Abstract
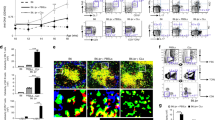
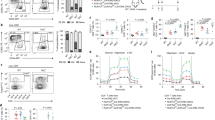
Autoimmune T-helper cells drive pathogenic autoantibody production in systemic lupus erythematosus (SLE), but the mechanisms maintaining those T cells are unknown. Autoreactive T cells are normally eliminated by functional inactivation (anergy) and activation-induced cell death (AICD) or apoptosis through death receptor (Fas) signaling1,2,3. However, mutations in the genes encoding Fas and its ligand (FasL) are rare in classical SLE4. By gene microarray profiling, validated by functional and biochemical studies, we establish here that activated T cells of lupus patients resist anergy and apoptosis by markedly upregulating and sustaining cyclooxygenase-2 (COX-2) expression. Inhibition of COX-2 caused apoptosis of the anergy-resistant lupus T cells by augmenting Fas signaling and markedly decreasing the survival molecule c-FLIP (cellular homolog of viral FLICE inhibitory protein). Studies with COX-2 inhibitors and Cox-2-deficient mice confirmed that this COX-2/FLIP antiapoptosis program is used selectively by anergy-resistant lupus T cells, and not by cancer cells or other autoimmune T cells. Notably, the gene encoding COX-2 is located in a lupus-susceptibility region on chromosome 1. We also found that only some COX-2 inhibitors were able to suppress the production of pathogenic autoantibodies to DNA by causing autoimmune T-cell apoptosis, an effect that was independent of prostaglandin E2 (PGE2). These findings could be useful in the design of lupus therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Refaeli, Y., Van Parijs, L., London, C.A., Tschopp, J. & Abbas, A.K. Biochemical mechanisms of IL-2-regulated Fas-mediated T cell apoptosis. Immunity 8, 615–623 (1998).
Green, D.R., Droin, N. & Pinkoski, M. Activation-induced cell death in T cells. Immunol. Rev. 193, 70–81 (2003).
Rathmell, J.C. & Thompson, C.B. Pathways of apoptosis in lymphocyte development, homeostasis, and disease. Cell 109, S97–S107 (2002).
Vaishnaw, A.K. et al. The spectrum of apoptotic defects and clinical manifestations, including systemic lupus erythematosus, in humans with CD95 (Fas/APO-1) mutations. Arthritis Rheum. 42, 1833–1842 (1999).
Yi, Y., McNerney, M. & Datta, S.K. Regulatory defects in Cbl and mitogen-activated protein kinase (extracellular signal-related kinase) pathways cause persistent hyperexpression of CD40 ligand in human lupus T cells. J. Immunol. 165, 6627–6634 (2000).
Pablos, J.L., Santiago, B., Carreira, P.E., Galindo, M. & Gomez-Reino, J.J. Cyclooxygenase-1 and -2 are expressed by human T cells. Clin. Exp. Immunol. 115, 86–90 (1999).
Lin, Z., Fillmore, G.C., Um, T.H., Elenitoba-Johnson, K.S. & Lim, M.S. Comparative microarray analysis of gene expression during activation of human peripheral blood T cells and leukemic Jurkat T cells. Lab. Invest. 83, 765–776 (2003).
Mitchell, T. et al. Immunological adjuvants promote activated T cell survival via induction of Bcl-3. Nat. Immunol. 2, 397–402 (2001).
Wakeland, E.K., Liu, K., Graham, R.R. & Behrens, T.W. Delineating the genetic basis of systemic lupus erythematosus. Immunity 15, 397–408 (2001).
Stack, E. & DuBois, R.N. Role of cyclooxygenase inhibitors for the prevention of colorectal cancer. Gastroenterol. Clin. North Am. 30, 1001–1010 (2001).
Zhu, J. et al. Using cyclooxygenase-2 inhibitors as molecular platforms to develop a new class of apoptosis-inducing agents. J. Natl. Cancer Inst. 94, 1745–1757 (2002).
Reddy, S.T. & Herschman, H.R. Ligand-induced prostaglandin synthesis requires expression of the TIS10/PGS-2 prostaglandin synthase gene in murine fibroblasts and macrophages. J. Biol. Chem. 269, 15473–15480 (1994).
Irmler, M. et al. Inhibition of death receptor signals by cellular FLIP. Nature 388, 190–195 (1997).
Lin, M.T., Lee, R.C., Yang, P.C., Ho, F.M. & Kuo, M.L. Cyclooxygenase-2 inducing Mcl-1-dependent survival mechanism in human lung adenocarcinoma CL1.0 cells. Involvement of phosphatidylinositol 3-kinase/Akt pathway. J. Biol. Chem. 276, 48997–49002 (2001).
Jones, R.G. et al. CD28-dependent activation of protein kinase B/Akt blocks Fas-mediated apoptosis by preventing death-inducing signaling complex assembly. J. Exp. Med. 196, 335–348 (2002).
Schirmer, M., Vallejo, A.N., Weyand, C.M. & Goronzy, J.J. Resistance to apoptosis and elevated expression of Bcl-2 in clonally expanded CD4+CD28− T cells from rheumatoid arthritis patients. J. Immunol. 161, 1018–1025 (1998).
Sharief, M.K. & Semra, Y.K. Heightened expression of survivin in activated T lymphocytes from patients with multiple sclerosis. J. Neuroimmunol. 119, 358–64 (2001).
Kirchhoff, S., Muller, W.W., Li-Weber, M. & Krammer, P.H. Up-regulation of c-FLIPshort and reduction of activation-induced cell death in CD28-costimulated human T cells. Eur. J. Immunol. 30, 2765–2774 (2000).
Zheng, Y., Vig, M., Lyons, J., Van Parijs, L. & Beg, A.A. Combined deficiency of p50 and cRel in CD4+ T cells reveals an essential requirement for nuclear factor κB in regulating mature T cell survival and in vivo function. J. Exp. Med. 197, 861–874 (2003).
Dixon, D.A. et al. Regulation of cyclooxygenase-2 expression by the translational silencer TIA-1. J. Exp. Med. 198, 475–481 (2003).
Parfenova, H. et al. Dynamics of nuclear localization sites for COX-2 in vascular endothelial cells. Am. J. Physiol. Cell Physiol. 281, C166–C178 (2001).
Ballif, B.A., Mincek, N.V., Barratt, J.T., Wilson, M.L. & Simmons, D.L. Interaction of cyclooxygenases with an apoptosis- and autoimmunity-associated protein. Proc. Natl. Acad. Sci. USA 93, 5544–5549 (1996).
Lu, L., Kaliyaperumal, A., Boumpas, D.T. & Datta, S.K. Major peptide autoepitopes for nucleosome-specific T cells of human lupus. J. Clin. Invest. 104, 345–355 (1999).
Kaliyaperumal, A., Michaels, M.A. & Datta, S.K. Naturally processed chromatin peptides reveal a major autoepitope that primes pathogenic T and B cells of lupus. J. Immunol. 168, 2530–2537 (2002).
Lander, S.A., Wallace, D.J. & Weisman, M.H. Celecoxib for systemic lupus erythematosus: case series and literature review of the use of NSAIDs in SLE. Lupus 11, 340–347 (2002).
Acknowledgements
This work was supported by grants from the National Institutes of Health (R37-AR39157 and RO1-AI41985). We thank C.–S. Chen for giving us purified and recrystallized celecoxib and rofecoxib, and A. Traynor for providing the samples from five lupus patients included in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Xu, L., Zhang, L., Yi, Y. et al. Human lupus T cells resist inactivation and escape death by upregulating COX-2. Nat Med 10, 411–415 (2004). https://doi.org/10.1038/nm1005
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1005
This article is cited by
-
Metabolites of prostaglandin synthases as potential biomarkers of Lyme disease severity and symptom resolution
Inflammation Research (2019)
-
Constitutive expression of murine c-FLIPR causes autoimmunity in aged mice
Cell Death & Disease (2014)
-
DNA-Microarrays
Zeitschrift für Rheumatologie (2011)
-
Lymphopenia relating to T-lymphocyte apoptosis in systemic lupus erythematosus
Clinical Rheumatology (2011)
-
Distinct gene signature revealed in white blood cells, CD4+ and CD8+ T cells in (NZBx NZW) F1 lupus mice after tolerization with anti-DNA Ig peptide
Genes & Immunity (2010)