Abstract
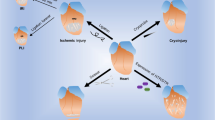
Despite advances in treatment of patients who suffer from ischemic heart disease, morbidity related to myocardial infarction is increasing in Western societies. Acute and chronic immune responses elicited by myocardial ischemia have an important role in the functional deterioration of the heart. Research on modulation of the inflammatory responses was focused on effector mediators such as leukocytes. However, increasing evidence indicates that various endogenous ligands that act as 'danger signals', also called danger-associated molecular patterns (DAMPs), are released upon injury and modulate inflammation. Originally described as part of the first-line defense against invading microorganisms, several Toll-like receptors (TLRs) on leukocytes and parenchymal cells have now been shown to respond to such signals and to have a pivotal role in noninfectious pathological cardiovascular conditions, such as ischemia–reperfusion injury and heart failure. From a therapeutic perspective, DAMPs are attractive targets owing to their specific induction after injury. In this Review, we will discuss innate immune activation through TLRs in cardiac ischemia mediated by DAMPs.
Key Points
-
Innate immune responses are critical mediators of tissue damage and repair after myocardial infarction
-
Cells involved in innate immunity also recognize and become activated by molecules released after cell death or by matrix degradation products, so-called 'danger signals' or danger-associated molecular patterns (DAMPs)
-
Toll-like receptors on cardiac and circulating cells recognize pathogen-associated molecular patterns as well as DAMPs and are important mediators of inflammatory reactions after cardiac ischemia
-
Preclinical studies show that DAMPs are promising targets to enhance myocardial viability and repair after myocardial infarction
-
As several DAMPs are necessary for proper wound healing, therapeutic interference with DAMP-related signaling in patients after myocardial infarction necessitates careful evaluation and further research
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Virchow, R. Cellular pathology. As based upon physiological and pathological histology. Lecture XVI—Atheromatous affection of arteries. 1858. Nutr. Rev. 47, 23–25 (1989).
Van, A. W. I. Landerer's mechanical theory of inflammation. Ann. Surg. 2, 479–485 (1885).
Lefer, A. M. & Rovetto, M. J. Influence of a myocardial depressant factor on physiologic properties of cardiac muscle. Proc. Soc. Exp. Biol. Med. 134, 269–273 (1970).
Lefer, A. M., Cowgill, R., Marshall, F. F., Hall, L. M. & Brand, E. D. Characterization of a myocardial depressant factor present in hemorrhagic shock. Am. J. Physiol. 213, 492–498 (1967).
Brand, E. D. & Lefer, A. M. Myocardial depressant factor in plasma from cats in irreversible post-oligemic shock. Proc. Soc. Exp. Biol. Med. 122, 200–203 (1966).
Arslan, F., de Kleijn, D. P., Timmers, L., Doevendans, P. A. & Pasterkamp, G. Bridging innate immunity and myocardial ischemia/reperfusion injury: the search for therapeutic targets. Curr. Pharm. Des. 14, 1205–1216 (2008).
Chao, W. Toll-like receptor signaling: a critical modulator of cell survival and ischemic injury in the heart. Am. J. Physiol. Heart Circ. Physiol. 296, H1–12 (2009).
Frangogiannis, N. G. Chemokines in the ischemic myocardium: from inflammation to fibrosis. Inflamm. Res. 53, 585–595 (2004).
Kelly, R. A. & Smith, T. W. Cytokines and cardiac contractile function. Circulation 95, 778–781 (1997).
Mann, D. L. Inflammatory mediators and the failing heart: past, present, and the foreseeable future. Circ. Res. 91, 988–998 (2002).
Frangogiannis, N. G. The immune system and cardiac repair. Pharmacol. Res. 58, 88–111 (2008).
Rezkalla, S. H. & Kloner, R. A. Coronary no-reflow phenomenon: from the experimental laboratory to the cardiac catheterization laboratory. Catheter. Cardiovasc. Interv. 72, 950–957 (2008).
Vinten-Johansen, J. Involvement of neutrophils in the pathogenesis of lethal myocardial reperfusion injury. Cardiovasc. Res. 61, 481–497 (2004).
Dobaczewski, M., Gonzalez-Quesada, C. & Frangogiannis, N. G. The extracellular matrix as a modulator of the inflammatory and reparative response following myocardial infarction. J. Mol. Cell Cardiol. 48, 504–511 (2009).
Spinale, F. G. Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiol. Rev. 87, 1285–1342 (2007).
Matzinger, P. The danger model: a renewed sense of self. Science 296, 301–305 (2002).
Seong, S. Y. & Matzinger, P. Hydrophobicity: an ancient damage-associated molecular pattern that initiates innate immune responses. Nat. Rev. Immunol. 4, 469–478 (2004).
Ionita, M. G., Arslan, F., de Kleijn, D. P. & Pasterkamp, G. Endogenous inflammatory molecules engage Toll-like receptors in cardiovascular disease. J. Innate. Immun. 2, 307–315 (2010).
Blasius, A. L. & Beutler, B. Intracellular toll-like receptors. Immunity 32, 305–315 (2010).
Arslan, F. et al. Myocardial ischemia/reperfusion injury is mediated by leukocytic toll-like receptor-2 and reduced by systemic administration of a novel anti-toll-like receptor-2 antibody. Circulation 121, 80–90 (2010).
Akira, S. & Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511 (2004).
Hansson, G. K. Immune and inflammatory mechanisms in the development of atherosclerosis. Br. Heart J. 69, S38–S41 (1993).
Brodison, A. & Swann, J. W. Myocarditis: a review. J. Infect. 37, 99–103 (1998).
Hansson, G. K. & Edfeldt, K. Toll to be paid at the gateway to the vessel wall. Arterioscler. Thromb. Vasc. Biol. 25, 1085–1087 (2005).
Li, M., Zhou, Y., Feng, G. & Su, S. B. The critical role of Toll-like receptor signaling pathways in the induction and progression of autoimmune diseases. Curr. Mol. Med. 9, 365–374 (2009).
Frantz, S., Ertl, G. & Bauersachs, J. Toll-like receptor signaling in the ischemic heart. Front. Biosci. 13, 5772–5779 (2008).
Kumar, H., Kawai, T. & Akira, S. Toll-like receptors and innate immunity. Biochem. Biophys. Res. Commun. 388, 621–625 (2009).
Arslan, F., Keogh, B., McGuirk, P. & Parker, A. E. TLR2 and TLR4 in ischemia reperfusion injury. Mediators Inflamm. doi:10.1155/2010/704202.
Favre, J. et al. Toll-like receptors 2-deficient mice are protected against postischemic coronary endothelial dysfunction. Arterioscler. Thromb. Vasc. Biol. 27, 1064–1071 (2007).
Knuefermann, P. et al. Toll-like receptor 2 mediates Staphylococcus aureus-induced myocardial dysfunction and cytokine production in the heart. Circulation 110, 3693–3698 (2004).
Boyd, J. H., Mathur, S., Wang, Y., Bateman, R. M. & Walley, K. R. Toll-like receptor stimulation in cardiomyoctes decreases contractility and initiates an NF-kappaB dependent inflammatory response. Cardiovasc. Res. 72, 384–393 (2006).
Sakata, Y. et al. Toll-like receptor 2 modulates left ventricular function following ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 292, H503–H509 (2007).
Landmesser, U., Wollert, K. C. & Drexler, H. Potential novel pharmacological therapies for myocardial remodelling. Cardiovasc. Res. 81, 519–527 (2009).
Cohn, J. N., Ferrari, R. & Sharpe, N. Cardiac remodeling—concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J. Am. Coll. Cardiol. 35, 569–582 (2000).
Shishido, T. et al. Toll-like receptor-2 modulates ventricular remodeling after myocardial infarction. Circulation 108, 2905–2910 (2003).
Timmers, L. et al. Toll-like receptor 4 mediates maladaptive left ventricular remodeling and impairs cardiac function after myocardial infarction. Circ. Res. 102, 257–264 (2008).
Satoh, M. et al. Activated toll-like receptor 4 in monocytes is associated with heart failure after acute myocardial infarction. Int. J. Cardiol. 109, 226–234 (2006).
Timmers, L. et al. Targeted deletion of nuclear factor kappaB p50 enhances cardiac remodeling and dysfunction following myocardial infarction. Circ. Res. 104, 699–706 (2009).
Piper, H. M., Abdallah, Y. & Schafer, C. The first minutes of reperfusion: a window of opportunity for cardioprotection. Cardiovasc. Res. 61, 365–371 (2004).
Eefting, F. et al. Role of apoptosis in reperfusion injury. Cardiovasc. Res. 61, 414–426 (2004).
Inserte, J., Barrabes, J. A., Hernando, V. & Garcia-Dorado, D. Orphan targets for reperfusion injury. Cardiovasc. Res. 83, 169–178 (2009).
Gustafsson, A. B. & Gottlieb, R. A. Heart mitochondria: gates of life and death. Cardiovasc. Res. 77, 334–343 (2008).
Garrido, C., Gurbuxani, S., Ravagnan, L. & Kroemer, G. Heat shock proteins: endogenous modulators of apoptotic cell death. Biochem. Biophys. Res. Commun. 286, 433–442 (2001).
Kol, A., Lichtman, A. H., Finberg, R. W., Libby, P. & Kurt-Jones, E. A. Cutting edge: heat shock protein (HSP) 60 activates the innate immune response: CD14 is an essential receptor for HSP60 activation of mononuclear cells. J. Immunol. 164, 13–17 (2000).
Ohashi, K., Burkart, V., Flohe, S. & Kolb, H. Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. J. Immunol. 164, 558–561 (2000).
Tissieres, A., Mitchell, H. K. & Tracy, U. M. Protein synthesis in salivary glands of Drosophila melanogaster: relation to chromosome puffs. J. Mol. Biol. 84, 389–398 (1974).
Young, J. C. Mechanisms of the Hsp70 chaperone system. Biochem. Cell. Biol. 88, 291–300 (2010).
Daugaard, M., Rohde, M. & Jaattela, M. The heat shock protein 70 family: highly homologous proteins with overlapping and distinct functions. FEBS Lett. 581, 3702–3710 (2007).
Currie, R. W. & White, F. P. Trauma-induced protein in rat tissues: a physiological role for a “heat shock” protein? Science 214, 72–73 (1981).
Currie, R. W. Effects of ischemia and perfusion temperature on the synthesis of stress-induced (heat shock) proteins in isolated and perfused rat hearts. J. Mol. Cell Cardiol. 19, 795–808 (1987).
Yellon, D. M. & Latchman, D. S. Stress proteins and myocardial protection. J. Mol. Cell Cardiol. 24, 113–124 (1992).
Gupta, M., Vavasis, C. & Frishman, W. H. Heat shock proteins in cardiovascular disease a new therapeutic target. Cardiol. Rev. 12, 26–30 (2004).
Fan, G. C., Chu, G. & Kranias, E. G. Hsp20 and its cardioprotection. Trends Cardiovasc. Med. 15, 138–141 (2005).
Tanonaka, K., Yoshida, H., Toga, W., Furuhama, K. & Takeo, S. Myocardial heat shock proteins during the development of heart failure. Biochem. Biophys. Res. Commun. 283, 520–525 (2001).
Knowlton, A. A. et al. Differential expression of heat shock proteins in normal and failing human hearts. J. Mol. Cell Cardiol. 30, 811–818 (1998).
Yellon, D. M. et al. Whole body heat stress fails to limit infarct size in the reperfused rabbit heart. Cardiovasc. Res. 26, 342–346 (1992).
Legare, J. F., Oxner, A., Heimrath, O., Myers, T. & Currie, R. W. Heat shock treatment results in increased recruitment of labeled PMN following myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 293, H3210–H3215 (2007).
Zal, B. et al. Heat-shock protein 60-reactive CD4+CD28null T cells in patients with acute coronary syndromes. Circulation 109, 1230–1235 (2004).
Lin, L. et al. HSP60 in heart failure: abnormal distribution and role in cardiac myocyte apoptosis. Am. J. Physiol. Heart Circ. Physiol. 293, H2238–H2247 (2007).
Frantz, S. et al. Toll4 (TLR4) expression in cardiac myocytes in normal and failing myocardium. J. Clin. Invest. 104, 271–280 (1999).
Kim, S. C. et al. Extracellular heat shock protein 60, cardiac myocytes, and apoptosis. Circ. Res. 105, 1186–1195 (2009).
Olivetti, G. et al. Apoptosis in the failing human heart. N. Engl. J. Med. 336, 1131–1141 (1997).
Sharma, A. K., Dhingra, S., Khaper, N. & Singal, P. K. Activation of apoptotic processes during transition from hypertrophy to heart failure in guinea pigs. Am. J. Physiol. Heart Circ. Physiol. 293, H1384–H1390 (2007).
Dorn, G. W. Apoptotic and non-apoptotic programmed cardiomyocyte death in ventricular remodelling. Cardiovasc. Res. 81, 465–473 (2009).
Park, M. et al. Apoptosis predominates in nonmyocytes in heart failure. Am. J. Physiol. Heart Circ. Physiol. 297, H785–H791 (2009).
Muller, S., Ronfani, L. & Bianchi, M. E. Regulated expression and subcellular localization of HMGB1, a chromatin protein with a cytokine function. J. Intern. Med. 255, 332–343 (2004).
Scaffidi, P., Misteli, T. & Bianchi, M. E. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature 418, 191–195 (2002).
Lotze, M. T. & Tracey, K. J. High-mobility group box 1 protein (HMGB1): nuclear weapon in the immune arsenal. Nat. Rev. Immunol. 5, 331–342 (2005).
Bonaldi, T. et al. Monocytic cells hyperacetylate chromatin protein HMGB1 to redirect it towards secretion. EMBO J. 22, 5551–5560 (2003).
Rendon-Mitchell, B. et al. IFN-gamma induces high mobility group box 1 protein release partly through a TNF-dependent mechanism. J. Immunol. 170, 3890–3897 (2003).
Chen, G. et al. Bacterial endotoxin stimulates macrophages to release HMGB1 partly through. J. Leukoc. Biol. 76, 994–1001 (2004).
Li, W., Sama, A. E. & Wang, H. Role of HMGB1 in cardiovascular diseases. Curr. Opin. Pharmacol. 6, 130–135 (2006).
Park, J. S. et al. Involvement of toll-like receptors 2 and 4 in cellular activation by high mobility group box 1 protein. J. Biol. Chem. 279, 7370–7377 (2004).
Ivanov, S. et al. A novel role for HMGB1 in TLR9-mediated inflammatory responses to CpG-DNA. Blood 110, 1970–1981 (2007).
Tian, J. et al. Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE. Nat. Immunol. 8, 487–496 (2007).
Kohno, T. et al. Role of high-mobility group box 1 protein in post-infarction healing process and left ventricular remodelling. Cardiovasc. Res. 81, 565–573 (2009).
Takahashi, K. et al. Modulated inflammation by injection of high-mobility group box 1 recovers post-infarction chronically failing heart. Circulation 118, S106–S114 (2008).
Goldstein, R. S. et al. Elevated high-mobility group box 1 levels in patients with cerebral and myocardial ischemia. Shock 25, 571–574 (2006).
Kitahara, T. et al. High-mobility group box 1 restores cardiac function after myocardial infarction in transgenic mice. Cardiovasc. Res. 80, 40–46 (2008).
Limana, F. et al. Exogenous high-mobility group box 1 protein induces myocardial regeneration after infarction via enhanced cardiac C-kit+ cell proliferation and differentiation. Circ. Res. 97, e73–e83 (2005).
Rossini, A. et al. HMGB1-stimulated human primary cardiac fibroblasts exert a paracrine action on human and murine cardiac stem cells. J. Mol. Cell Cardiol. 44, 683–693 (2008).
Andrassy, M. et al. High-mobility group Box-1 in ischemia-reperfusion injury of the heart. Circulation 117, 3216–3226 (2008).
Tzeng, H. P. et al. Negative inotropic effects of high-mobility group box 1 protein in isolated contracting cardiac myocytes. Am. J. Physiol. Heart Circ. Physiol. 294, H1490–H1496 (2008).
Wang, H. et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science 285, 248–251 (1999).
Yang, H. et al. Reversing established sepsis with antagonists of endogenous high-mobility group box 1. Proc. Natl Acad. Sci. USA 101, 296–301 (2004).
Natanson, C. et al. Role of endotoxemia in cardiovascular dysfunction and mortality. Escherichia coli and Staphylococcus aureus challenges in a canine model of human septic shock. J. Clin. Invest. 83, 243–251 (1989).
Ono, M. Molecular links between tumor angiogenesis and inflammation: inflammatory stimuli of macrophages and cancer cells as targets for therapeutic strategy. Cancer Sci. 99, 1501–1506 (2008).
vid Dong, Z. M., Aplin, A. C. & Nicosia, R. F. Regulation of angiogenesis by macrophages, dendritic cells, and circulating myelomonocytic cells. Curr. Pharm. Des. 15, 365–379 (2009).
Srikrishna, G. & Freeze, H. H. Endogenous damage-associated molecular pattern molecules at the crossroads of inflammation and cancer. Neoplasia 11, 615–628 (2009).
Sharma, A., Ray, R. & Rajeswari, M. R. Overexpression of high mobility group (HMG) B1 and B2 proteins directly correlates with the progression of squamous cell carcinoma in skin. Cancer Invest. 26, 843–851 (2008).
Dobaczewski, M. et al. Extracellular matrix remodeling in canine and mouse myocardial infarcts. Cell Tissue Res. 324, 475–488 (2006).
Comper, W. D. & Laurent, T. C. Physiological function of connective tissue polysaccharides. Physiol. Rev. 58, 255–315 (1978).
Waldenstrom, A., Martinussen, H. J., Gerdin, B. & Hallgren, R. Accumulation of hyaluronan and tissue edema in experimental myocardial infarction. J. Clin. Invest. 88, 1622–1628 (1991).
Termeer, C. et al. Oligosaccharides of Hyaluronan activate dendritic cells via toll-like receptor 4. J. Exp. Med. 195, 99–111 (2002).
Scheibner, K. A. et al. Hyaluronan fragments act as an endogenous danger signal by engaging TLR2. J. Immunol. 177, 1272–1281 (2006).
Gariboldi, S. et al. Low molecular weight hyaluronic acid increases the self-defense of skin epithelium by induction of beta-defensin 2 via TLR2 and TLR4. J. Immunol. 181, 2103–2110 (2008).
Jiang, D. et al. Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat. Med. 11, 1173–1179 (2005).
Zheng, L., Riehl, T. E. & Stenson, W. F. Regulation of colonic epithelial repair in mice by Toll-like receptors and hyaluronic acid. Gastroenterology 137, 2041–2051 (2009).
Liao, Y. F., Gotwals, P. J., Koteliansky, V. E., Sheppard, D. & Van De, W. L. The EIIIA segment of fibronectin is a ligand for integrins alpha 9beta 1 and alpha 4beta 1 providing a novel mechanism for regulating cell adhesion by alternative splicing. J. Biol. Chem. 277, 14467–14474 (2002).
Schoneveld, A. H. et al. Atherosclerotic lesion development and Toll like receptor 2 and 4 responsiveness. Atherosclerosis 197, 95–104 (2008).
Arslan, F. et al. Lack of fibronectin-EDA promotes survival and prevents adverse remodeling and heart function deterioration after myocardial infarction. Circ. Res. 108, 582–592 (2011).
Li, Y., Heuser, J. S., Cunningham, L. C., Kosanke, S. D. & Cunningham, M. W. Mimicry and antibody-mediated cell signaling in autoimmune myocarditis. J. Immunol. 177, 8234–8240 (2006).
Fairweather, D. et al. Complement receptor 1 and 2 deficiency increases coxsackievirus B3-induced myocarditis, dilated cardiomyopathy, and heart failure by increasing macrophages, IL-1beta, and immune complex deposition in the heart. J. Immunol. 176, 3516–3524 (2006).
Zhang, P., Cox, C. J., Alvarez, K. M. & Cunningham, M. W. Cutting edge: cardiac myosin activates innate immune responses through TLRs. J. Immunol. 183, 27–31 (2009).
Erridge, C. & Samani, N. J. Saturated fatty acids do not directly stimulate Toll-like receptor signaling. Arterioscler. Thromb. Vasc. Biol. 29, 1944–1949 (2009).
Tsan, M. F. & Gao, B. Endogenous ligands of Toll-like receptors. J. Leukoc. Biol. 76, 514–519 (2004).
Tsan, M. F. & Baochong, G. Pathogen-associated molecular pattern contamination as putative endogenous ligands of Toll-like receptors. J. Endotoxin. Res. 13, 6–14 (2007).
Zhu, X. et al. Toll-like receptor 2 activation by bacterial peptidoglycan-associated lipoprotein activates cardiomyocyte inflammation and contractile dysfunction. Crit. Care Med. 35, 886–892 (2007).
Schoneveld, A. H. et al. Toll-like receptor 2 stimulation induces intimal hyperplasia and atherosclerotic lesion development. Cardiovasc. Res. 66, 162–169 (2005).
Bjorkbacka, H. Multiple roles of Toll-like receptor signaling in atherosclerosis. Curr. Opin. Lipidol. 17, 527–533 (2006).
Hardarson, H. S. et al. Toll-like receptor 3 is an essential component of the innate stress response in virus-induced cardiac injury. Am. J. Physiol. Heart Circ. Physiol. 292, H251–H258 (2007).
Richer, M. J., Lavallee, D. J., Shanina, I. & Horwitz, M. S. Toll-like receptor 3 signaling on macrophages is required for survival following coxsackievirus B4 infection. PLoS One 4, e4127 (2009).
Oyama, J. et al. Reduced myocardial ischemia-reperfusion injury in toll-like receptor 4-deficient mice. Circulation 109, 784–789 (2004).
Fairweather, D. et al. IL-12 receptor beta 1 and Toll-like receptor 4 increase IL-1 beta- and IL-18-associated myocarditis and coxsackievirus replication. J. Immunol. 170, 4731–4737 (2003).
Vink, A. et al. In vivo evidence for a role of toll-like receptor 4 in the development of intimal lesions. Circulation 106, 1985–1990 (2002).
Triantafilou, K. et al. Human cardiac inflammatory responses triggered by Coxsackie B viruses are mainly Toll-like receptor (TLR) 8-dependent. Cell Microbiol. 7, 1117–1126 (2005).
Yu, Q. et al. IL-18 induction of osteopontin mediates cardiac fibrosis and diastolic dysfunction in mice. Am. J. Physiol. Heart Circ. Physiol. 297, H76–H85 (2009).
Bulicheva, N. et al. Effect of cell-free DNA of patients with cardiomyopathy and rDNA on the frequency of contraction of electrically paced neonatal rat ventricular myocytes in culture. Ann. N. Y. Acad. Sci. 1137, 273–277 (2008).
Knuefermann, P. et al. Bacterial DNA induces myocardial inflammation and reduces cardiomyocyte contractility: role of toll-like receptor 9. Cardiovasc. Res. 78, 26–35 (2008).
Chen, L. et al. TLR signals promote IL-6/IL-17-dependent transplant rejection. J. Immunol. 182, 6217–6225 (2009).
Acknowledgements
The authors would like to thank Professor N. G. Frangogiannis for his critical review of the manuscript. This work is supported by research grants from The Netherlands Organization for Scientific Research and Utrecht University Mozaïek grant (contract 017.004.004 to F. Arslan) and Netherlands Heart Foundation (contract 2010T001 to F. Arslan).
Author information
Authors and Affiliations
Contributions
F. Arslan researched data and wrote the manuscript. G. Pasterkamp and D. P. de Kleijn reviewed and edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Arslan, F., de Kleijn, D. & Pasterkamp, G. Innate immune signaling in cardiac ischemia. Nat Rev Cardiol 8, 292–300 (2011). https://doi.org/10.1038/nrcardio.2011.38
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2011.38
This article is cited by
-
Protective effects of macrophage-specific integrin α5 in myocardial infarction are associated with accentuated angiogenesis
Nature Communications (2023)
-
Macrophages in cardiac remodelling after myocardial infarction
Nature Reviews Cardiology (2023)
-
The role of periostin in cardiac fibrosis
Heart Failure Reviews (2023)
-
Spike-based adenovirus vectored COVID-19 vaccine does not aggravate heart damage after ischemic injury in mice
Communications Biology (2022)
-
Dexmedetomidine attenuates myocardial ischemia-reperfusion injury in vitro by inhibiting NLRP3 Inflammasome activation
BMC Anesthesiology (2021)