Abstract
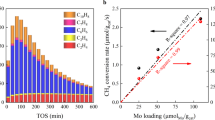
Developing efficient catalytic processes to convert methane into useful feedstocks relies critically upon devising new coupling processes that use abundant, thermodynamically ‘mild’ oxidants together with selective catalysts. We report here on elemental sulfur as a promising ‘soft’ oxidant for selective methane conversion to ethylene over MoS2, RuS2, TiS2, PdS and Pd/ZrO2 catalysts. Experiments and density functional theory reveal that methane conversion is directly correlated with surface metal–sulfur bond strengths. Surfaces with weakly bound sulfur are more basic and activate methane C–H bonds more readily. In contrast, experimental and theoretical selectivities scale inversely with surface metal–sulfur bond strengths, and surfaces with the strongest metal–sulfur bonds afford the highest ethylene selectivities. High CH4/S ratios, short contact times and the provision of a support maximizes the coupling of CHx intermediates and selectivity to ethylene, because these conditions yield surfaces with stronger metal–sulfur bonding (for example, Pd16S7), which suppresses the over-oxidation of methane.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tullo, A. H. Celanese takes an ethanol plunge. Chem. Eng. News 89, 20–21 (2011).
Crabtree, R. H. Aspects of methane chemistry. Chem. Rev. 95, 987–1007 (1995).
Lunsford, J. H. Catalytic conversion of methane to more useful chemicals and fuels: a challenge for the 21st century. Catal. Today 63, 165–174 (2000).
Holmen, A. Direct conversion of methane to fuels and chemicals. Catal. Today 142, 2–8 (2009).
Horn, R., Williams, K. A., Degenstein, N. J. & Schmidt, L. D. Syngas by catalytic partial oxidation of methane on rhodium: mechanistic conclusions from spatially resolved measurements and numerical simulations. J. Catal. 242, 92–102 (2006).
York, A. P. E., Xiao, T. C., Green, M. L. H. & Claridge J. B. Methane oxyforming for synthesis gas production. Catal. Rev. Sci. Eng. 49, 511–560 (2007).
Bar-Nahum, I., Khenkin, A. M. & Neumann, R. Mild, aqueous, aerobic, catalytic oxidation of methane to methanol and acetaldehyde catalyzed by a supported bipyrimidinylplatinum–polyoxometalate hybrid compound. J. Am. Chem. Soc. 126, 10236–10237 (2004).
Otsuka, K. & Wang, Y. Direct conversion of methane into oxygenates. Appl. Catal. A 222, 145–161 (2001).
Dry, M. E. Practical and theoretical aspects of the catalytic Fischer–Tropsch process. Appl. Catal. A 138, 319–344 (1996).
Flores, R. M. Coalbed methane: from hazard to resource. Int. J. Coal Geol. 35, 3–26 (1998).
DOE Basic Energy Sciences Advisory Committee. Directing Matter and Energy: Five Challenges for Science and the Imagination (National Academy Press, 2007).
Xu, Y. D., Bao, X. H. & Lin, L. W. Direct conversion of methane under nonoxidative conditions. J. Catal. 216, 386–395 (2003).
Podkolzin, S. G., Stangland, E. E., Jones, M. E., Peringer, E. & Lercher, J. A. Methyl chloride production from methane over lanthanum-based catalysts. J. Am. Chem. Soc. 129, 2569–2576 (2007).
Peringer, E., Salzinger, M., Hutt, M., Lemonidou, A. A. & Lercher J. A. Modified lanthanum catalysts for oxidative chlorination of methane. Top. Catal. 52, 1220–1231 (2009).
Zavyalova, U., Holena, M., Schlögl, R. & Baerns, M. Statistical analysis of past catalytic data on oxidative methane coupling for new insights into the composition of high-performance catalysts. ChemCatChem 3, 1935–1947 (2011) and references therein.
Lang, S. M., Bernhardt, T. M., Barnett R. N. & Landman, U. Methane activation and catalytic ethylene formation on free Au2+. Angew. Chem. Int. Ed. 49, 980–983 (2010).
Sorokin, A. B. et al. Oxidation of methane and ethylene in water at ambient conditions. Catal. Today 157, 149–154 (2010).
Periana, R. A. et al. Mercury-catalyzed, high-yield system for the oxidation of methane to methanol. Science 259, 340–343 (1993).
Groothaert, M. H., Smeets, P. J., Sels, B. F., Jacobs, P. A. & Schoonheydt, R. A. Selective oxidation of methane by the bis(mu-oxo)dicopper core stabilized on ZSM-5 and mordenite zeolites. J. Am. Chem. Soc. 127, 1394–1395 (2005).
An, Z. J., Pan, X. L., Liu, X. M., Han, X. W. & Bao, X. H. Combined redox couples for catalytic oxidation of methane by dioxygen at low temperatures. J. Am. Chem. Soc. 128, 16028–16029 (2006).
Luzgin, M. V. et al. Understanding methane aromatization on a Zn-modified high-silica zeolite. Angew. Chem. Int. Ed. 47, 4559–4562 (2008).
Palkovits, R., Antonietti, M., Kuhn, P., Thomas, A. & Schuth, F. Solid catalysts for the selective low-temperature oxidation of methane to methanol. Angew. Chem. Int. Ed. 48, 6909–6912 (2009).
Gretz, E., Oliver, T. F. & Sen, A. Carbon–hydrogen bond activation by electrophilic transition-metal compounds—palladium(II)-mediated oxidation of arenes and alkanes including methane. J. Am. Chem. Soc. 109, 8109–8111 (1987).
Linstrom, P. J. & Mallard, W. G. (eds) NIST Chemistry WebBook; NIST Standard Reference Database Number 69 (National Institute of Standards and Technology, 2011). Available at http://webbook.nist.gov.
Stull, D. R., Westrum, E. F. & Sinke, G. C. The Chemical Thermodynamics of Organic Compounds 243–244 (Wiley, 1969).
Polyakov, M. et al. Mechanochemical activation of MoS2—surface properties and catalytic activities in hydrogenation and isomerization of alkenes and in H2/D2 exchange. J. Catal. 260, 236–244 (2008).
Chianelli, R. R., et al. Periodic trends in hydrodesulfurization: in support of the Sabatier principle. Appl. Catal. A 227, 83–96 (2002).
Byskov, L. S., Norskov, J. K., Clausen, B. S. & Topsoe, H. DFT calculations of unpromoted and promoted MoS2-based hydrodesulfurization catalysts. J. Catal. 187, 109–122 (1999).
Besenbacher, F. et al. Recent STM, DFT and HAADF-STEM studies of sulfide-based hydrotreating catalysts: insight into mechanistic, structural and particle size effects. Catal. Today 130, 86–96 (2008).
Corma, A., Martinez, C., Ketley, G. & Blair, G. On the mechanism of sulfur removal during catalytic cracking. Appl. Catal. A 208, 135–152 (2001).
Kabe, T., Ishihara, A. & Qian, W. Hydrodesulfurization and Hydrodenitrogenation: Chemistry and Engineering (Wiley-VCH, 1999).
Chianelli, R. R. et al. Catalytic properties of single layers of transition metal sulfide catalytic materials. Catal. Rev. Sci. Eng. 48, 1–41 (2006).
Topsoe, H., Clausen, B. S. & Massoth, F. E. in Hydrotreating Catalysis—Science and Technology, Vol. 11 (eds Anderson, J.R. & Boudart, M.) (Springer, 1996).
Bezverkhyy, I., Afanasiev, P., Geantet, C. & Lacroix, M. Highly active (Co)MoS2/Al2O3 hydrodesulfurization catalysts prepared in aqueous solution. J. Catal. 204, 495–497 (2001).
Wang, H. M. & Iglesia, E. Thiophene hydrodesulfurization catalysis on supported Ru clusters: mechanism and site requirements for hydrogenation and desulfurization pathways. J. Catal. 273, 245–256 (2010).
Anderson, J. R., Chang, Y. F., Pratt, K. C. & Foger, K. Reaction of methane and sulfur—oxidative coupling and carbon-disulfide formation. React. Kinet. Catal. Lett. 49, 261–269 (1993).
Didenko, L. P., Linde, V. R. & Savchenko, V. I. Partial catalytic oxidation and condensation of methane by oxygen and sulfur. Catal. Today 42, 367–370 (1998).
Nehb, W. & Vydra, K. F. Sulfur. Ullman's Encyclopedia of Industrial Chemistry (Wiley-VCH, 2002).
Stair, P. C. The application of UV Raman spectroscopy for the characterization of catalysts and catalytic reactions. Adv. Catal. 51, 75–98 (2007).
Chin, Y., Buda C., Neurock, M. & Iglesia, E. Reactivity of chemisorbed oxygen atoms and their catalytic consequences during CH4/O2 catalysis on supported Pt clusters. J. Catal. 283, 10–24 (2011).
Chin, Y., Buda, B., Neurock, M. & Iglesia, E. Selectivity of chemisorbed oxygen in C–H bond activation and CO oxidation and kinetic consequences for CH4–O2 catalysis on Pt and Rh clusters. J. Am. Chem. Soc. 133, 15958–15978 (2011).
Chin, Y. & Iglesia, E. Elementary steps, the role of chemisorbed oxygen, and the effects of cluster size in catalytic CH4–O2 reactions on palladium. J. Phys. Chem. 115, 17845–17855 (2011).
Jüngst, E. & Nehb, W. Handbook of Heterogeneous Catalysis Ch. 12.4 (Wiley-VCH, 2008).
Acknowledgements
The authors thank the Dow Chemical Company for support as part of the Methane Challenge project. The authors thank S. Domke, P. Nickias, D. Barton and M. Kaminsky for stimulating discussions.
Author information
Authors and Affiliations
Contributions
Q.Z., S.L.W. and C.X. performed the experiments, analysed the data, and wrote portions of the manuscript. O.U. and M.N. performed DFT calculations and wrote portions of the manuscript. All authors contributed to writing and revising the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 8633 kb)
Rights and permissions
About this article
Cite this article
Zhu, Q., Wegener, S., Xie, C. et al. Sulfur as a selective ‘soft’ oxidant for catalytic methane conversion probed by experiment and theory. Nature Chem 5, 104–109 (2013). https://doi.org/10.1038/nchem.1527
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1527
This article is cited by
-
Catalytic reforming of methane with H2S via dynamically stabilized sulfur on transition metal oxides and sulfides
Nature Catalysis (2023)
-
Carbon oxygenate transformations by actinide compounds and catalysts
Nature Reviews Chemistry (2017)
-
Methane Activation by Heterogeneous Catalysis
Catalysis Letters (2015)
-
Oxidation goes soft
Nature Chemistry (2013)