Abstract
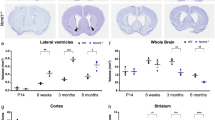
Mecp2 is an X-linked gene encoding a nuclear protein that binds specifically to methylated DNA (ref. 1) and functions as a general transcriptional repressor by associating with chromatin-remodeling complexes2,3. Mecp2 is expressed at high levels in the postnatal brain1,4, indicating that methylation-dependent regulation of gene expression may have a crucial role in the mammalian central nervous system. Consistent with this notion is the recent demonstration that MECP2 mutations cause Rett syndrome5,6,7,8 (RTT, MIM 312750), a childhood neurological disorder that represents one of the most common causes of mental retardation in females9,10,11. Here we show that Mecp2-deficient mice exhibit phenotypes that resemble some of the symptoms of RTT patients. Mecp2-null mice were normal until 5 weeks of age, when they began to develop disease, leading to death between 6 and 12 weeks. Mutant brains showed substantial reduction in both weight and neuronal cell size, but no obvious structural defects or signs of neurodegeneration. Brain-specific deletion of Mecp2 at embryonic day (E) 12 resulted in a phenotype identical to that of the null mutation, indicating that the phenotype is caused by Mecp2 deficiency in the CNS rather than in peripheral tissues. Deletion of Mecp2 in postnatal CNS neurons led to a similar neuronal phenotype, although at a later age. Our results indicate that the role of Mecp2 is not restricted to the immature brain, but becomes critical in mature neurons. Mecp2 deficiency in these neurons is sufficient to cause neuronal dysfunction with symptomatic manifestation similar to Rett syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lewis, J.D. et al. Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 69, 905–914 (1992).
Nan, X. et al. Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393, 386–389 (1998).
Jones, P.L. et al. Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nature Genet. 19, 187–191 (1998).
Coy, J.F., Sedlacek, Z., Bachner, D., Delius, H. & Poustka, A. A complex pattern of evolutionary conservation and alternative polyadenylation within the long 3"-untranslated region of the methyl-CpG-binding protein 2 gene (MeCP2) suggests a regulatory role in gene expression. Hum. Mol. Genet. 8, 1253–1262 (1999).
Amir, R.E. et al. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nature Genet. 23, 185–188 (1999).
Wan, M. et al. Rett syndrome and beyond: recurrent spontaneous and familial MECP2 mutations at CpG hotspots. Am. J. Hum. Genet. 65, 1520–1529 (1999).
Van den Veyver, I.B. & Zoghbi, H.Y. Methyl-CpG-binding protein 2 mutations in Rett syndrome. Curr. Opin. Genet. Dev. 10, 275–279 (2000).
Dragich, J., Houwink-Manville, I. & Schanen, C. Rett syndrome: a surprising result of mutation in MECP2. Hum. Mol. Genet. 9, 2365–2375 (2000).
Rett, A. Uber ein eigenartiges hirnatrophisches Syndrom bei Hyperammonamie im Kindersalter. Wien Med. Wochenschr. 116, 723–726 (1966).
Hagberg, B., Aicardi, J., Dias, K. & Ramos, O. A progressive syndrome of autism, dementia, ataxia, and loss of purposeful hand use in girls: Rett's syndrome: report of 35 cases. Ann. Neurol. 14, 471–479 (1983).
Armstrong, D.D. Review of Rett syndrome. J. Neuropathol. Exp. Neurol. 56, 843–849 (1997).
Tate, P., Skarnes, W. & Bird, A. The methyl-CpG binding protein MeCP2 is essential for embryonic development in the mouse. Nature Genet. 12, 205–208 (1996).
Ellaway, C. & Christodoulou, J. Rett syndrome: clinical update and review of recent genetic advances. J. Paediatr. Child Health 35, 419–426 (1999).
Schanen, N.C. et al. Neonatal encephalopathy in two boys in families with recurrent Rett syndrome. J. Child Neurol. 13, 229–231 (1998).
Schanen, C. & Francke, U. A severely affected male born into a Rett syndrome kindred supports X-linked inheritance and allows extension of the exclusion map. Am. J. Hum. Genet. 63, 267–269 (1998).
Villard, L., Cardoso, A.K., Chelly, P.J., Tardieu, P.M. & Fontes, M. Two affected boys in a Rett syndrome family: clinical and molecular findings. Neurology 55, 1188–1193 (2000).
Meloni, I. et al. A mutation in the rett syndrome gene, MECP2, causes X-linked mental retardation and progressive spasticity in males. Am. J. Hum. Genet. 67, 982–985 (2000).
Clayton-Smith, J., Watson, P., Ramsden, S. & Black, G.C. Somatic mutation in MECP2 as a non-fatal neurodevelopmental disorder in males. Lancet 356, 830–832 (2000).
Orrico, A. et al. MECP2 mutation in male patients with non-specific X-linked mental retardation. FEBS Lett. 481, 285–288 (2000).
Trumpp, A., Depew, M.J., Rubenstein, J.L., Bishop, J.M. & Martin, G.R. Cre-mediated gene inactivation demonstrates that FGF8 is required for cell survival and patterning of the first branchial arch. Genes Dev. 13, 3136–3148 (1999).
Fan, G. et al. DNA hypomethylation perturbs the function and survival of CNS neurons in postnatal animals. J. Neurosci. 21, 788–797 (2001).
Guy, J., Hendrich, B., Martin, J.E. & Bird, A. A mouse Mecp2-null mutation causes neurological symptoms that mimic Rett syndrome. Nature Genet. 27, 322–326 (2001).
Jellinger, K., Armstrong, D., Zoghbi, H.Y. & Percy, A.K. Neuropathology of Rett syndrome. Acta Neuropathol. 76, 142–158 (1988).
Hagberg, G., Stenbom, Y. & Witt Engerstrom, I. Head growth in Rett syndrome. Acta Paediatr. 89, 198–202 (2000).
Bauman, M.L., Kemper, T.L. & Arin, D.M. Microscopic observations of the brain in Rett syndrome. Neuropediatrics 26, 105–108 (1995).
Bauman, M.L., Kemper, T.L. & Arin, D.M. Pervasive neuroanatomic abnormalities of the brain in three cases of Rett's syndrome. Neurology 45, 1581–1586 (1995).
Minichiello, L. et al. Essential role for TrkB receptors in hippocampus-mediated learning. Neuron 24, 401–414 (1999).
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nature Genet. 21, 70–71 (1999).
Dragatsis, I. & Zeitlin, S. CaMKIIα-Cre transgene expression and recombination patterns in the mouse brain. Genesis 26, 133–135 (2000).
Acknowledgements
We thank U. Francke, L. Jackson-Grusby and G. Fan for critical reading of the manuscript; J. Reis, R. Flannery, J. Loring and J. Dausman for technical support; A. Bird for providing mice carrying an independently generated Mecp2 conditional allele; and the Howard Hughes Medical Institute, the National Institutes of Health, the Rett Syndrome Research Foundation and the Cure Autism Now Foundation for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, R., Akbarian, S., Tudor, M. et al. Deficiency of methyl-CpG binding protein-2 in CNS neurons results in a Rett-like phenotype in mice. Nat Genet 27, 327–331 (2001). https://doi.org/10.1038/85906
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85906
This article is cited by
-
tRNA epitranscriptomic alterations associated with opioid-induced reward-seeking and long-term opioid withdrawal in male mice
Neuropsychopharmacology (2024)
-
RNA methyltransferase NSun2 deficiency promotes neurodegeneration through epitranscriptomic regulation of tau phosphorylation
Acta Neuropathologica (2023)
-
Extension of the Lifespan of a Mouse Model of Rett Syndrome by Intracerebroventricular Delivery of MECP2
Neuroscience Bulletin (2023)
-
Progress on the roles of MEF2C in neuropsychiatric diseases
Molecular Brain (2022)
-
Modeling human neurodevelopmental diseases with brain organoids
Cell Regeneration (2022)