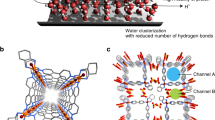
Abstract
Proton transport plays an important role in many biological processes due to the ability of protons to rapidly translocate along chains of hydrogen-bonded water molecules. Molecular dynamics simulations have predicted that confinement in hydrophobic nanochannels should enhance the rate of proton transport. Here, we show that 0.8-nm-diameter carbon nanotube porins, which promote the formation of one-dimensional water wires, can support proton transport rates exceeding those of bulk water by an order of magnitude. The transport rates in these narrow nanotube pores also exceed those of biological channels and Nafion. With larger 1.5-nm-diameter nanotube porins, proton transport rates comparable to bulk water are observed. We also show that the proton conductance of these channels can be modulated by the presence of Ca2+ ions. Our results illustrate the potential of small-diameter carbon nanotube porins as a proton conductor material and suggest that strong spatial confinement is a key factor in enabling efficient proton transport.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Grotthuss, C. J. T. Sur la décomposition de l'eau et des corps q'uelle tient en dissolution à l'aide de l’électricité galvanique. Ann. Chim. LVIII, 54–74 (1806).
Wraight, C. A. Chance and design—Proton transfer in water, channels and bioenergetic proteins. Biochim. Biophys. Acta 1757, 886–912 (2006).
Kashket, E. R. The proton motive force in bacteria – a critical-assessment of methods. Ann. Rev. Microbiol. 39, 219–242 (1985).
DeCoursey, T. E. & Grinstein, S. in Inflammation: Basic Principles and Clinical Correlates (eds Gallin, J. I. & Snyderman, R.) 639–659 (Lippincott Williams & Wilkins, 1999).
Demaurex, N., Downey, G. P., Waddell, T. K. & Grinstein, S. Intracellular pH regulation during spreading of human neutrophils. J. Cell Biol. 133, 1391–1402 (1996).
DeCoursey, T. E. Hypothesis: do voltage-gated H+ channels in alveolar epithelial cells contribute to CO2 elimination by the lung? Am. J. Physiol. 278, C1–C10 (2000).
Chernyshev, A., Armstrong, K. M. & Cukierman, S. Proton transfer in gramicidin channels is modulated by the thickness of monoglyceride bilayers. Biophys. J. 84, 238–250 (2003).
Moffat, J. C. et al. Proton transport through influenza a virus M2 protein reconstituted in vesicles. Biophys. J. 94, 434–445 (2008).
Zhou, J. et al. Function of protonatable residues in the flagellar motor of Escherichia coli: a critical role for Asp 32 of MotB. J. Bacteriol. 180, 2729–2735 (1998).
Aksimentiev, A., Balabin, I. A., Fillingame, R. H. & Schulten, K. Insights into the molecular mechanism of rotation in the Fo sector of ATP synthase. Biophys. J. 86, 1332–1344 (2004).
Peighambardoust, S. J., Rowshanzamir, S. & Amjadi, M. Review of the proton exchange membranes for fuel cell applications. Int. J. Hydrogen. Energ. 35, 9349–9384 (2010).
Slade, S., Campbell, S. A., Ralph, T. R. & Walsh, F. C. Ionic conductivity of an extruded Nafion 1100 EW series of membranes. J. Electrochem. Soc. 149, A1556–A1564 (2002).
Maniwa, Y. et al. Water-filled single-wall carbon nanotubes as molecular nanovalves. Nature Mater. 6, 135–141 (2007).
Kolesnikov, A. I. et al. Anomalously soft dynamics of water in a nanotube: a revelation of nanoscale confinement. Phys. Rev. Lett. 93, 035503 (2004).
Lee, C. Y., Choi, W., Han, J.-H. & Strano, M. S. Coherence resonance in a single-walled carbon nanotube ion channel. Science 329, 1320–1324 (2010).
Kalra, A., Garde, S. & Hummer, G. Osmotic water transport through carbon nanotube membranes. Proc. Natl Acad. Sci. USA 100, 10175–10180 (2003).
Dellago, C., Naor, M. M. & Hummer, G. Proton transport through water-filled carbon nanotubes. Phys. Rev. Lett. 90, 105902 (2003).
Holt, J. K. et al. Fast mass transport through sub-2-nanometer carbon nanotubes. Science 312, 1034–1037 (2006).
Choi, W. et al. Diameter-dependent ion transport through the interior of isolated single-walled carbon nanotubes. Nature Commun. 4, 2397 (2013).
Geng, J. et al. Stochastic transport through carbon nanotubes in lipid bilayers and live cell membranes. Nature 514, 612–618 (2014).
Pascal, T. A., Goddard, W. A. & Jung, Y. Entropy and the driving force for the filling of carbon nanotubes with water. Proc. Natl Acad. Sci. USA 108, 11794–11798 (2011).
Weisman, R. B. & Bachilo, S. M. Dependence of optical transition energies on structure for single-walled carbon nanotubes in aqueous suspension: an empirical Kataura plot. Nano Lett. 3, 1235–1238 (2003).
Deamer, D. W. & Nichols, J. W. Proton-hydroxide permeability of liposomes. Proc. Natl Acad. Sci. USA 80, 165–168 (1983).
Han, J. Y. & Burgess, K. Fluorescent indicators for intracellular pH. Chem. Rev. 110, 2709–2728 (2010).
Gensure, R. H., Zeidel, M. L. & Hill, W. G. Lipid raft components cholesterol and sphingomyelin increase H+/OH- permeability of phosphatidylcholine membranes. Biochem. J. 398, 485–495 (2006).
Krishnamoorthy, G. Temperature jump as a new technique to study the kinetics of fast transport of protons across membranes. Biochemistry 25, 6666–6671 (1986).
Day, T. J. F., Soudackov, A. V., Cuma, M., Schmitt, U. W. & Voth, G. A. A second generation multistate empirical valence bond model for proton transport in aqueous systems. J. Chem. Phys. 117, 5839–5849 (2002).
Pomes, R. & Roux, B. Free energy profiles for H+ conduction along hydrogen-bonded chains of water molecules. Biophys. J. 75, 33–40 (1998).
Braun-Sand, S., Burykin, A., Chu, Z. T. & Warshel, A. Realistic simulations of proton transport along the gramicidin channel: Demonstrating the importance of solvation effects. J. Phys. Chem. B 109, 583–592 (2005).
Dellago, C. & Hummer, G. Kinetics and mechanism of proton transport across membrane nanopores. Phys. Rev. Lett. 97, 245901 (2006).
Akeson, M. & Deamer, D. W. Proton conductance by the gramicidin water wire. Model for proton conductance in the F1F0 ATPases? Biophys. J. 60, 101–109 (1991).
Chernyshev, A. & Cukierman, S. Thermodynamic view of activation energies of proton transfer in various gramicidin A channels. Biophys. J. 82, 182–192 (2002).
Golovanov, A. P. et al. The divalent cation-binding sites of gramicidin-a transmembrane ion-channel. Biopolym. 31, 425–434 (1991).
Mclanugh, S. G., Szabo, G., Eisenman, G. & Ciani, S. M. Surface charge and conductance of phospholipid membranes. Proc. Natl Acad. Sci. USA 67, 1268–1275 (1970).
Liu, Z. F. et al. Organizing single-walled carbon nanotubes on gold using a wet chemical self-assembling technique. Langmuir 16, 3569–3573 (2000).
Minati, L. et al. Characterization of thiol-functionalized carbon nanotubes on gold surfaces. Surf. Sci. 604, 1414–1419 (2010).
Stewart, J. C. Colorimetric determination of phospholipids with ammonium ferrothiocyanate. Analyt. Biochem. 104, 10–14 (1980).
Huang, C. & Mason, J. T. Geometric packing constraints in egg phosphatidylcholine vesicles. Proc. Natl Acad. Sci. USA 75, 308–310 (1978).
Acknowledgements
We thank A.T. Pham for the images used in Fig. 1b,c, and Y. Yu for assistance with cryo-EM imaging. A.B. acknowledges SULI summer internship programme funding from the US Department of Energy. This work was supported by the US Department of Energy, Office of Basic Energy Sciences, Division of Materials Sciences and Engineering under Award SCW0972. Work at the Lawrence Livermore National Laboratory was performed under the auspices of the US Department of Energy under Contract DE-AC52-07NA27344. Work at the Molecular Foundry was supported by the Office of Science, Office of Basic Energy Sciences, of the US Department of Energy under Contract No. DE-AC02-05CH11231.
Author information
Authors and Affiliations
Contributions
R.H.T and A.N. designed the experiments, R.H.T. performed the proton transport experiments. K.K. and R.H.T. performed Raman characterization studies. F.I.A. and R.H.T. performed the TEM and cryogenic TEM characterization. A.B., K.K. and R.H.T. synthesized the CNT porins. R.H.T. and A.N. wrote the manuscript, and all authors commented on it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1988 kb)
Rights and permissions
About this article
Cite this article
Tunuguntla, R., Allen, F., Kim, K. et al. Ultrafast proton transport in sub-1-nm diameter carbon nanotube porins. Nature Nanotech 11, 639–644 (2016). https://doi.org/10.1038/nnano.2016.43
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2016.43
This article is cited by
-
Transient water wires mediate selective proton transport in designed channel proteins
Nature Chemistry (2023)
-
Breakdown of the Nernst–Einstein relation in carbon nanotube porins
Nature Nanotechnology (2023)
-
Construction of angstrom-scale ion channels with versatile pore configurations and sizes by metal-organic frameworks
Nature Communications (2023)
-
Search for a Grotthuss mechanism through the observation of proton transfer
Communications Chemistry (2023)
-
High-resolution discrimination of homologous and isomeric proteinogenic amino acids in nanopore sensors with ultrashort single-walled carbon nanotubes
Nature Communications (2023)