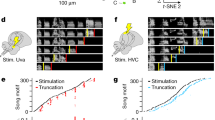
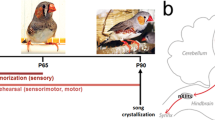
Abstract
Premotor circuits help generate imitative behaviors and can be activated during observation of another animal′s behavior, leading to speculation that these circuits participate in sensory learning that is important to imitation. Here we tested this idea by focally manipulating the brain activity of juvenile zebra finches, which learn to sing by memorizing and vocally copying the song of an adult tutor. Tutor song–contingent optogenetic or electrical disruption of neural activity in the pupil′s song premotor nucleus HVC prevented song copying, indicating that a premotor structure important to the temporal control of birdsong also helps encode the tutor song. In vivo multiphoton imaging and neural manipulations delineated a pathway and a candidate synaptic mechanism through which tutor song information is encoded by premotor circuits. These findings provide evidence that premotor circuits help encode sensory information about the behavioral model before shaping and executing imitative behaviors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Dehaene-Lambertz, G. et al. Functional organization of perisylvian activation during presentation of sentences in preverbal infants. Proc. Natl. Acad. Sci. USA 103, 14240–14245 (2006).
Ferrari, P.F., Gallese, V., Rizzolatti, G. & Fogassi, L. Mirror neurons responding to the observation of ingestive and communicative mouth actions in the monkey ventral premotor cortex. Eur. J. Neurosci. 17, 1703–1714 (2003).
Gallese, V., Fadiga, L., Fogassi, L. & Rizzolatti, G. Action recognition in the premotor cortex. Brain 119, 593–609 (1996).
Prather, J.F., Peters, S., Nowicki, S. & Mooney, R. Precise auditory-vocal mirroring in neurons for learned vocal communication. Nature 451, 305–310 (2008).
Prather, J.F., Peters, S., Nowicki, S. & Mooney, R. Persistent representation of juvenile experience in the adult songbird brain. J. Neurosci. 30, 10586–10598 (2010).
Arbib, M.A., Liebal, K. & Pika, S. Primate vocalization, gesture, and the evolution of human language. Curr. Anthropol. 49, 1053–1063 discussion 1063–1076 (2008).
Iacoboni, M. et al. Cortical mechanisms of human imitation. Science 286, 2526–2528 (1999).
Rizzolatti, G. & Arbib, M.A. Language within our grasp. Trends Neurosci. 21, 188–194 (1998).
Rizzolatti, G., Fogassi, L. & Gallese, V. Neurophysiological mechanisms underlying the understanding and imitation of action. Nat. Rev. Neurosci. 2, 661–670 (2001).
Doupe, A.J. & Kuhl, P.K. Birdsong and human speech: common themes and mechanisms. Annu. Rev. Neurosci. 22, 567–631 (1999).
Immelman, K. Song development in the zebra finch and other estrildid finches. in Bird Vocalizations (ed. Hinde, R.A.), 61–74 (Cambridge University Press, 1969).
Marler, P. & Tamura, M. Culturally transmitted patterns of vocal behavior in sparrows. Science 146, 1483–1486 (1964).
Mooney, R. Neural mechanisms for learned birdsong. Learn. Mem. 16, 655–669 (2009).
Hahnloser, R.H. & Kotowicz, A. Auditory representations and memory in birdsong learning. Curr. Opin. Neurobiol. 20, 332–339 (2010).
London, S.E. & Clayton, D.F. Functional identification of sensory mechanisms required for developmental song learning. Nat. Neurosci. 11, 579–586 (2008).
Bolhuis, J.J., Hetebrij, E., Den Boer-Visser, A.M., De Groot, J.H. & Zijlstra, G.G. Localized immediate early gene expression related to the strength of song learning in socially reared zebra finches. Eur. J. Neurosci. 13, 2165–2170 (2001).
Gobes, S.M. & Bolhuis, J.J. Birdsong memory: a neural dissociation between song recognition and production. Curr. Biol. 17, 789–793 (2007).
Phan, M.L., Pytte, C.L. & Vicario, D.S. Early auditory experience generates long-lasting memories that may subserve vocal learning in songbirds. Proc. Natl. Acad. Sci. USA 103, 1088–1093 (2006).
Terpstra, N.J. & Bolhuis, J.J. & den Boer-Visser, A.M. An analysis of the neural representation of birdsong memory. J. Neurosci. 24, 4971–4977 (2004).
Akutagawa, E. & Konishi, M. New brain pathways found in the vocal control system of a songbird. J. Comp. Neurol. 518, 3086–3100 (2010).
Bauer, E.E. et al. A synaptic basis for auditory-vocal integration in the songbird. J. Neurosci. 28, 1509–1522 (2008).
Nottebohm, F., Stokes, T.M. & Leonard, C.M. Central control of song in the canary, Serinus canarius. J. Comp. Neurol. 165, 457–486 (1976).
Hahnloser, R.H., Kozhevnikov, A.A. & Fee, M.S. An ultra-sparse code underlies the generation of neural sequences in a songbird. Nature 419, 65–70 (2002).
Long, M.A. & Fee, M.S. Using temperature to analyse temporal dynamics in the songbird motor pathway. Nature 456, 189–194 (2008).
Nick, T.A. & Konishi, M. Neural song preference during vocal learning in the zebra finch depends on age and state. J. Neurobiol. 62, 231–242 (2005).
Roberts, T.F., Tschida, K.A., Klein, M.E. & Mooney, R. Rapid spine stabilization and synaptic enhancement at the onset of behavioural learning. Nature 463, 948–952 (2010).
Basham, M.E., Nordeen, E.J. & Nordeen, K.W. Blockade of NMDA receptors in the anterior forebrain impairs sensory acquisition in the zebra finch (Poephila guttata). Neurobiol. Learn. Mem. 66, 295–304 (1996).
Boyden, E.S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Nagel, G. et al. Light activation of channelrhodopsin-2 in excitable cells of Caenorhabditis elegans triggers rapid behavioral responses. Curr. Biol. 15, 2279–2284 (2005).
Yizhar, O., Fenno, L.E., Davidson, T.J., Mogri, M. & Deisseroth, K. Optogenetics in neural systems. Neuron 71, 9–34 (2011).
Zhang, F. et al. Multimodal fast optical interrogation of neural circuitry. Nature 446, 633–639 (2007).
Kelley, D.B. & Nottebohm, F. Projections of a telencephalic auditory nucleus-field L-in the canary. J. Comp. Neurol. 183, 455–469 (1979).
Vates, G.E., Broome, B.M., Mello, C.V. & Nottebohm, F. Auditory pathways of caudal telencephalon and their relation to the song system of adult male zebra finches. J. Comp. Neurol. 366, 613–642 (1996).
Wild, J.M., Williams, M.N., Howie, G.J. & Mooney, R. Calcium-binding proteins define interneurons in HVC of the zebra finch (Taeniopygia guttata). J. Comp. Neurol. 483, 76–90 (2005).
Mooney, R. & Prather, J.F. The HVC microcircuit: the synaptic basis for interactions between song motor and vocal plasticity pathways. J. Neurosci. 25, 1952–1964 (2005).
Tumer, E.C. & Brainard, M.S. Performance variability enables adaptive plasticity of 'crystallized' adult birdsong. Nature 450, 1240–1244 (2007).
Vu, E.T., Mazurek, M.E. & Kuo, Y.C. Identification of a forebrain motor programming network for the learned song of zebra finches. J. Neurosci. 14, 6924–6934 (1994).
Yu, A.C. & Margoliash, D. Temporal hierarchical control of singing in birds. Science 273, 1871–1875 (1996).
Engert, F. & Bonhoeffer, T. Dendritic spine changes associated with hippocampal long-term synaptic plasticity. Nature 399, 66–70 (1999).
Matsuzaki, M., Honkura, N., Ellis-Davies, G.C. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004).
Fortune, E.S. & Margoliash, D. Parallel pathways and convergence onto HVc and adjacent neostriatum of adult zebra finches (Taeniopygia guttata). J. Comp. Neurol. 360, 413–441 (1995).
Cardin, J.A., Raksin, J.N. & Schmidt, M.F. Sensorimotor nucleus NIf is necessary for auditory processing but not vocal motor output in the avian song system. J. Neurophysiol. 93, 2157–2166 (2005).
Coleman, M.J. & Mooney, R. Synaptic transformations underlying highly selective auditory representations of learned birdsong. J. Neurosci. 24, 7251–7265 (2004).
Hahnloser, R.H. & Fee, M.S. Sleep-related spike bursts in HVC are driven by the nucleus interface of the nidopallium. J. Neurophysiol. 97, 423–435 (2007).
Roy, A. & Mooney, R. Song decrystallization in adult zebra finches does not require the song nucleus NIf. J. Neurophysiol. 102, 979–991 (2009).
Fortune, E.S. & Margoliash, D. Cytoarchitectonic organization and morphology of cells of the field L complex in male zebra finches (Taenopygia guttata). J. Comp. Neurol. 325, 388–404 (1992).
Bolhuis, J.J. & Gahr, M. Neural mechanisms of birdsong memory. Nat. Rev. Neurosci. 7, 347–357 (2006).
Arbib, M.A. From grasp to language: embodied concepts and the challenge of abstraction. J. Physiol. Paris 102, 4–20 (2008).
Fabbri-Destro, M. & Rizzolatti, G. Mirror neurons and mirror systems in monkeys and humans. Physiology (Bethesda) 23, 171–179 (2008).
Roberts, T.F., Klein, M.E., Kubke, M.F., Wild, J.M. & Mooney, R. Telencephalic neurons monosynaptically link brainstem and forebrain premotor networks necessary for song. J. Neurosci. 28, 3479–3489 (2008).
Ölveczky, B.P., Andalman, A.S. & Fee, M.S. Vocal experimentation in the juvenile songbird requires a basal ganglia circuit. PLoS Biol. 3, e153 (2005).
Dutar, P., Vu, H.M. & Perkel, D.J. Multiple cell types distinguished by physiological, pharmacological and anatomic properties in nucleus HVC of the adult zebra finch. J. Neurophysiol. 80, 1828–1838 (1998).
Cardin, J.A., Raksin, J.N. & Schmidt, M.F. Sensorimotor nucleus NIf is necessary for auditory processing but not vocal motor output in the avian song system. J. Neurophysiol. 93, 2157–2166 (2005).
Acknowledgements
The authors thank H. Greenside, S. Lisberger, D. Purves, K. Tschida and F. Wang for reading and commenting on the manuscript, K. Hamaguchi (Duke University Medical Center) for data analysis software support and design of microdialysis probes, T. Otchy, K. Bohon, A. Bae and S. Lotfi for technical assistance and assistance with data processing and J. Baltzegar for animal husbandry and laboratory support. This research was supported by grants from the National Science Foundation (R.M.), the US National Institutes of Health (R.M. (R01 DC02524) and B.P.Ö. (R01 NS066408)), a Rubicon fellowship from the Netherlands Organization for Scientific Research (NWO) to S.M.H.G. and grants from the Klingenstein, Sloan and McKnight Foundations (B.P.Ö.).
Author information
Authors and Affiliations
Contributions
This manuscript combines the synergistic results of two independently conceived and executed research studies arriving at similar conclusions. T.F.R. and R.M. designed all experiments involving tutor song-contingent optogenetic or electrical manipulations of HVC activity, in vivo multiphoton imaging of HVC dendritic spines, pharmacological manipulations of NMDA receptors in HVC during tutoring and tutor song-contingent microstimulation of NIf and Field L. T.F.R. conducted all of the aforementioned experiments and analyzed resultant data with input from R.M., S.M.H.G. and B.P.Ö. designed, executed and analyzed the results of experiments involving permanent or reversible NIf lesions. M.M. adapted optogenetic methods for use in the zebra finch and characterized the effects of hChR2 activation on HVC neurons in brain slices. T.F.R. and R.M. wrote the manuscript with extensive input from B.P.Ö. and S.M.H.G. All authors read and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Table 1 (PDF 2434 kb)
Supplementary Video 1
Tutor song-triggered optogenetic disruption of neural activity in the pupils' HVC. Video of a juvenile zebra finch being tutored for the first time, using tutor song-contingent optogenetic methods to disrupt neural activity in HVC. The song of the tutor (bird on the right in this video) is detected by software and used to drive brief pulses of blue light delivered to the HVC of the pupil (bird on left in this video) via chronically implanted fiber optic cables (MOV 5127 kb)
Rights and permissions
About this article
Cite this article
Roberts, T., Gobes, S., Murugan, M. et al. Motor circuits are required to encode a sensory model for imitative learning. Nat Neurosci 15, 1454–1459 (2012). https://doi.org/10.1038/nn.3206
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3206
This article is cited by
-
Characterizing Sensory Phenotypes of Subgroups with a Known Genetic Etiology Pertaining to Diagnoses of Autism Spectrum Disorder and Intellectual Disability
Journal of Autism and Developmental Disorders (2023)
-
Wireless and battery-free technologies for neuroengineering
Nature Biomedical Engineering (2021)
-
Memory-specific correlated neuronal activity in higher-order auditory regions of a parrot
Scientific Reports (2021)
-
Expression of FoxP2 in the basal ganglia regulates vocal motor sequences in the adult songbird
Nature Communications (2021)
-
AAV1 is the optimal viral vector for optogenetic experiments in pigeons (Columba livia)
Communications Biology (2021)