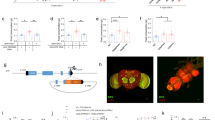
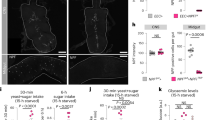
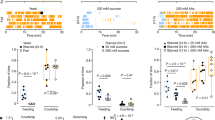
Abstract
Omnivores, including humans, have an inborn tendency to avoid noxious or unfamiliar foods. Such defensive foraging behaviors are modifiable, however, in response to physiological needs. Here we describe a method for assessing risk-sensitive food acquisition in Drosophila melanogaster. Food-deprived fly larvae become more likely to feed on noxious foods (adulterated with quinine) as the duration of deprivation increases. The neuropeptide F receptor NPFR1, a mammalian neuropeptide Y (NPY) receptor homolog, centrally regulates the response to noxious food in D. melanogaster. Overexpression of NPFR1 was sufficient to cause nondeprived larvae to more readily take in noxious food, whereas loss of NPFR1 signaling led to the opposite phenotype. Moreover, NPFR1 neuronal activity may be directly regulated by the insulin-like signaling pathway. Upregulation of insulin-like receptor signaling in NPFR1 cells suppressed the feeding response to noxious food. Our results suggest that the coordinated activities of the conserved NPY- and insulin-like receptor signaling systems are essential for the dynamic regulation of noxious food intake according to the animal's energy state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Charnov, E.L. Optimal foraging, the marginal value theorem. Theor. Popul. Biol. 9, 129–136 (1976).
Stricker, E.M. Neurobiology of Food and Fluid Intake (Plenum, New York, 1990).
Bateson, M. Recent advances in our understanding of risk-sensitive foraging preferences. Proc. Nutr. Soc. 61, 509–516 (2002).
Hewes, R.S. & Taghert, P.H. Neuropeptides and neuropeptide receptors in the Drosophila melanogaster genome. Genome Res. 11, 1126–1142 (2001).
Brown, M.R. et al. Identification of a Drosophila brain-gut peptide related to the neuropeptide Y family. Peptides 20, 1035–1042 (1999).
Wu, Q. et al. Developmental control of foraging and social behavior by the Drosophila neuropeptide Y-like system. Neuron 39, 147–161 (2003).
Bannon, A.W. et al. Behavioral characterization of neuropeptide Y knockout mice. Brain Res. 868, 79–87 (2000).
Segal-Lieberman, G., Trombly, D.J., Juthani, V., Wang, X. & Maratos-Flier, E. NPY ablation in C57BL/6 mice leads to mild obesity and to an impaired refeeding response to fasting. Am. J. Physiol. Endocrinol. Metab. 284, E1131–E1139 (2003).
Brogiolo, W. et al. An evolutionarily conserved function of the Drosophila insulin receptor and insulin-like peptides in growth control. Curr. Biol. 11, 213–221 (2001).
Rulifson, E.J., Kim, S.K. & Nusse, R. Ablation of insulin-producing neurons in flies: growth and diabetic phenotypes. Science 296, 1118–1120 (2002).
Ikeya, T., Galic, M., Belawat, P., Nairz, K. & Hafen, E. Nutrient-dependent expression of insulin-like peptides from neuroendocrine cells in the CNS contributes to growth regulation in Drosophila. Curr. Biol. 12, 1293–1300 (2002).
Oldham, S. & Hafen, E. Insulin/IGF and target of rapamycin signaling: a TOR de force in growth control. Trends Cell Biol. 13, 79–85 (2003).
Bruning, J.C. et al. Role of brain insulin receptor in control of body weight and reproduction. Science 289, 2122–2125 (2000).
Gerozissis, K. Brain insulin: regulation, mechanisms of action and functions. Cell. Mol. Neurobiol. 23, 1–25 (2003).
Moore, P. Controlling how many cells make a fly. J. Biol. 2, 16–16.5 (2003).
Song, J., Wu, L., Chen, Z., Kohanski, R.A. & Pick, L. Axons guided by insulin receptor in Drosophila visual system. Science 300, 502–505 (2003).
Osborne, K.A. et al. Natural behavior polymorphism due to a cGMP-dependent protein kinase of Drosophila. Science 277, 834–836 (1997).
Shen, P. & Cai, H.N. Drosophila neuropeptide F mediates integration of chemosensory stimulation and conditioning of the nervous system by food. J. Neurobiol. 47, 16–25 (2001).
Kitamoto, T. Targeted expression of temperature-sensitive dynamin to study neural mechanisms of complex behavior in Drosophila. J. Neurogenet. 16, 205–228 (2002).
Cao, H., Thompson, H.M., Krueger, E.W. & McNiven, M.A. Disruption of Golgi structure and function in mammalian cells expressing a mutant dynamin. J. Cell Sci. 113, 1993–2002 (2000).
Jones, S.M., Howell, K.E., Henley, J.R., Cao, H. & McNiven, M.A. Role of dynamin in the formation of transport vesicles from the trans-Golgi network. Science 279, 573–577 (1998).
Kreitzer, G., Marmorstein, A., Okamoto, P., Vallee, R. & Rodriguez-Boulan, E. Kinesin and dynamin are required for post-Golgi transport of a plasma-membrane protein. Nat. Cell Biol. 2, 125–127 (2000).
Haass, M., Hock, M., Richardt, G. & Schomig, A. Neuropeptide Y differentiates between exocytotic and nonexocytotic noradrenaline release in guinea-pig heart. Naunyn Schmiedebergs Arch. Pharmacol. 340, 509–515 (1989).
Wen, T., Parrish, C.A., Xu, D., Wu, Q. & Shen, P. Drosophila neuropeptide F and its receptor NPFR1 define a signaling pathway that acutely modulates alcohol sensitivity. Proc. Natl. Acad. Sci. USA 102, 2141–2146 (2005).
Colombani, J. et al. A nutrient sensor mechanism controls Drosophila growth. Cell 114, 739–749 (2003).
Kimball, S.R. & Jefferson, L.S. Regulation of protein synthesis by branched-chain amino acids. Curr. Opin. Clin. Nutr. Metab. Care 4, 39–43 (2001).
Thomas, G. The S6 kinase signaling pathway in the control of development and growth. Biol. Res. 35, 305–313 (2002).
Barcelo, H. & Stewart, M.J. Altering Drosophila S6 kinase activity is consistent with a role for S6 kinase in growth. Genesis 34, 83–85 (2002).
Thorsell, A. & Heilig, M. Diverse functions of neuropeptide Y revealed using genetically modified animals. Neuropeptides 36, 182–193 (2002).
Thorsell, A. et al. Behavioral insensitivity to restraint stress, absent fear suppression of behavior and impaired spatial learning in transgenic rats with hippocampal neuropeptide Y overexpression. Proc. Natl. Acad. Sci. USA 97, 12852–12857 (2000).
Levine, A.S., Kotz, C.M. & Gosnell, B.A. Sugars and fats: the neurobiology of preference. J. Nutr. 133, 831S–834S (2003).
Levine, A.S., Jewett, D.C., Cleary, J.P., Kotz, C.M. & Billington, C.J. Our journey with neuropeptide Y: effects on ingestive behaviors and energy expenditure. Peptides 25, 505–510 (2004).
Larhammar, D. Evolution of neuropeptide Y, peptide YY and pancreatic polypeptide. Regul. Pept. 62, 1–11 (1996).
Davis, G.W., DiAntonio, A., Petersen, S.A. & Goodman, C.S. Postsynaptic PKA controls quantal size and reveals a retrograde signal that regulates presynaptic transmitter release in Drosophila. Neuron 20, 305–315 (1998).
Gao, X., Neufeld, T.P. & Pan, D. Drosophila PTEN regulates cell growth and proliferation through PI3K-dependent and -independent pathways. Dev. Biol. 221, 404–418 (2000).
Leevers, S.J., Weinkove, D., MacDougall, L.K., Hafen, E. & Waterfield, M.D. The Drosophila phosphoinositide 3-kinase Dp110 promotes cell growth. EMBO J. 15, 6584–6594 (1996).
Lin, D.M. & Goodman, C.S. Ectopic and increased expression of Fasciclin II alters motoneuron growth cone guidance. Neuron 13, 507–523 (1994).
Torroja, L., Chu, H., Kotovsky, I. & White, K. Neuronal overexpression of APPL, the Drosophila homologue of the amyloid precursor protein (APP), disrupts axonal transport. Curr. Biol. 9, 489–492 (1999).
Rao, S., Lang, C., Levitan, E.S. & Deitcher, D.L. Visualization of neuropeptide expression, transport, and exocytosis in Drosophila melanogaster. J. Neurobiol. 49, 159–172 (2001).
Piccin, A. et al. Efficient and heritable functional knock-out of an adult phenotype in Drosophila using a GAL4-driven hairpin RNA incorporating a heterologous spacer. Nucleic Acids Res. 29, E55 (2001).
Garczynski, S.F., Brown, M.R., Shen, P., Murray, T.F. & Crim, J.W. Characterization of a functional neuropeptide F receptor from Drosophila melanogaster. Peptides 23, 773–780 (2002).
Acknowledgements
We thank D.L. Deitcher, E. Hafen, T. Kitamoto, D. Pan, J.H. Park, Exelixis Inc. and the Bloomington Fly Stock Center for fly lines, and J.S. Willis for the critical reading of the manuscript. This work was funded by a National Institutes of Health grant DK-58348 to P.S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Representative images of feeding and nonfeeding larvae in quinine assays. (PDF 117 kb)
Supplementary Fig. 2
Dissection of the midgut of feeding and nonfeeding larvae. (PDF 587 kb)
Supplementary Fig. 3
Comparison of the size of adult eyes. (PDF 407 kb)
Supplementary Fig. 4
Model of the central regulation of risk-sensitive feeding response to noxious food. (PDF 94 kb)
Rights and permissions
About this article
Cite this article
Wu, Q., Zhao, Z. & Shen, P. Regulation of aversion to noxious food by Drosophila neuropeptide Y– and insulin-like systems. Nat Neurosci 8, 1350–1355 (2005). https://doi.org/10.1038/nn1540
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1540
This article is cited by
-
Heterogeneous receptor expression underlies non-uniform peptidergic modulation of olfaction in Drosophila
Nature Communications (2023)
-
The fly liquid-food electroshock assay (FLEA) suggests opposite roles for neuropeptide F in avoidance of bitterness and shock
BMC Biology (2021)
-
Availability of food determines the need for sleep in memory consolidation
Nature (2021)
-
Adipokinetic hormone (AKH), energy budget and their effect on feeding and gustatory processes of foraging honey bees
Scientific Reports (2021)
-
Impact of alkaloids in food consumption, metabolism and survival in a blood-sucking insect
Scientific Reports (2020)