Abstract
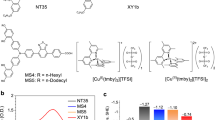
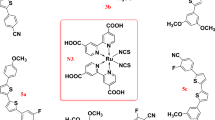
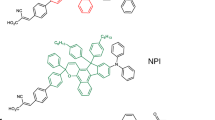
Dye-sensitized solar cells (DSCs) have achieved impressive conversion efficiencies for solar energy of over 11% with an electrolyte that contains triiodide/iodide as a redox couple. Although triiodide/iodide redox couples work efficiently in DSCs, they suffer from two major disadvantages: electrolytes that contain triiodide/iodide corrode electrical contacts made of silver (which reduces the options for the scale up of DSCs to module size) and triiodide partially absorbs visible light. Here, we present a new disulfide/thiolate redox couple that has negligible absorption in the visible spectral range, a very attractive feature for flexible DSCs that use transparent conductors as current collectors. Using this novel, iodide-free redox electrolyte in conjunction with a sensitized heterojunction, we achieved an unprecedented efficiency of 6.4% under standard illumination test conditions. This novel redox couple offers a viable pathway to develop efficient DSCs with attractive properties for scale up and practical applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Shah, A. et al. Photovoltaic technology: the case for thin-film solar cells. Science 285, 692–698 (1999).
Brabec, C. et al. Plastic solar cells. Adv. Funct. Mater. 11, 15–28 (2001).
O'Regan, B. & Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353, 737–740 (1991).
Chiba, Y. et al. Dye-sensitized solar cells with conversion efficiency of 11.1%. J. Appl. Phys. 45, L638–L640 (2006).
Nazeeruddin, M. et al. Engineering of efficient panchromatic sensitizers for nanocrystalline TiO2-based solar cells. J. Am. Chem. Soc. 123, 1613–1624 (2001).
Gao, F. et al. Enhance the optical absorptivity of nanocrystalline TiO2 film with high molar extinction coefficient ruthenium sensitizers for high performance dye-sensitized solar cells. J. Am. Chem. Soc. 130, 10720–10728 (2008).
Martinson, A. B. F. et al. New architectures for dye-sensitized solar cells. Chem. Eur. J. 14, 4458–4467 (2008).
Zhang, Z. et al. The 2,2,6,6-tetramethyl-1-piperidinyloxy radical: an efficient, iodine- free redox mediator for dye-sensitized solar cells. Adv. Funct. Mater. 18, 341–346 (2008).
Snaith, H. J. et al. Dye-sensitized solar cells incorporating a ‘liquid’ hole-transporting material. Nano. Lett. 6, 2000–2003 (2006).
Cameron, P. J. et al. Electrochemical studies of the Co(iii)/Co(ii)(dbbip)2 redox couple as a mediator for dye-sensitized nanocrystalline solar cells. Coord. Chem. Rev. 248, 1447–1453 (2004).
Yanagida, S. et al. Iodine/iodide-free dye-sensitized solar cells. Acc. Chem. Res. 42, 1827–1838 (2009).
O'Regan, B. et al. Electrodeposited nanocomposite n–p heterojunctions for solid-state dye-sensitized photovoltaics. Adv. Mater. 12, 1263–1267 (2000).
Bach, U. et al. Solid-state dye-sensitized mesoporous TiO2 solar cells with high photon-to-electron conversion efficiencies. Nature 395, 583–585 (1998).
Marsan, B. Cellules photovoltaïques électrochimiques à electrolyte polymère de configuration n-CdSe//POE modifié-M2S/xS//ITO. PhD thesis, INRS-Énergie, Univ. Québec (1988).
Philias, J.-M. & Marsan, B. All-solid-state photoelectrochemical cell based on a polymer electrolyte containing a new transparent and highly electropositive redox couple. Electrochim. Acta 44, 2915–2926 (1999).
Kay, A. et al. Artificial photosynthesis. 2. Investigations on the mechanism of photosensitization of nanocrystalline TiO2 solar cells by chlorophyll derivatives. J. Phys. Chem. 98, 952–959 (1994).
Ardo, S. & Meyer, G. Photodriven heterogeneous charge transfer with transition-metal compounds anchored to TiO2 semiconductor surfaces. J. Chem. Soc. Rev. 38, 115–164 (2009).
Nusbaumer, H. et al. An alternative efficient redox couple for the dye-sensitized solar cell system. Chem. Eur. J. 9, 3756–3763 (2003).
Sapp, S. A. et al. Substituted polypyridine complexes of cobalt(ii/iii) as efficient electron-transfer mediators in dye-sensitized solar cells. J. Am. Chem. Soc. 124, 11215–11222 (2002).
Cazzanti, S. et al. Efficient non-corrosive electron-transfer mediator mixtures for dye-sensitized solar cells. J. Am. Chem. Soc. 128, 9996–9997 (2006).
Wang, P. et al. Charge separation and efficient light energy conversion in sensitized mesoscopic solar cells based on binary ionic liquids. J. Am. Chem. Soc. 127, 6850–6856 (2005).
Wang, M. et al. The influence of charge transport and recombination on the performance of dye-sensitized solar cells. ChemPhysChem 10, 290–299 (2009).
O'Regan, B. et al. Measuring charge transport from transient photovoltage rise times. A new tool to investigate electron transport in nanoparticle films. J. Phys. Chem. B 110, 17155–17160 (2006).
Huang, S. et al. Charge recombination in dye-sensitized nanocrystalline TiO2 solar cells. J. Phys. Chem. B 101, 2576–2582 (1997).
Wang, M. et al. High-performance liquid and solid dye-sensitized solar cells based on a novel metal-free organic sensitizer. Adv. Mater. 20, 4460–4463 (2008).
Acknowledgements
The authors thank P. Comte for the TiO2 film fabrication, and C. Graetzel and K. Sivula for discussions. M.W., J.-E.M., R.H.-B., S.M.Z. and M.G. thank the Swiss National Science Foundation, and N.C., L.B. and B.M. thank the Natural Sciences and Engineering Research Council of Canada for financial support.
Author information
Authors and Affiliations
Contributions
M.G., B.M. and S.M.Z. contributed to the conception and design of the experiments, analysis of the data and writing of the paper. N.C. and L.B. carried out the experiments and contributed to the materials. J.-E.M. and R.H.-B. analysed the data and contributed to the analysis tools. M.W. carried out the experiments, analysed the data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 540 kb)
Rights and permissions
About this article
Cite this article
Wang, M., Chamberland, N., Breau, L. et al. An organic redox electrolyte to rival triiodide/iodide in dye-sensitized solar cells. Nature Chem 2, 385–389 (2010). https://doi.org/10.1038/nchem.610
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.610
This article is cited by
-
Review on fabrication methodologies and its impacts on performance of dye-sensitized solar cells
Environmental Science and Pollution Research (2022)
-
Optimization of Pt loading on the counter electrode for efficient and bifacial dye-sensitized solar cells with polymer gel electrolyte
Korean Journal of Chemical Engineering (2022)
-
Structural, optical, surface, and photocatalytic properties of SnO2 films produced by ultrasonic spray pyrolysis
Journal of Sol-Gel Science and Technology (2022)
-
Hydrophobic carbon/Whitlockite derived from expired yogurt as a counter electrode for dye-sensitized solar cell (DSSC)
Journal of Materials Science: Materials in Electronics (2022)
-
The effect of spinning cycle on structural, optical, surface and photocatalytic properties of sol–gel derived ZnO films
Journal of Sol-Gel Science and Technology (2021)