Science Foundation Functional Genomic Initiative, U.S. Department of Agriculture, U.S.-Israel Binational Science Foundation (BSF), and U.S.-Israel Binational Research and Development Fund (BARD) to V. C., and, in part, by a postdoctoral fellowship from the Japan Society for the Promotion of Science to S. U.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Ueki, S., Citovsky, V. The systemic movement of a tobamovirus is inhibited by a cadmium-ion-induced glycine-rich protein. Nat Cell Biol 4, 478–486 (2002). https://doi.org/10.1038/ncb806
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb806
This article is cited by
-
Comprehensive transcriptome analyses of Fusarium-infected root xylem tissues to decipher genes involved in chickpea wilt resistance
3 Biotech (2023)
-
Callose deposited at soybean sieve element inhibits long-distance transport of Soybean mosaic virus
AMB Express (2022)
-
Genome-wide identification, phylogenetic analysis, and expression profiling of glycine-rich RNA-binding protein (GRPs) genes in seeded and seedless grapes (Vitis vinifera)
Physiology and Molecular Biology of Plants (2021)
-
Cell wall Glycine-rich Protein2 is involved in tapetal differentiation and pollen maturation
Journal of Plant Research (2020)
-
Genetic analysis of a Piezo-like protein suppressing systemic movement of plant viruses in Arabidopsis thaliana
Scientific Reports (2019)