Abstract
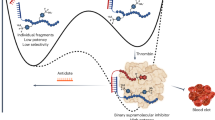
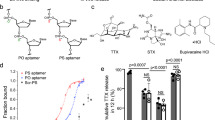
With an ever increasing number of people taking numerous medications, the need to safely administer drugs and limit unintended side effects has never been greater. Antidote control remains the most direct means to counteract acute side effects of drugs, but, unfortunately, it has been challenging and cost prohibitive to generate antidotes for most therapeutic agents. Here we describe the development of a set of antidote molecules that are capable of counteracting the effects of an entire class of therapeutic agents based upon aptamers. These universal antidotes exploit the fact that, when systemically administered, aptamers are the only free extracellular oligonucleotides found in circulation. We show that protein- and polymer-based molecules that capture oligonucleotides can reverse the activity of several aptamers in vitro and counteract aptamer activity in vivo. The availability of universal antidotes to control the activity of any aptamer suggests that aptamers may be a particularly safe class of therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dyke, C.K. et al. First-in-human experience of an antidote-controlled anticoagulant using RNA aptamer technology: a phase 1a pharmacodynamic evaluation of a drug-antidote pair for the controlled regulation of factor IXa activity. Circulation 114, 2490–2497 (2006).
EyetechStudyGroup. Preclinical and phase 1A clinical evaluation of an anti-VEGF pegylated aptamer (EYE001) for the treatment of exudative age-related macular degeneration. Retina 22, 143–152 (2002).
EyetechStudyGroup. Anti-vascular endothelial growth factor therapy for subfoveal choroidal neovascularization secondary to age-related macular degeneration: phase II study results. Ophthalmology 110, 979–986 (2003).
Gilbert, J.C. et al. First-in-human evaluation of anti von Willebrand factor therapeutic aptamer ARC1779 in healthy volunteers. Circulation 116, 2678–2686 (2007).
Lee, J.H. et al. A therapeutic aptamer inhibits angiogenesis by specifically targeting the heparin binding domain of VEGF165. Proc. Natl. Acad. Sci. USA 102, 18902–18907 (2005).
McNamara, J.O. et al. Multivalent 4–1BB binding aptamers costimulate CD8+ T cells and inhibit tumor growth in mice. J. Clin. Invest. 118, 376–386 (2008).
Nimjee, S.M., Rusconi, C.P. & Sullenger, B.A. Aptamers: an emerging class of therapeutics. Annu. Rev. Med. 56, 555–583 (2005).
Sullenger, B.A., Gallardo, H.F., Ungers, G.E. & Gilboa, E. Overexpression of TAR sequences renders cells resistant to human immunodeficiency virus replication. Cell 63, 601–608 (1990).
Anderson, J. et al. Safety and efficacy of a lentiviral vector containing three anti-HIV genes—CCR5 ribozyme, tat-rev siRNA, and TAR decoy—in SCID-hu mouse–derived T cells. Mol. Ther. 15, 1182–1188 (2007).
Ellington, A.D. & Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990).
Tuerk, C. & Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249, 505–510 (1990).
Becker, R.C., Rusconi, C. & Sullenger, B. Nucleic acid aptamers in therapeutic anticoagulation. Technology, development and clinical application. Thromb. Haemost. 93, 1014–1020 (2005).
Nimjee, S.M., Rusconi, C.P., Harrington, R.A. & Sullenger, B.A. The potential of aptamers as anticoagulants. Trends Cardiovasc. Med. 15, 41–45 (2005).
Fanikos, J. et al. Adverse drug events in hospitalized cardiac patients. Am. J. Cardiol. 100, 1465–1469 (2007).
Nimjee, S.M. et al. A novel antidote-controlled anticoagulant reduces thrombin generation and inflammation and improves cardiac function in cardiopulmonary bypass surgery. Mol. Ther. 14, 408–415 (2006).
Rusconi, C.P. et al. RNA aptamers as reversible antagonists of coagulation factor IXa. Nature 419, 90–94 (2002).
Oney, S. et al. Antidote-controlled platelet inhibition targeting von Willebrand factor with aptamers. Oligonucleotides 17, 265–274 (2007).
Rusconi, C.P., Yeh, A., Lyerly, H.K., Lawson, J.H. & Sullenger, B.A. Blocking the initiation of coagulation by RNA aptamers to factor VIIa. Thromb. Haemost. 84, 841–848 (2000).
Rusconi, C.P. et al. Antidote-mediated control of an anticoagulant aptamer in vivo. Nat. Biotechnol. 22, 1423–1428 (2004).
Kleinman, M.E. et al. Sequence- and target-independent angiogenesis suppression by siRNA via TLR3. Nature 452, 591–597 (2008).
White, R. et al. Generation of species cross-reactive aptamers using “toggle” SELEX. Mol. Ther. 4, 567–573 (2001).
Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 31, 3406–3415 (2003).
Hird, R.B. et al. Direct effects of protamine sulfate on myocyte contractile processes. Cellular and molecular mechanisms. Circulation 92, II433–II446 (1995).
Porsche, R. & Brenner, Z.R. Allergy to protamine sulfate. Heart Lung 28, 418–428 (1999).
Shigeta, O. et al. Low-dose protamine based on heparin-protamine titration method reduces platelet dysfunction after cardiopulmonary bypass. J. Thorac. Cardiovasc. Surg. 118, 354–360 (1999).
Welsby, I.J. et al. Hemodynamic changes after protamine administration: association with mortality after coronary artery bypass surgery. Anesthesiology 102, 308–314 (2005).
Gonzalez, H., Hwang, S.J. & Davis, M.E. New class of polymers for the delivery of macromolecular therapeutics. Bioconjug. Chem. 10, 1068–1074 (1999).
Moore, T.J., Cohen, M.R. & Furberg, C.D. Serious adverse drug events reported to the Food and Drug Administration, 1998–2005. Arch. Intern. Med. 167, 1752–1759 (2007).
Lazarou, J., Pomeranz, B.H. & Corey, P.N. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. J. Am. Med. Assoc. 279, 1200–1205 (1998).
Carr, J.A. & Silverman, N. The heparin-protamine interaction. A review. J. Cardiovasc. Surg. (Torino) 40, 659–666 (1999).
Stanker, L.H., Wyrobek, A., McKeown, C. & Balhorn, R. Identification of the binding site of two monoclonal antibodies to human protamine. Mol. Immunol. 30, 1633–1638 (1993).
Davis, M.E. et al. Self-assembling nucleic acid delivery vehicles via linear, water-soluble, cyclodextrin-containing polymers. Curr. Med. Chem. 11, 179–197 (2004).
Mao, H.Q. & Leong, K.W. Design of polyphosphoester-DNA nanoparticles for non-viral gene delivery. Adv. Genet. 53, 275–306 (2005).
Joachimi, A., Mayer, G. & Hartig, J.S. A new anticoagulant-antidote pair: control of thrombin activity by aptamers and porphyrins. J. Am. Chem. Soc. 129, 3036–3037 (2007).
Schröder, M. & Bowie, A.G. TLR3 in antiviral immunity: key player or bystander? Trends Immunol. 26, 462–468 (2005).
Heidel, J.D. et al. Administration in non-human primates of escalating intravenous doses of targeted nanoparticles containing ribonucleotide reductase subunit M2 siRNA. Proc. Natl. Acad. Sci. USA 104, 5715–5721 (2007).
Harrison, P. Platelet function analysis. Blood Rev. 19, 111–123 (2005).
Acknowledgements
We thank S.M. Nimjee and J. Layzer for helpful discussions and Calando Pharmaceuticals for providing the CDP and CDP-Im polymers. This work was supported by a grant from the US National Institutes of Health (HL065222 to B.A.S.), a predoctoral fellowship from the American Heart Association (0615443U to S.O.) and a grant from the US National Cancer Institute (CA 119347 to M.E.D).
Author information
Authors and Affiliations
Contributions
S.O. designed and performed research, analyzed data and wrote the manuscript; R.T.S.L designed and performed research and analyzed data; K.M.B. performed research; C.M.B. performed research and analyzed data; G.Q. performed research; J.D.H. designed and performed research, provided useful reagents, analyzed data and provided useful discussions; J.Y.-C.L. performed research; B.C.M. performed research; M.E.D. provided useful reagents and discussions; K.W.L. provided useful reagents and discussions; B.A.S. suggested the universal antidote idea, designed and coordinated research, analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
B.A.S. and M.E.D. are scientific founders of Regado Biosciences, Inc. and Calando Pharmaceuticals, Inc., respectively.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1 and 2 and Supplementary Figs. 1–3 (PDF 1018 kb)
Rights and permissions
About this article
Cite this article
Oney, S., Lam, R., Bompiani, K. et al. Development of universal antidotes to control aptamer activity. Nat Med 15, 1224–1228 (2009). https://doi.org/10.1038/nm.1990
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1990
This article is cited by
-
Novel nanotherapeutics for cancer immunotherapy by albumin nanoparticles functionalized with PD-1 and PD-L1 aptamers
Cancer Nanotechnology (2024)
-
RNA aptamers with specific binding affinity to CD40 (CD40Apt) represents a promising antagonist of the CD40-CD40L signaling for thyroid-associated ophthalmopathy (TAO) treatment in mouse
Journal of Translational Medicine (2023)
-
In vitro selection of aptamers and their applications
Nature Reviews Methods Primers (2023)
-
Chimeric protein probes for C5a receptors through fusion of the anaphylatoxin C5a core region with a small-molecule antagonist
Science China Chemistry (2019)
-
Cationic nanoparticle as an inhibitor of cell-free DNA-induced inflammation
Nature Communications (2018)