Abstract
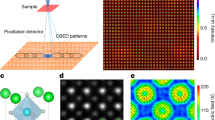
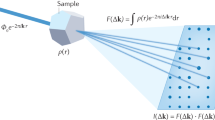
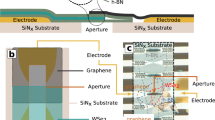
The electronic charge density distribution or the electrostatic atomic potential of a solid or molecule contains information not only on the atomic structure, but also on the electronic properties, such as the nature of the chemical bonds or the degree of ionization of atoms. However, the redistribution of charge due to chemical bonding is small compared with the total charge density, and therefore difficult to measure. Here, we demonstrate an experimental analysis of charge redistribution due to chemical bonding by means of high-resolution transmission electron microscopy (HRTEM). We analyse charge transfer on the single-atom level for nitrogen-substitution point defects in graphene, and confirm the ionicity of single-layer hexagonal boron nitride. Our combination of HRTEM experiments and first-principles electronic structure calculations opens a new way to investigate electronic configurations of point defects, other non-periodic arrangements or nanoscale objects that cannot be studied by an electron or X-ray diffraction analysis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hohenberg, P. & Kohn, W. Inhomogeneous electron gas. Phys. Rev. 136, B864–B871 (1964).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Coppens, P. & Stevens, E. D. Accurate X-ray diffraction and quantum chemistry: The study of charge density distributions. Adv. Quant. Chem. 10, 1–35 (1977).
Koritsanszky, T. S. & Coppens, P. Chemical applications of X-ray charge-density analysis. Chem. Rev. 101, 1583–1627 (2001).
Vainshtein, B. K. Modern Crystallography (Springer, 1964).
Zuo, J. M. Measurements of electron densities in solids: A real-space view of electronic structure and bonding in inorganic crystals. Rep. Prog. Phys. 67, 2053–2103 (2004).
Wu, L., Zhu, Y. & Tafto, J. Test of first-principle calculations of charge transfer and electron–hole distribution in oxide superconductors by precise measurements of structure factors. Phys. Rev. B 59, 6035–6038 (1999).
Shibata, S., Hirota, F. & Shioda, T. Molecular electron density from electron scattering. J. Mol. Struct. 485, 1–11 (1999).
Zuo, J. M., Kim, M., O’Keefe, M. & Spence, J. C. H. Direct observation of d-orbital holes and Cu–Cu bonding in Cu2O. Nature 401, 49–52 (1999).
Wu, L. et al. Valence-electron distribution in MgB2 by accurate diffraction measurements and first-principles calculations. Phys. Rev. B 69, 064501 (2004).
Hamers, R. J., Tromp, R. M. & Demuth, J. E. Surface electronic structure of Si-111 7×7 resolved in real space. Phys. Rev. Lett. 56, 1972–1975 (1986).
Browning, N. D., Chisholm, M. F. & Pennycook, S. J. Atomic-resolution chemical analysis using a scanning transmission electron microscope. Nature 366, 143–146 (1993).
Muller, D. A., Tzou, Y., Raj, R. & Silcox, J. Mapping sp2 and sp3 states of carbon at sub-nanometre spatial resolution. Nature 366, 725–727 (1993).
Batson, P. E. Simultaneous stem imaging and electron energy-loss spectroscopy with atomic-column sensitivity. Nature 366, 727–728 (1993.).
Muller, D. A. et al. Atomic-scale chemical imaging of composition and bonding by aberration-corrected microscopy. Science 319, 1073–1076 (2008).
Gross, L., Mohn, F., Moll, M., Liljeroth, P. & Meyer, G. The chemical structure of a molecule resolved by atomic force microscopy. Science 325, 1110–1114 (2009).
Weiss, C., Wagner, C., Temirov, R. & Tautz, F. S. Direct imaging of intermolecular bonds in scanning tunneling microscopy. J. Am. Chem. Soc. 132, 11864–11865 (2010).
Wolf, E. L. Principles of Electron Tunneling Spectroscopy (Oxford Univ. Press, 1989).
Cox, G. et al. Scanning tunneling microscopy of crystal dislocations in gallium arsenide. Phys. Rev. Lett. 64, 2402–2405 (1990).
Atamny, F., Spillecke, O. & Schlögl, R. On the STM imaging contrast of graphite: Towards a true atomic resolution. Phys. Chem. Chem. Phys. 1, 4113–4118 (1999).
Gemming, T., Mobius, G., Exner, M., Ernst, F. & Ruehle, M. Ab initio HRTEM simulations of ionic crystals: A case study of sapphire. J. Microsc. 190, 89–98 (1998).
Mogck, S., Kooi, B. J., De Hosson, J. Th. M. & Finnis, M. W. Ab initio transmission electron microscopy image simulations of coherent Ag–MgO interfaces. Phys. Rev. B 70, 245427 (2004).
Deng, B. & Marks, L. D. Theoretical structure factors for selected oxides and their effects in high-resolution electron-microscope (HREM) images. Acta Crystallogr. A 62, 208–216 (2006).
Deng, B., Marks, L. D. & Rondinelli, J. M. Charge defects glowing in the dark. Ultramicroscopy 107, 374–381 (2007).
Wei, D. et al. Synthesis of n-doped graphene by chemical vapor deposition and its electrical properties. Nano Lett. 9, 1752–1758 (2009).
Novoselov, K. S. et al. Electric field effect in atomically thin carbon films. Science 306, 666–669 (2004).
Geim, A. K. & Novoselov, K. S. The rise of graphene. Nature Mater. 6, 183–191 (2007).
Pauling, L. The structure and properties of graphite and boron nitride. Proc. Natl Acad. Sci. USA 56, 1646–1652 (1966).
Meyer, J. C., Chuvilin, A., Algara-Siller, G., Biskupek, J. & Kaiser, U. Selective sputtering and atomic resolution imaging of atomically thin boron nitride membranes. Nano Lett. 9, 2683–2689 (2009).
Jin, C., Lin, F., Suenaga, K. & Iijima, S. Fabrication of a freestanding boron nitride single layer and its defect assignments. Phys. Rev. Lett. 102, 195505 (2009).
Alem, N. et al. Atomically thin hexagonal boron nitride probed by ultrahigh-resolution transmission electron microscopy. Phys. Rev. B 80, 155425 (2009).
Krivanek, O. L. et al. Atom-by-atom structural and chemical analysis by annular dark-field electron microscopy. Nature 464, 571–574 (2010).
Kirkland, E. J. Advanced Computing in Electron Microscopy (Plenum, 1998).
Doyle, P. A. & Turner, P. S. Relativistic Hartree–Fock X-ray and electron scattering factors. Acta Crystallogr. A 24, 390–397 (1968).
Zhu, Z. H., Hatori, H., Wang, S. B. & Lu, G. Q. Insights into hydrogen atom adsorption on and the electrochemical properties of nitrogen-substituted carbon materials. J. Phys. Chem. B 109, 16744–16749 (2005).
Lim, S. H., Li, R., Ji, W. & Lin, J. Effects of nitrogenation on single-walled carbon nanotubes within density functional theory. Phys. Rev. B 76, 195406 (2007).
Meyer, J. C. et al. The structure of suspended graphene sheets. Nature 446, 60–63 (2007).
Meyer, J. C. et al. On the roughness of single- and bi-layer graphene membranes. Solid State Commun. 143, 101–109 (2007).
Thust, A. High-resolution transmission electron microscopy on an absolute contrast scale. Phys. Rev. Lett. 102, 220810 (2009).
Meyer, J. C., Girit, C. O., Crommie, M. & Zettl, A. Imaging and dynamics of light atoms and molecules on graphene. Nature 454, 319–322 (2008).
Kim, K. S. et al. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 457, 706–710 (2009).
Li, X. et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 324, 1312–1314 (2009).
Park, H. J., Meyer, J. C., Roth, S. & Skakalova, V. Growth and properties of few-layer graphene prepared by chemical vapor deposition. Carbon 48, 1088–1094 (2010).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Künzel, D., Markert, T., Groß, A. & Benoit, D. M. Bis(terpyridine)-based surface template structures on graphite: A force field and DFT study. Phys. Chem. Chem. Phys. 11, 8867–8878 (2009).
Blaha, P., Schwarz, K., Madsen, G. K. H., Kvasnicka, D. & Luitz, J. WIEN2k: An Augmented Plane Wave and Local Orbitals Program for Calculating Crystal Properties. (Vienna Univ. Technology, 2001).
Acknowledgements
We gratefully acknowledge financial support by the German Research Foundation (DFG) and the Ministry of Science, Research and the Arts (MWK) of the state Baden-Württemberg within the Sub-Angstrom Low-Voltage Electron Microscopy project (SALVE) and by the DFG within research project SFB 569. T.I. acknowledges the JSPS Postdoctoral Fellowship for Research Abroad. G.A-S. acknowledges the support of CONACyT-DAAD scholarship.
Author information
Authors and Affiliations
Contributions
J.C.M., A.C. and S.K. carried out TEM experiments. J.C.M., S.K. and A.C. analysed the data. S.K. carried out DFT calculations and TEM simulations based on WIEN2k. A.C. contributed to TEM simulations, discussions and analysis. H.J.P., V.S., S.R. and J.H.S. developed the synthesis of nitrogen-doped graphene. D.K. and A.G. carried out DFT calculations using the Vienna Ab initio Simulation Package. G.A-S. contributed to TEM simulations. T.I. and U.S. made Auger spectroscopy measurements. U.K. supervised part of the work. J.C.M. conceived and designed the study and wrote the paper. S.K. and U.K. co-wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 3488 kb)
Rights and permissions
About this article
Cite this article
Meyer, J., Kurasch, S., Park, H. et al. Experimental analysis of charge redistribution due to chemical bonding by high-resolution transmission electron microscopy. Nature Mater 10, 209–215 (2011). https://doi.org/10.1038/nmat2941
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat2941
This article is cited by
-
Imaging the electron charge density in monolayer MoS2 at the Ångstrom scale
Nature Communications (2023)
-
The helium-vacancy complexes and helium bubbles formation mechanism in chromium: a comprehensive first-principle study
Journal of Materials Science (2023)
-
Electronic structures and strengthening mechanisms of superhard high-entropy diborides
Rare Metals (2023)
-
Atomic electrostatic maps of 1D channels in 2D semiconductors using 4D scanning transmission electron microscopy
Nature Communications (2019)
-
Single-atom nickel confined nanotube superstructure as support for catalytic wet air oxidation of acetic acid
Communications Chemistry (2019)