Abstract
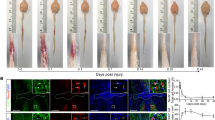
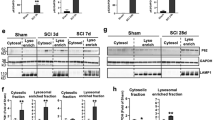
Traumatic spinal cord injury often results in complete loss of voluntary motor and sensory function below the site of injury. The long-term neurological deficits after spinal cord trauma may be due in part to widespread apoptosis of neurons and oligodendroglia in regions distant from and relatively unaffected by the initial injury1,2,3,4. The caspase family of cysteine proteases regulates the execution of the mammalian apoptotic cell death program5,6,7. Caspase-3 cleaves several essential downstream substrates involved in the expression of the apoptotic phenotype in vitro, including gelsolin, PAK2, fodrin, nuclear lamins and the inhibitory subunit of DNA fragmentation factor8,9,10,11,12. Caspase-3 activation in vitro can be triggered by upstream events, leading to the release of cytochrome c from the mitochondria and the subsequent transactivation of procaspase-9 by Apaf-1 (refs. 13, 14, 15). We report here that these upstream and downstream components of the caspase-3 apoptotic pathway are activated after traumatic spinal cord injury in rats, and occur early in neurons in the injury site and hours to days later in oligodendroglia adjacent to and distant from the injury site. Given these findings, targeting the upstream events of the caspase-3 cascade has therapeutic potential in the treatment of acute traumatic injury to the spinal cord.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Shuman, S.L., Bresnahan, J.C. & Beattie, M.S. Apoptosis of microglia and oligodendrocytes after spinal cord contusion in rats. J. Neurosci. Res. 50 , 798–808 (1997).
Liu, X.Z., et al. Neuronal and glial apoptosis after traumatic spinal cord injury. J. Neurosci. 17, 5395– 5406 (1997).
Crowe, M.J., Bresnahan, J.C., Shuman, S.L., Masters, J.N. & Beattie, M.S. Apoptosis and delayed degeneration after spinal cord injury in rats and monkeys. Nature Med. 3, 73–76 (1997).
Emery, E. et al. Apoptosis after traumatic human spinal cord injury. J. Neurosurg. 89, 911–20 (1998).
Cohen, G.M. Caspases: the executioners of apoptosis. Biochem. J. 326, 1–16 (1997).
Salvesen, G.S. & Dixit, V.M. Caspases: intracellular signaling by proteolysis. Cell 91, 443– 6 (1997).
Thornberry, N.A. The caspase family of cysteine proteases. Br. Med. Bull. 53, 478–490 (1997).
Enari, M. et al. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 391, 43– 50 (1998).
Sakahira, H., Enari, M. & Nagata, S. Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature 391, 96– 99 (1998).
Rudel, T. & Bokoch, G.M. Membrane and morphological changes in apoptotic cells regulated by caspase-mediated activation of PAK2. Science 276, 1571–1574 ( 1997).
Kluck, R.M., Bossy-Wetzel, E., Green, D.R. & Newmeyer, D.D. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275, 1132– 1136 (1997).
Kothakota, S. et al. Caspase-3-generated fragment of gelsolin: effector of morphological change in apoptosis. Science 278, 294– 298 (1997).
Pan, G., Humke, E.W. & Dixit, V.M. Activation of caspases triggered by cytochrome c in vitro. FEBS. Lett. 426, 151– 154 (1998).
Hu, Y., Ding, L., Spencer, D.M. & Nunez, G. WD-40 repeat region regulates Apaf-1 self-association and procaspase-9 activation. J. Biol. Chem. 273, 33489–33494 (1998).
Li, P. et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91, 479–489 (1997).
Liu, X., Kim, C.N., Yang, J., Jemmerson, R. & Wang, X. Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86, 147–157 (1996).
Leoni, L.M. et al. Induction of an apoptotic program in cell-free extracts by 2-chloro-2'- deoxyadenosine 5´-triphosphate and cytochrome c. Proc. Natl. Acad. Sci. USA 95, 9567– 9571 (1998).
Liu, X., Zou, H., Slaughter, C. & Wang, X. DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89, 175– 184 (1997).
Liu, X. et al. The 40-kDa subunit of DNA fragmentation factor induces DNA fragmentation and chromatin condensation during apoptosis. Proc. Natl. Acad. Sci. USA 95, 8461–8466 ( 1998).
Tang, D. & Kidd, V.J. Cleavage of DFF-45/ICAD by multiple caspases is essential for its function during apoptosis. J. Biol. Chem. 273, 28549–28552 ( 1998).
Yakovlev, A.G., et al. Activation of CPP32-like caspases contributes to neuronal apoptosis and neurological dysfunction after traumatic brain injury. J. Neurosci. 17, 7415–7424 (1997).
Pike, B.R. et al. Regional calpain and caspase-3 proteolysis of alpha-spectrin after traumatic brain injury. Neuroreport 9, 2437–2442 (1998).
Namura, S. et al. Activation and cleavage of caspase-3 in apoptosis induced by experimental cerebral ischemia. J. Neurosci. 18, 3659–3668 (1998).
Gruner, J. A monitored contusion model of spinal cord injury in the rat. J. Neurotrauma 9, 123–128 (1992).
Basso, D., Beattie, M. & Breshnahan, J. Graded histological and locomotor outcomes after spinal cord contusion using the NYU weight-drop device versus transection. Exp. Neurol. 139, 244–256 (1996).
Acknowledgements
We thank X. Wang for suggestions, B. Maley for assistance with the confocal images, and D.M. Gash and J.P. Herman for critically reviewing earlier versions of the manuscript. These studies were supported by PHS grant NS-30248 (J.E.S.) and grants from the Kentucky Spinal Cord and Head Injury Research Trust (J.E..S and P.E.K.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Springer, J., Azbill, R. & Knapp, P. Activation of the caspase-3 apoptotic cascade in traumatic spinal cord injury. Nat Med 5, 943–946 (1999). https://doi.org/10.1038/11387
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/11387
This article is cited by
-
The Role of Resveratrol on Spinal Cord Injury: from Bench to Bedside
Molecular Neurobiology (2024)
-
Restoring mitochondrial cardiolipin homeostasis reduces cell death and promotes recovery after spinal cord injury
Cell Death & Disease (2022)
-
Progression in translational research on spinal cord injury based on microenvironment imbalance
Bone Research (2022)
-
Neural stem cell-derived small extracellular vesicles attenuate apoptosis and neuroinflammation after traumatic spinal cord injury by activating autophagy
Cell Death & Disease (2019)
-
Clotam enhances anti-proliferative effect of vincristine in Ewing sarcoma cells
Apoptosis (2019)