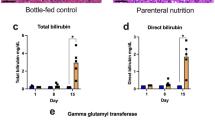
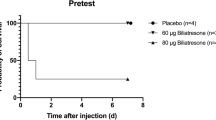
Abstract
Since its introduction into clinical practice, parenteral nutrition has revolutionized the care of premature neonates. Serum transaminase and bilirubin levels are commonly elevated in infants on parenteral nutrition, but their normalization is typical in the setting of short-term administration of parenteral nutrition uncomplicated by sepsis. Premature infants who require long-term parenteral nutrition are, however, at severe risk for developing life-threatening hepatic complications. These complications include cirrhosis, liver failure, and the concomitant risks of sepsis, coagulopathy and death. Premature infants and those with short-bowel syndrome are most susceptible to these morbid outcomes. Although it has been more than a quarter of a century since parenteral nutrition was first introduced and its association with hepatic complications described, the precise etiology of parenteral nutrition associated cholestasis (PNAC) remains a mystery; however, our understanding of the molecular components that contribute to PNAC has improved substantially. In this Review, we summarize the fundamentals of PNAC, describe animal models of the disease, review the hepatic bile acid transporters that are crucial for bile acid homeostasis, and define the roles that endotoxin, genetics, and the components of parenteral nutrition are likely to have in the molecular pathogenesis of this life-threatening condition.
Key Points
-
Parenteral nutrition provides valuable life-sustaining nutrients to patients unable to tolerate enteral nutrition, but prolonged administration is associated with severe, life-threatening liver disease, especially in premature neonates
-
The precise etiology of parenteral nutrition associated cholestasis (PNAC) is probably multifactorial, with risk factors including prematurity, history of bowel resection and/or lack of enteral feeds, catheter infections and/or sepsis, and a lack of or presence of specific components in the parenteral nutrition itself
-
Older children and adults are less prone to severe cases of PNAC than are neonates, and animal models suggest that there is a developmental component to bile acid transporter gene expression; these factors indicate that the expression of critical hepatic protective genes might be involved in susceptibility to PNAC
-
Animal models of parenteral nutrition administration suggest that parenteral nutrition itself and/or particular components of parenteral nutrition might affect key hepatic bile acid transporters, apoptotic signaling, and other detoxification mechanisms at the gene level, thereby promoting cholestasis and hepatocellular injury
-
Few therapeutic options are available for PNAC, with severe cases necessitating liver or combined liver–bowel transplantation
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rhoads JE (2001) The development of TPN: an interview with pioneer surgical nutritionist Jonathan E Rhoads, MD [Interview by Carolyn T Spencer and Charlene Compher]. J Am Diet Assoc 101: 747–750
Guenter P et al. (2004) The impact of nursing practice on the history and effectiveness of total parenteral nutrition. JPEN J Parenter Enteral Nutr 28: 54–59
[No authors listed] (1981) Nutrition classics. Surgery, Volume 64, 1968. Long-term total parenteral nutrition with growth, development, and positive nitrogen balance: Stanley J. Dudrick, Douglas W. Wilmore, Harry M. Vars, Jonathan E. Rhoads. Nutr Rev 39: 278–281
Rager R and Finegold MJ (1975) Cholestasis in immature newborn infants: is parenteral alimentation responsible. J Pediatr 86: 264–269
Finegold MJ (1982) Total parenteral alimentation and cholestasis. Am J Clin Pathol 77: 378–379
Drongowski RA and Coran AG (1989) An analysis of factors contributing to the development of total parenteral nutrition-induced cholestasis. JPEN J Parenter Enteral Nutr 13: 586–589
Sokol RJ (1997) Total parenteral nutrition-related liver disease. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 38: 418–428
Kelly DA (1998) Liver complications of pediatric parenteral nutrition—epidemiology. Nutrition 14: 153–157
Teitelbaum DH (1997) Parenteral nutrition-associated cholestasis. Curr Opin Pediatr 9: 270–275
Kelly DA (2006) Intestinal failure-associated liver disease: what do we know today. Gastroenterology 130 (Suppl 1): S70–S77
Beale EF et al. (1979) Intrahepatic cholestasis associated with parenteral nutrition in premature infants. Pediatrics 64: 342–347
Btaiche IF and Khalidi N (2002) Parenteral nutrition-associated liver complications in children. Pharmacotherapy 22: 188–211
Beath SV et al. (1996) Parenteral nutrition-related cholestasis in postsurgical neonates: multivariate analysis of risk factors. J Pediatr Surg 31: 604–606
Zambrano E et al. (2004) Total parenteral nutrition induced liver pathology: an autopsy series of 24 newborn cases. Pediatr Dev Pathol 7: 425–432
Mullick FG et al. (1994) Total parenteral nutrition: a histopathologic analysis of the liver changes in 20 children. Mod Pathol 7: 190–194
Cohen C and Olsen MM (1981) Pediatric total parenteral nutrition: liver histopathology. Arch Pathol Lab Med 105: 152–156
Colomb V et al. (1992) Long-term parenteral nutrition in children: liver and gallbladder disease. Transplant Proc 24: 1054–1055
Shulman RJ (2000) New developments in total parenteral nutrition for children. Curr Gastroenterol Rep 2: 253–258
Bell RL et al. (1986) Total parenteral nutrition-related cholestasis in infants. JPEN J Parenter Enteral Nutr 10: 356–359
Balistreri WF et al. (1983) Immaturity of the enterohepatic circulation in early life: factors predisposing to “physiologic” maldigestion and cholestasis. J Pediatr Gastroenterol Nutr 2: 346–354
Lester R et al. (1983) Diversity of bile acids in the fetus and newborn infant. J Pediatr Gastroenterol Nutr 2: 355–364
de Belle RC et al. (1979) Intestinal absorption of bile salts: immature development in the neonate. J Pediatr 94: 472–476
Denehy CM and Ryan JR (1986) Development of gallbladder contractility in the guinea pig. Pediatr Res 20: 214–217
Suchy FJ et al. (1986) Taurocholate transport and Na+-K+-ATPase activity in fetal and neonatal rat liver plasma membrane vesicles. Am J Physiol 251: 665–673
Karpen SJ (2002) Nuclear receptor regulation of hepatic function. J Hepatol 36: 832–850
Zollner G et al. (2006) Role of nuclear receptors in the adaptive response to bile acids and cholestasis: pathogenetic and therapeutic considerations. Mol Pharm 3: 231–251
Gao B et al. (2004) Differential expression of bile salt and organic anion transporters in developing rat liver. J Hepatol 41: 201–208
Chen HL et al. (2004) Developmental expression of canalicular transporter genes in human liver [abstract]. J Pediatr Gastroenterol Nutr. 39 (Suppl): aS68
Chen HL et al. (2005) Developmental expression of canalicular transporter genes in human liver. J Hepatol 43: 472–477
Touloukian RJ and Seashore JH (1975) Hepatic secretory obstruction with total parenteral nutrition in the infant. J Pediatr Surg 10: 353–360
Aynsley-Green A (1983) Plasma hormone concentrations during enteral and parenteral nutrition in the human newborn. J Pediatr Gastroenterol Nutr 2 (Suppl 1): S108–S112
Aynsley-Green A (1989) The endocrinology of feeding in the newborn. Baillieres Clin Endocrinol Metab 3: 837–868
Lucas A et al. (1983) Metabolic and endocrine consequences of depriving preterm infants of enteral nutrition. Acta Paediatr Scand 72: 245–249
Fedorowski T et al. (1979) Transformation of chenodeoxycholic acid and ursodeoxycholic acid by human intestinal bacteria. Gastroenterology 77: 1068–1073
Lichtman SN et al. (1990) Hepatic inflammation in rats with experimental small intestinal bacterial overgrowth. Gastroenterology 98: 414–423
Lichtman SN et al. (1991) Biliary tract disease in rats with experimental small bowel bacterial overgrowth. Hepatology 13: 766–772
Lichtman SN et al. (1991) Hepatic injury associated with small bowel bacterial overgrowth in rats is prevented by metronidazole and tetracycline. Gastroenterology 100: 513–519
Freund HR et al. (1985) A possible beneficial effect of metronidazole in reducing TPN-associated liver function derangements. J Surg Res 38: 356–363
Nanji AA and Anderson FH (1984) Cholestasis associated with parenteral nutrition develops more commonly with hematologic malignancy than with inflammatory bowel disease. JPEN J Parenter Enteral Nutr 8: 325
Fouin-Fortunet H et al. (1982) Hepatic alterations during total parenteral nutrition in patients with inflammatory bowel disease: a possible consequence of lithocholate toxicity. Gastroenterology 82: 932–937
Cruccetti A et al. (2003) Surgical infants on total parenteral nutrition have impaired cytokine responses to microbial challenge. J Pediatr Surg 38: 138–142
Zheng YJ et al. (2004) Endotoxin and cytokine released during parenteral nutrition. JPEN J Parenter Enteral Nutr 28: 163–168
Forrest EH et al. (2002) Improvement in cholestasis associated with total parenteral nutrition after treatment with an antibody against tumour necrosis factor alpha. Liver 22: 317–320
Trauner M et al. (1999) Inflammation-induced cholestasis. J Gastroenterol Hepatol 14: 946–959
Crawford JM and Boyer JL (1998) Clinicopathology conferences: inflammation-induced cholestasis. Hepatology 28: 253–260
Trauner M et al. (1998) Endotoxin downregulates rat hepatic ntcp gene expression via decreased activity of critical transcription factors. J Clin Invest 101: 2092–2100
Ghose R et al. (2004) Endotoxin leads to rapid subcellular re-localization of hepatic RXRα: a novel mechanism for reduced hepatic gene expression in inflammation. Nucl Recept 2: 4
Li D et al. (2002) Interleukin-1 beta-mediated suppression of RXR:RAR transactivation of the Ntcp promoter is JNK-dependent. J Biol Chem 277: 31416–31422
Green RM (1996) Regulation of hepatocyte bile salt transporters by endotoxin and inflammatory cytokines in rodents. Gastroenterology 111: 193–198
Denson LA et al. (2000) Interleukin-1beta suppresses retinoid transactivation of two hepatic transporter genes involved in bile formation. J Biol Chem 275: 8835–8843
Lavoie JC et al. (2005) Reduced bile flow associated with parenteral nutrition is independent of oxidant load and parenteral multivitamins. J Pediatr Gastroenterol Nutr 41: 108–114
Zlotkin SH and Anderson GH (1982) The development of cystathionase activity during the first year of life. Pediatr Res 16: 65–68
Cooke RJ et al. (1984) Effect of taurine supplementation on hepatic function during short-term parenteral nutrition in the premature infant. J Pediatr Gastroenterol Nutr 3: 234–238
Guertin F et al. (1991) Effect of taurine on total parenteral nutrition-associated cholestasis. JPEN J Parenter Enteral Nutr 15: 247–251
Spencer AU et al. (2005) Pediatric short bowel syndrome: redefining predictors of success. Ann Surg 242: 403–409
Moreno A et al. (1994) Aluminum in the neonate related to parenteral nutrition. Acta Paediatr 83: 25–29
Rabinow BE et al. (1989) Aluminum in parenteral products: analysis, reduction, and implications for pediatric TPN. J Parenter Sci Technol 43: 132–139
Moukarzel AA et al. (1992) Excessive chromium intake in children receiving total parenteral nutrition. Lancet 339: 385–388
Bougle D et al. (1993) Chromium and parenteral nutrition in children. J Pediatr Gastroenterol Nutr 17: 72–74
Kafritsa Y et al. (1998) Long-term outcome of brain manganese deposition in patients on home parenteral nutrition. Arch Dis Child 79: 263–265
Sheldon GF et al. (1978) Hepatic dysfunction during hyperalimentation. Arch Surg 113: 504–508
Mashima Y (1979) Effect of calorie overload on puppy livers during parenteral nutrition. JPEN J Parenter Enteral Nutr 3: 139–145
Vileisis RA et al. (1980) Prospective controlled study of parenteral nutrition-associated cholestatic jaundice: effect of protein intake. J Pediatr 96: 893–897
Graham MF (1984) Inhibition of bile flow in the isolated perfused rat liver by a synthetic parenteral amino acid mixture: associated net amino acid fluxes. Hepatology 4: 69–73
Shenai JP and Borum PR (1984) Tissue carnitine reserves of newborn infants. Pediatr Res 18: 679–682
Bowyer BA et al. (1988) L-carnitine therapy in home parenteral nutrition patients with abnormal liver tests and low plasma carnitine concentrations. Gastroenterology 94: 434–438
Buchman AL et al. (2001) Choline deficiency causes reversible hepatic abnormalities in patients receiving parenteral nutrition: proof of a human choline requirement: a placebo-controlled trial. JPEN J Parenter Enteral Nutr 25: 260–268
Cavicchi M et al. (2000) Prevalence of liver disease and contributing factors in patients receiving home parenteral nutrition for permanent intestinal failure. Ann Intern Med 132: 525–532
Colomb V et al. (2000) Role of lipid emulsions in cholestasis associated with long-term parenteral nutrition in children. JPEN J Parenter Enteral Nutr 24: 345–350
Carter BA et al. (2006) Soy lipid-derived stigmasterol (Stig) antagonizes bile acid (BA)-activation of the FXR dependent BA sinusoidal efflux pump genes, OSTα/β, in primary mouse hepatocytes and HepG2cells [abstract #69]. J Pediatr Gastroenterol Nutr 43: E35
Carter BA et al. (2005) Soy-lipid derived stigmasterol suppresses FXR target genes BSEP and FGF-19, in human hepatoblastoma cells—potential role in total parenteral nutrition-associated cholestasis (TPNAC) [abstract #190]. J Pediatr Gastroenterol Nutr 41: 552
Clayton PT et al. (1993) Phytosterolemia in children with parenteral nutrition-associated cholestatic liver disease. Gastroenterology 105: 1806–1813
Gura KM et al. (2006) Reversal of parenteral nutrition-associated liver disease in two infants with short bowel syndrome using parenteral fish oil: implications for future management. Pediatrics 118: e197–e201
Tazuke Y et al. (2004) Hepatic P-glycoprotein changes with total parenteral nutrition administration. JPEN J Parenter Enteral Nutr 28: 1–6
Tazuke Y (2004) Effects of lipid administration on liver apoptotic signals in a mouse model of total parenteral nutrition (TPN). Pediatr Surg Int 20: 224–228
Javid PJ et al. (2005) The route of lipid administration affects parenteral nutrition-induced hepatic steatosis in a mouse model. J Pediatr Surg 40: 1446–1453
Beuers U (2006) Drug insight: mechanisms and sites of action of ursodeoxycholic acid in cholestasis. Nat Clin Pract Gastroenterol Hepatol 3: 318–328
Gunsar C et al. (2002) The biochemical and histopathological effects of ursodeoxycholic acid and metronidazole on total parenteral nutrition-associated hepatic dysfunction: an experimental study. Hepatogastroenterology 49: 497–500
De Marco G et al. (2006) Early treatment with ursodeoxycholic acid for cholestasis in children on parenteral nutrition because of primary intestinal failure. Aliment Pharmacol Ther 24: 387–394
Beau P et al. (1994) Is ursodeoxycholic acid an effective therapy for total parenteral nutrition-related liver disease. J Hepatol 20: 240–244
Lindor KD and Burnes J (1991) Ursodeoxycholic acid for the treatment of home parenteral nutrition-associated cholestasis: a case report. Gastroenterology 101: 250–253
Spagnuolo MI et al. (1996) Ursodeoxycholic acid for treatment of cholestasis in children on long-term total parenteral nutrition: a pilot study. Gastroenterology 111: 716–719
Tsai S et al. (2005) Failure of cholecystokinin-octapeptide to prevent TPN-associated gallstone disease. J Pediatr Surg 40: 263–267
Teitelbaum DH et al. (2005) Use of cholecystokinin-octapeptide for the prevention of parenteral nutrition-associated cholestasis. Pediatrics 115: 1332–1340
Heubi JE et al. (2002) Tauroursodeoxycholic acid (TUDCA) in the prevention of total parenteral nutrition-associated liver disease. J Pediatr 141: 237–242
Barshes NR (2006) Isolated orthotopic liver transplantation for parenteral nutrition-associated liver injury. JPEN J Parenter Enteral Nutr 30: 526–529
Acknowledgements
We would like to thank Saul J Karpen, MD, PhD, and members of the Texas Children's Liver Center Laboratory for intellectual contributions to the experimental data present in this manuscript. The authors would also like to thank Milton J Finegold, MD, Chief of Pediatric Pathology at Texas Children's Hospital, Baylor College of Medicine, for allowing us to include in this manuscript slides of gross and histopathological PNAC from his personal collection. Work in BA Carter's laboratory has been supported by the American Gastroenterological Association/Roche, Child Health Research Center K12 HD41648-04, and the Children's Digestive Health and Nutrition Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Carter, B., Shulman, R. Mechanisms of Disease: update on the molecular etiology and fundamentals of parenteral nutrition associated cholestasis. Nat Rev Gastroenterol Hepatol 4, 277–287 (2007). https://doi.org/10.1038/ncpgasthep0796
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpgasthep0796
This article is cited by
-
Human induced pluripotent stem cell derived hepatocytes provide insights on parenteral nutrition associated cholestasis in the immature liver
Scientific Reports (2021)
-
Modified Rat-Tail-Fixed Model of Parenteral Nutrition to Study Liver Injury
Indian Journal of Surgery (2020)
-
Cholestatic liver (dys)function during sepsis and other critical illnesses
Intensive Care Medicine (2016)