Abstract
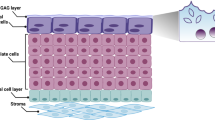
Although the urinary bladder urothelium has classically been thought of as a passive barrier to ions and solutes, a number of novel properties have been recently attributed to urothelial cells. Studies have revealed that the urothelium is involved in sensory mechanisms (i.e. the ability to express a number of sensor molecules or respond to thermal, mechanical and chemical stimuli) and can release chemical mediators. Localization of afferent nerves next to the urothelium suggests that urothelial cells could be targets for neurotransmitters released from bladder nerves or that chemicals released by urothelial cells could alter afferent nerve excitability. Taken together, these and other findings highlighted in this article suggest a sensory function for the urothelium. Elucidation of mechanisms that influence urothelial function might provide insights into the pathology of bladder dysfunction.
Key Points
-
The urothelium, a specialized lining of the urinary tract, has historically been viewed as a passive barrier to ions and solutes
-
There is evidence that the urothelium responds to both physiological and chemical stimuli and can release a number of signaling molecules
-
Release of chemical mediators from urothelial cells indicates that these cells have specialized sensory and signaling properties that could allow reciprocal communication with neighboring urothelial cells, as well as afferent and efferent nerves or other cells (i.e. myofibroblasts and immune or inflammatory cells) within the bladder wall
-
Various types of transient receptor potential channels including TRPV1 are expressed in the urothelium as well as in bladder afferent nerves
-
Results from TRPV1-null mice demonstrate that TRPV1 receptors are essential for normal mechanically evoked purinergic signaling by the urothelium, and indicate that the function of these receptors extends beyond pain sensation to include participation in normal bladder function
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Apodaca G (2004) The uroepithelium: not just a passive barrier. Traffic 5: 117–128
Lewis SA (2000) Everything you wanted to know about the bladder epithelium but were afraid to ask. Am J Physiol Renal Physiol 278: F867–F874
Wang E et al. (2003) Hydrostatic pressure-regulated ion transport in bladder uroepithelium. Am J Physiol Renal Physiol 285: F651–F663
Tammela T et al. (1993) Urothelial permeability of the isolated whole bladder. Neurourol Urodyn 12: 39–47
Romih R et al. (2005) Differentiation of epithelial cells in the urinary tract. Cell Tissue Res 320: 259–268
Martin BF (1972) Cell replacement and differentiation in transitional epithelium: a histological and autoradiographic study of the guinea-pig bladder and ureter. J Anatomy 112: 433–455
Hicks M (1975) The mammalian urinary bladder: an accommodating organ. Biol Rev 50: 215–246
Brady CM et al. (2004) Parallel changes in bladder suburothelial vanilloid receptor TRPV1 and pan-neuronal marker PGP9.5 immunoreactivity in patients with neurogenic detrusor overactivity after intravesical resiniferatoxin treatment. BJU Int 93: 770–776
Acharya P et al. (2004) Distribution of the tight junction proteins ZO-1, occludin, and claudin-4, -8, and -12 in bladder epithelium. Am J Physiol Renal Physiol 287: F305–F318
Parsons CL et al. (1979) Antibacterial activity of bladder surface mucin duplicated by exogenous glycosaminoglycan (heparin). Infect Immun 24: 552–557
Lavelle J et al. (2002) Bladder permeability barrier: recovery from selective injury of surface epithelial cells. Am J Physiol Renal Physiol 283: F242–F253
Bassuk JA et al. (2003) Induction of urothelial cell proliferation by fibroblast growth factor-7 in RAG1-deficient mice. Adv Exp Med Biol 539: 623–633
de Boer WI et al. (1996) Functions of fibroblast and transforming growth factors in primary organ-like cultures of normal human urothelium. Lab Invest 75: 147–156
Anderson G et al. (2003) Intracellular bacterial biofilm-like pods in urinary tract infections. Science 301: 105–107
Apodaca G et al. (2003) Disruption of bladder epithelium barrier function after spinal cord injury. Am J Physiol Renal Physiol 284: F966–F976
Truschel ST et al. (2001) Involvement of nitric oxide (NO) in bladder afferent and urothelial abnormalities following chronic spinal cord injury [abstract #842.9]. Soc Neurosci Abstr Program
Keay SK et al. (2004) An antiproliferative factor from interstitial cystitis patients is a Frizzled 8 protein-related sialoglycopeptide. Proc Natl Acad Sci USA 101: 11803–11808
Conrads TP et al.: CKAP4/p63 is a receptor for the frizzled-8 protein-related antiproliferative factor from interstitial cystitis patients. J Biol Chem, in press
Schilling J and Hultgren S (2002) Recent advances into the pathogenesis of recurrent urinary tract infections: the bladder as a reservoir for uropathogenic Escherichia coli. Int J Antimicrob Agents 19: 457–460
Birder L et al. (2001) Vanilloid receptor expression suggests a sensory role for urinary bladder epithelial cells. Proc Natl Acad Sci USA 98: 13396–13401
Birder L et al. (2002) Altered urinary bladder function in mice lacking the vanilloid receptor TRPV1. Nat Neurosci 5: 856–860
Dickson A et al. (2006) Peptidergic sensory and parasympathetic fiber sprouting in the mucosa of the rat urinary bladder in a chronic model of cyclophosphamide-induced cystitis. Neuroscience 139: 671–685
Jen PY et al. (1995) Immunohistochemical localization of neuromarkers and neuropeptides in human fetal and neonatal urinary bladder. Br J Pharmacol 75: 230–235
Kunze A et al. (2006) Quantitative immunohistochemical study of the innervation of the guinea-pig lower urinary tract. Br J Urol 98: 424–429
Sui GP et al. (2002) Gap junctions and connexin expression in human suburothelial interstitial cells. BJU Int 90: 118–129
Wiseman OJ et al. (2002) The ultrastructure of bladder lamina propria nerves in healthy subjects and patients with detrusor hyperreflexia. J Urol 168: 2040–2045
Brading AF and McCloskey KD (2005) Mechanisms of disease: specialized interstitial cells of the urinary tract—an assessment of current knowledge. Nat Clin Pract Urol 2: 546–554
Birder L et al. (2002) Beta-adrenoceptor agonists stimulate endothelial nitric oxide synthase in rat urinary bladder urothelial cells. J Neurosci 22: 8063–8070
Birder L et al. (2003) Feline interstitial cystitis results in mechanical hypersensitivity and altered ATP release from bladder urothelium. Am J Physiol Renal Physiol 285: F423–F429
Burnstock G (2001) Purine-mediated signalling in pain and visceral perception. Trends Pharmacol Sci 22: 182–188
Chess-Williams R (2002) Muscarinic receptors of the urinary bladder: detrusor, urothelial and prejunctional. Auton Autacoid Pharmacol 22: 133–145
Chopra B et al. (2005) Expression and function of bradykinin B1 and B2 receptors in normal and inflamed rat urinary bladder urothelium. J Physiol 562: 859–871
Ferguson DR et al. (1997) ATP is released from rabbit urinary bladder epithelial cells by hydrostatic pressure changes—a possible sensory mechanism? J Physiol 505: 503–511
Beckel J et al. (2006) Expression of functional nicotinic acetylcholine receptors in rat bladder epithelial cells. Am J Physiol 290: F103–F110
Petruska JC and Mendell LM (2004) The many functions of nerve growth factor: multiple actions on nociceptors. Neurosci Lett 361: 168–171
Wolf-Johnston AS et al. (2003) Increased NGF and TRPV1 expression in urinary bladder and sensory neurons from cats with feline interstitial cystitis [abstract 608.4]. Soc Neurosci Abstr Program
Ossovskaya VS and Bunnett NW (2005) Protease-activated receptors: contribution to physiology and disease. Physiol Rev 84: 579–621
Carattino MD et al. (2005) Mutations in the pore region modify epithelial sodium channel gating by shear stress. J Biol Chem 280: 4393–4401
Kanai AJ et al. (2006) Origin of spontaneous activity in neonatal and adult rat bladders and its enhancement by stretch and muscarinic agonists. Am J Physiol Renal Physiol, in press
Wang E et al. (2005) ATP and purinergic receptor-dependent membrane traffic in bladder umbrella cells. J Clin Invest 115: 2412–2422
Cockayne DA et al. (2000) Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature 407: 1011–1015
Brady CM et al. (2004) P2X3-immunoreactive nerve fibres in neurogenic detrusor overactivity and the effect of intravesical resiniferatoxin. Eur Urol 46: 247–253
Sun Y et al. (2001) Augmented stretch activated adenosine triphosphate release from bladder uroepithelial cells in patients with interstitial cystitis. J Urol 166: 1951–1956
Burnstock G (2006) Purinergic signaling. Br J Pharmacol 147 (Suppl 1): S172–S181
Andersson KE and Hedlund P (2002) Pharmacologic perspective on the physiology of the lower urinary tract. Urology 60: 13–20
Tominaga M et al. (2001) Potentiation of capsaicin receptor activation by metabotropic ATP receptors: a possible mechanism for ATP-evoked pain and hypersensitivity. Proc Natl Acad Sci USA 29: 820–825
Nickel JC (2003) Interstitial cystitis—an elusive clinical target? J Urol 170: 816–817
Parsons CL et al. (1998) The role of urinary potassium in the pathogenesis and diagnosis of interstitial cystitis. J Urol 159: 1862–1867
Gillenwater JY and Wein AJ (1998) Summary of the National Institute of Arthritis, Diabetes, Digestive and Kidney Diseases workshop on interstitial cystitis, National Institutes of Health, Bethesda, Maryland, August 28–29, 1987. J Urol 140: 203–206
Birder L et al. (2004) Alterations in P2X and P2Y purinergic receptor expression in urinary bladder from normal cats and cats with interstitial cystitis. Am J Physiol Renal Physiol 287: F1084–F1091
Wellner MC and Isenberg G (1994) Stretch effects on whole-cell currents of guinea pig urinary bladder myocytes. J Physiol 480: 439–448
Caterina MJ (2001) The vanilloid receptor: a molecular gateway to the pain pathway. Ann Rev Neurosci 24: 602–607
Caterina MJ et al. (1997) The capsaicin receptor: a heat activated ion channel in the pain pathway. Nature 389: 816–824
Maggi CA (1992) Therapeutic potential of capsaicin-like molecules: studies in animals and humans. Life Sci 51: 1777–1781
Chancellor MB and de Groat WC (1999) Intravesical capsaicin and resiniferatoxin therapy: spicing up the ways to treat the overactive bladder. J Urol 162: 3–11
Staskin DR and MacDiarmid SA (2006) Using anticholinergics to treat overactive bladder: the issue of treatment tolerability. Am J Med 119: 9–15
Pathak AS and Aboseif SR (2005) Overactive bladder: drug therapy versus nerve stimulation. Nat Clin Pract Urol 2: 310–311
Abrams P et al. (2006) Muscarinic receptors: their distribution and function in body systems, and the implications for treating overactive bladder. Br J Pharmacol 148: 565–578
Hegde SS (2006) Muscarinic receptors in the bladder: from basic research to therapeutics. Br J Pharmacol 147 (Suppl 2): S80–S87
Yoshida M et al. (2006) Non-neuronal cholinergic system in human bladder urothelium. Urology 67: 425–430
Tiwari A and Naruganahalli KS (2006) Current and emerging investigational medical therapies for the treatment of overactive bladder. Expert Opin Investig Drugs 15: 1017–1037
Chancellor MB (2005) Urgency, botulinum toxin and how botulinum toxin can help urgency. J Urol 174: 818
Toft BR and Nordling J (2006) Recent developments of intravesical therapy of painful bladder syndrome/interstitial cystitis: a review. Current Opin Urol 16: 268–272
Apostolidis A et al. (2006) Proposed mechanism for the efficacy of injected botulinum toxin in the treatment of human detrusor overactivity. Eur Urol 49: 644–650
Smith CP et al. (2005) Enhanced ATP release from rat bladder urothelium during chronic bladder inflammation: effect of botulinum toxin A. Neurochem Int 47: 291–297
Birder L et al. (1998) Adrenergic- and capsaicin-evoked nitric oxide release from urothelium and afferent nerves in urinary bladder. Am J Physiol Renal Physiol 275: F226–F229
Inoue T and Gabella G (1991) A vascular network closely linked to the epithelium of the urinary bladder of the rat. Cell Tissue Res 263: 137–143
Toh KL and Ng CK (2006) Urodynamic studies in the evaluation of young men presenting with lower urinary tract symptoms. Int J Urol 13: 520–523
Yassin A et al. (2006) Alpha-adrenoceptors are a common demoninator in the pathophysiology of erectile function and BPH/LUTS—implications for clinical practice. Andrologia 38: 1–12
Chan Q et al. (2005) Function of the lower urinary tract in mice lacking alpha1D-adrenoceptor. J Urol 174: 370–374
Ishihama H et al. (2006) Activation of alpha1D adrenergic receptors in the rat urothelium facilitates the micturition reflex. J Urol 175: 358–364
Buffington CA et al. (2002) Norepinephrine content and adrenoceptor function in the bladder of cats with feline interstitial cystitis. J Urol 167: 1876–1880
Birder L et al. (2005) Altered inducible nitric oxide synthase expression and nitric oxide production in urinary bladder from cats with feline interstitial cystitis. J Urol 173: 625–629
Andersson KE and Persson K (1995) Nitric oxide synthase and the lower urinary tract: possible implications for physiology and pathophysiology. Scand J Urol Nephrol Suppl 175: 43–53
Pandita RK et al. (2000) Intravesical oxyhemoglobin initiates bladder overactivity in conscious, normal rats. J Urol 164: 545–550
de Groat WC et al. (1997) Modification of urinary bladder function after spinal cord injury. Adv Neurol 72: 347–364
Ozawa H et al. (1999) Effect of intravesical nitric oxide therapy on cyclophosphamide-induced cystitis. J Urol 162: 2211–2216
Korkmaz A et al. (2005) Peroxynitrite may be involved in bladder damage caused by cyclophosphamide in rats. J Urol 173: 1793–1796
Hosseini A et al. (2004) Nitric oxide as an objective marker for evaluation of treatment response in patients with classic interstitial cystitis. J Urol 172: 2261–2265
Kolios G et al. (2004) Nitric oxide in inflammatory bowel disease: a universal messenger in an unsolved puzzle. Immunology 113: 427–437
Acknowledgements
This work was supported by NIH grants to L Birder (RO1 DK54824 and RO1 DK57284).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Lori Birder has associations with the following companies: Hydra Biosciences Inc., Roche Palo Alto LLC, Dynogen Pharmaceuticals Inc., Pfizer Inc., and Omeros Corporation.
William C de Groat has associations with the following companies: Abbott Laboratories, Roche Palo Alto LLC, Eli Lilly and Company, Pfizer Inc., Novartis AG, Dynogen Pharmaceuticals Inc., Johnson & Johnson, Lilly ICOS LLC, Astellas Pharma Inc., Hydra Biosciences Inc., Boehringer Ingleheim, Allergan Inc. and Sanofi Aventis.
Rights and permissions
About this article
Cite this article
Birder, L., de Groat, W. Mechanisms of Disease: involvement of the urothelium in bladder dysfunction. Nat Rev Urol 4, 46–54 (2007). https://doi.org/10.1038/ncpuro0672
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpuro0672
This article is cited by
-
De-obstruction of bladder outlet in humans reverses organ remodelling by normalizing the expression of key transcription factors
BMC Urology (2024)
-
A biophysically comprehensive model of urothelial afferent neurons: implications for sensory signalling in urinary bladder
Journal of Computational Neuroscience (2024)
-
Role of calcium channel blockers in lower urinary tract symptoms in benign prostatic hyperplasia: a literature review
African Journal of Urology (2022)
-
The urothelium: a multi-faceted barrier against a harsh environment
Mucosal Immunology (2022)
-
Potential role of oxidative stress in the pathogenesis of diabetic bladder dysfunction
Nature Reviews Urology (2022)