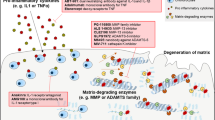
Key Points
-
Osteoarthritis (OA) is a painful, complex disease that affects millions of people worldwide. There is currently no disease-modifying therapy for OA, and existing drugs to treat symptoms of OA are largely ineffective. The lack of a validated animal model and the multifactorial nature of the disease make the discovery and development of new drugs even more challenging.
-
OA is a disease of the whole joint that involves many pathophysiological processes that arise from a dysregulation in the function of cytokines and growth factors, prostaglandins, cartilage matrix fragments, neuropeptides, reactive oxygen intermediates, proteolytic enzymes and protease inhibitors. Dysregulation of these factors sets in motion a cycle of degeneration of cartilage, bone, ligaments and synovium that coincides with an inflammatory response and peripheral and central nervous system sensitization.
-
Current treatments for OA include acetaminophen, non-steroidal anti-inflammatories, cyclooxygenase 2 (COX2) inhibitors, and intra-articular hyaluronic acid or steroid injections. However, disease-modifying efficacy has not been demonstrated for any of these drugs. These drugs are only moderately effective, leaving the patients with a substantial pain burden and often gastrointestinal side effects after chronic administration.
-
Disease-modifying drugs currently in various stages of clinical development include inhibitors against matrix metalloproteinases, interleukin-1β-convertase (ICE) and cathepsin K, and the nutraceutical glucosamine. Inhibitors against lipoxygenase and COX2, as well as nitric oxide analgesics and novel NSAIDS, are being tested in the clinic for the symptomatic treatment of OA.
-
The complex pathology of OA calls for a systems biology approach to dissecting the molecular pathways of disease initiation and progression, and to this end much effort is now focused on identifying and validating biomarkers and disease models for OA.
Abstract
Osteoarthritis is a painful and disabling disease that affects millions of patients. Its aetiology is largely unknown, but is most likely multi-factorial. Osteoarthritis poses a dilemma: it often begins attacking different joint tissues long before middle age, but cannot be diagnosed until it becomes symptomatic decades later, at which point structural alterations are already quite advanced. In this review, osteoarthritis is considered as a disease of the whole joint that may result from multiple pathophysiological mechanisms, one of which is the dysregulation of lipid homeostasis. No proven disease-modifying therapy exists for osteoarthritis and current treatment options for chronic osteoarthritic pain are insufficient, but new pharmacotherapeutic options are emerging.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Harris, E. D. Jr. The Bone and Joint decade: a catalyst for progress. Arthritis Rheum 44, 1969–1970 (2004).
WHO Scientific group. The burden of musculoskeletal conditions at the start of the new millennium. World Health Organ Tech. Rep. Ser. 919, 1–218 (2003). The tremendous challenge of the disease is summarized in this report of the epidemiology of musculoskeletal diseases.
Merx, H. et al. International variation in hip replacement rates. Ann. Rheum. Dis. 62, 222–226 (2003).
NIH Consensus Statement on Total Knee Replacement December 8–10, 2003. J. Bone Joint Surg. Am. 86, 1328–1335 (2004).
Kean, W. F., Kean, R. & Buchanan, W. W. Osteoarthritis: symptoms, signs and source of pain. Inflammo-pharmacology 12, 3–31 (2004).
Badley, E. M. & Wang, P. P. Arthritis and the aging population: projections of arthritis prevalence in Canada 1991 to 2031. J. Rheumatol. 25, 138–144 (1998).
Decision Resources, Inc. Osteoarthritis: Immune and Inflammatory Disorders Study (Decision Resources, Inc., Waltham, Massachusetts, 2004).
Hannan, M. T., Felson, D. T. & Pincus, T. Analysis of the discordance between radiographic changes and knee pain in osteoarthritis of the knee. J. Rheumatol. 27, 1513–1517 (2000).
Flower, R. J. The development of COX2 inhibitors. Nature Rev. Drug. Discov. 2, 179–191 (2003). A comprehensive review of COX2 inhibitor development.
Topol, E. J. Failing the public health — Rofecoxib, Merck, and the FDA. N. Engl. J. Med. 351, 1707–1711 (2004).
Petit-Zeman, S. Characteristics of COX 2 inhibitors questioned. Nature Rev. Drug. Discov. 3, 726–727 (2004). This paper and reference10 provide evidence that the safety of COX2 inhibitors will be re-evaluated.
Felson, D. T. An update on the pathogenesis and epidemiology of osteoarthritis. Radiol. Clin. N. Am. 42, 1–9 (2004).
Spector, T. D. & MacGregor, A. J. Risk factors for osteoarthritis: genetics. Osteoarthr. Cartil. 12, S39–S44 (2004).
Richette, P. & Bardin, T. Structure-modifying agents for osteoarthritis: an update. Joint Bone Spine 71, 18–23 (2004).
Jordan, K. M. et al. EULAR Recommendations 2003: an evidence based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann. Rheum. Dis. 62, 1145–1155 (2003). Recent clinical recommendations issued by an osteoarthritis task force.
Zhang, W., Jones, A. & Doherty, M. Does paracetamol (acetaminophen) reduce the pain of osteoarthritis? A meta-analysis of randomised controlled trials. Ann. Rheum. Dis. 63, 901–907 (2004).
Neame, R., Zhang, W. & Doherty, M. A historic issue of the Annals: three papers examine paracetamol in osteoarthritis. Ann. Rheum. Dis. 63, 897–900 (2004).
Lee, C., Straus, W. L., Balshaw, R., Barlas, S., Vogel, S. & Schnitzer, T. J. A comparison of the efficacy and safety of nonsteroidal antiinflammatory agents versus acetaminophen in the treatment of osteoarthritis: a meta-analysis. Arthritis Rheum. 51, 746–754 (2004).
Vane, J. R., Bakhle, Y. S. & Botting, R. M. Cyclooxygenases 1 and 2. Ann. Rev. Pharmacol. Toxicol. 38, 97–120 (1998).
Samad, T. A. et al. Interleukin-1β-mediated induction of COX-2 in the CNS contributes to inflammatory pain hypersensitivity. Nature 410, 471–475 (2001).
Watson, M. C., Brookes, S. T., Kirwan, J. R. & Faulkner, A. in The Cochrane Library, Issue 1 10.1002/14651858.CD000142 (Update Software Ltd, Oxford, 2003).
Warner, T. D. et al. Nonsteroid drug selectivities for cyclo-oxygenase-1 rather than cyclo-oxygenase-2 are associated with human gastrointestinal toxicity: a full in vitro analysis. Proc. Natl Acad. Sci. USA 96, 7563–7568 (1999).
Bensen, W. G. et al. Treatment of osteoarthritis with celecoxib, a cyclooxygenase-2 inhibitor: a randomized controlled trial. Mayo Clin. Proc. 74, 1095–1105 (1999).
Day, R. et al. A randomized trial of the efficacy and tolerability of the COX-2 inhibitor rofecoxib vs ibuprofen in patients with osteoarthritis. Rofecoxib/Ibuprofen Comparator Study Group. Arch. Intern. Med. 160, 1781–1787 (2000).
McAlindon, T. E., LaValley, M. P., Gulin, J. P. & Felson, D. T. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. JAMA 283, 1469–1475 (2000).
Zerkak, D. & Dougados, M. The use of glucosamine therapy in osteoarthritis. Curr. Pain Headache Rep. 8, 507–511 (2004).
Chard, J & Dieppe, P. Glucosamine for osteoarthritis: magic, hype, or confusion? It's probably safe — but there's no good evidence that it works. BMJ 322, 1439–1440 (2001). This paper questions the validity of glucosamine as a treatment for OA.
Biggee, B. A. & McAlindon, T. Glucosamine for osteoarthritis: part I, review of the clinical evidence. Med. Health RI 87, 176–179 (2004).
Lin, J., Zhang, W., Jones, A. & Doherty, M. Efficacy of topical non-steroidal anti-inflammatory drugs in the treatment of osteoarthritis: meta–analysis of randomised controlled trials. BMJ 329, 324 (2004).
Mason, L., Moore, R. A., Edwards, J. E., Derry, S. & McQuay, H. J. Topical NSAIDs for chronic musculoskeletal pain: systematic review and meta-analysis. BMC Musculoskelet. Disord. 5, 28 (2004).
Ayral, X. Injections in the treatment of osteoarthritis. Best Pract. Res. Clin. Rheumatol. 15, 609–626 (2001).
Gossec, L. & Dougados, M. Intra-articular treatments in osteoarthritis: from the symptomatic to the structure modifying. Ann. Rheum. Dis. 63, 478–482 (2004).
Creamer, P. Intra-articular corticosteroid treatment in osteoarthritis. Curr. Opin. Rheumatol. 11, 417–421 (1999).
Lohmander, L. S. et al. Intra-articular hyaluronan injections in the treatment of osteoarthritis of the knee: a randomised, double blind, placebo controlled multicentre trial. Hyaluronan Multicentre Trial Group. Ann. Rheum. Dis. 55, 424–431 (1996).
Brandt, K. D., Smith, G. N. Jr & Simon, L. S. Intraarticular injection of hyaluronan as treatment for knee osteoarthritis: what is the evidence? Arthritis Rheum. 43, 1192–1203 (2000).
Lo, G. H., LaValley, M., McAlindon, T. & Felson, D. T. Intra-articular hyaluronic acid in treatment of knee osteoarthritis: a meta-analysis. JAMA. 290, 3115–3221 (2003).
Berenbaum, F. Signaling transduction. Curr. Opin. Rheumatol. 16, 616–622 (2004).
Haringman, J. J., Ludikhuize, J. & Tak, P. P. Chemokines in joint disease: the key to inflammation? Ann. Rheum. Dis. 63, 1186–1194 (2004).
Aigner, T. et al. Functional genomics of osteoarthritis. Pharmacogenomics 3, 635–650 (2002).
Fan, Z. et al. Regulation of anabolic and catabolic gene expression in normal and osteoarthritic adult human articular chondrocytes by osteogenic protein-1. Clin. Exp. Rheumatol. 22, 103–106 (2004).
Sandell, L. J. & Aigner, T. Articular cartilage and changes in osteoarthritis. An introduction: cell biology of osteoarthritis. Arthritis Res. 3, 107–113 (2001).
Matsumoto, T., Gargosky, S. E., Iwasaki, K. & Rosenfeld, R. G. Identification and characterization of insulin-like growth factors (IGFs), IGF-binding proteins (IGFBPs), and IGFBP proteases in human synovial fluid. J. Clin. Endocrinol. Metab. 81, 150–155 (1996).
Lajeunesse, D. The role of bone in the treatment of osteoarthritis. Osteoarthr. Cartil. 12, S34–S38 (2004).
Dumond, H. et al. Evidence for a key role of leptin in osteoarthritis. Arthritis Rheum. 48, 3118–3129 (2003). A recent perspective of leptin playing an important role in OA.
Figenschau, Y. et al. Human articular chondrocytes express functional leptin receptors. Biochem. Biophys. Res. Commun. 287, 190–197 (2001).
Lohmander, L. S., Atley, L. M., Pietka, T. A. & Eyre, D. R. The release of crosslinked peptides from type II collagen into human synovial fluid is increased soon after joint injury and in osteoarthritis. Arthritis Rheum. 48, 3130–3139 (2003).
Rudolphi, K. et al. Pralnacasan, an inhibitor of interleukin-1β converting enzyme, reduces joint damage in two murine models of osteoarthritis. Osteoarthr. Cartil. 11, 738–746 (2003).
Zhang, X., Mao, Z. & Yu, Ch. Suppression of early experimental osteoarthritis by gene transfer of interleukin-1 receptor antagonist and interleukin-10. J. Orthop. Res. 22, 742–750 (2004).
Fuchs, S. et al. Differential induction and regulation of matrix metalloproteinases in osteoarthritic tissue and fluid synovial fibroblasts. Osteoarthr. Cartil. 12, 409–418 (2004).
Kevorkian, L. et al. Expression profiling of metalloproteinases and their inhibitors in cartilage. Arthritis Rheum. 50, 131–141 (2004).
Francois, M. et al. Peroxisome proliferator-activated receptor-γ down regulates chondrocyte matrix meatlloproteinase-1 via a novel composite element. J. Biol. Chem. 279, 28411–28418 (2004).
Bordji, K. et al. Evidence for the presence of peroxisome proliferator-activated receptor (PPAR)α and γ and retinoid Z receptor cartilage. J. Biol. Chem. 275, 12243–12250 (2000).
Martel-Pelletier, M., Pelletier, J. P. & Fahni, H. Cyclooxygenase-2 and prostaglandins in articular tissues. Semin. Arthritis Rheum. 33, 155–167 (2003).
Fahmi, H., Pelletier, J. P., Mineau, F. & Martel-Pelletier, J. 15d-PGJ2 is acting as “dual agent” on the regulation of COX-2 expression in human osteoarthritic chondrocytes. Osteoarthr. Cartil. 10, 845–848 (2002).
Aupperle, K. R. et al. NF-κB regulation by I kappa B kinase in primary fibroblast-like synoviocytes. J. Immunol. 163, 427–433 (1999).
Aupperle, K. R. et al. Expression and regulation of inducible IκB kinase (IKK-i) in human fibroblast-like synoviocytes. Cell. Immunol. 214, 54–59 (2001).
Tegeder, I. et al. Specific inhibition of IκB kinase reduces hyperalgesia in inflammatory and neuropathic pain models. J. Neurosci. 24, 1673–1645 (2004).
Jobin, C. et al. Specific NF-κB blockade selectively inhibits tumour necrosis factor α-induced COX2 but not constitutive Cox-1 gene expression in HT29 cells. Immunology 95, 537–543 (1998).
Pelletier, J. -P. Rationale for the use of structure-modifying drugs and agents in the treatment of osteoarthritis. Osteoarthr. Cartil. 12 (Suppl. A), 63–68 (2003).
Buckland-Wright, C. Subchondral bone changes in hand and knee osteoarthritis detected by radiography. Osteoarthr. Cartil. 12, (Suppl. A) 10–19 (2004).
Felson, D. T. & Neogi, T. Osteoarthritis: is it a disease of cartilage or bone? Arthritis Rheum. 50, 341–344 (2004). A recent paper discussing the contribution of both cartilage and bone to the progression of the disease.
Grynpas, M. D., Alpert, B., Katz, I., Lieberman, I. & Pritzker, K. P. Subchondral bone in osteoarthritis. Calcif. Tissue Int. 49, 20–26 (1991).
Hayami, T. et al. The role of subchondral bone remodeling in osteoarthritis: reduction of cartilage degeneration and prevention of osteophyte formation by alendronate in the rat anterior cruciate ligament transection model. Arthritis Rheum. 50, 1193–1206 (2004).
Doschak, M. R., Wohl, G. R., Hanley, D. A., Bray, R. C. & Zernicke, R. F. Antiresorptive therapy conserves some periarticular bone and ligament mechanical properties after anterior cruciate ligament disruption in the rabbit knee. J. Orthop. Res. 22, 942–948 (2004).
Myers, S. L., Brandt, K. D., Burr, D. B., O'Connor, B. L. & Albrecht, M. Effects of a bisphosphonate on bone histomorphometry and dynamics in the canine cruciate deficiency model of osteoarthritis. J. Rheumatol. 26, 2645–2653 (1999).
Bingham, C. et al. Clinically significant placebo improvement occurs by 6 months and is maintained through 24 months in a study of knee OA pain and function. Osteoarthr. Cartil. 12, S132 Abs. P323 (2004).
Menkes, C. J. & Lane, N. E. Are osteophytes good or bad? Osteoarthr. Cartil. 12, S53–S54 (2004).
Uchino, M. et al. Growth factor expression in the osteophytes of the human femoral head in osteoarthritis. Clin. Orthop. 377, 119–125 (2000).
Brandt, K. D. Osteophytes in osteoarthritis. Clinical aspects. Osteoarthr. Cartil. 7, 334–335 (1999).
Van Osch, G. J., Van der Kraan, P. M., Van Valburg, A. A. & Van den Berg, W. B. The relation between cartilage damage and osteophyte size in a murine model for osteoarthritis in the knee. Rheumatol. Int. 16, 115–119 (1996).
Lanyon, P., O'Reilly, S., Jones, A. & Doherty, M. Radiographic assessment of symptomatic knee osteoarthritis in the community: definitions and normal joint space. Ann. Rheum. Dis. 57, 595–601 (1998).
Ghosh, P. Vascular mechanisms in osteoarthritis. Best Practice Res. Clin. Rheumatol. 15, 693–710 (2001).
Simkin, P. A. in Osteoarthritic Joint Pain (Novartis Foundation Symposium) 179–190 (Wiley, Chichester, 2004).
Takeda, S. et al. Leptin regulates bone formation via the sympathetic nervous system. Cell 111, 305–317 (2002).
Ducy, P. et al. Leptin inhibits bone formation through a hypothalamic relay: a central control of bone mass. Cell 100, 197–207 (2000).
Ahima, R. S. & Flier, J. S. Leptin. Annu. Rev. Physiol. 62, 413–437 (2000).
Dean, G., Hoyland, J. A., Denton, J., Donn, R. P. & Freemont, A. J. Mast cells in the synovium and synovial fluid in osteoarthritis. Br. J. Rheumatol. 32, 671–675 (1993).
Lindblad, S. & Hedfors, E. Arthroscopic and immunohistologic characterization of knee joint synovitis in osteoarthritis. Arthritis Rheumatol. 30, 1081–1088 (1987).
Halverson, P. B. & Derfus, B. A. Calcium crystal-induced inflammation. Curr Opin. Rheumatol. 13, 221–224 (2001).
Kato, T., Xiang, Y., Nakamura, H. & Nishioka, K. Neoantigens in osteoarthritic cartilage. Curr. Opin. Rheumatol. 16, 604–608 (2004).
Abramson, S. B. Inflammation in osteoarthritis. J. Rheumatol. Suppl. 70, 70–76 (2004).
Poole, A. R. Biochemical/immunochemical biomarkers of osteoarthritis: utility for prediction of incident or progressive osteoarthritis. Rheum. Dis. Clin. North Am. 29, 803–818 (2003). This paper provides a classic review on biomarkers in OA.
Mapp, P. I. Innervation of the synovium. Ann. Rheum. Dis. 54, 398–403 (1995).
Opree, A. & Kress, M. Involvement of the proinflammatory cytokines tumor necrosis factor-α, IL-1β, and IL-6 but not IL-8 in the development of heat hyperalgesia: effects on heat-evoked calcitonin gene-related peptide release from rat skin. J. Neurosci. 20, 6289–6293 (2000).
Safieh-Garabedian, B., Poole, S., Allchorne, A., Winter, J. & Woolf, C. J. Contribution of interleukin-1β to the inflammation-induced increase in nerve growth factor levels and inflammatory hyperalgesia. Br. J. Pharmacol. 115, 1265–1275 (1995).
Woolf, C. J., Allchorne, A., Safieh-Garabedian, B. & Poole, S. Cytokines, nerve growth factor and inflammatory hyperalgesia: the contribution of tumour necrosis factor α. Br. J. Pharmacol. 121, 417–424 (1997).
Chen, X., Tanner, K. & Levine, J. D. Mechanical sensitization of cutaneous C-fiber nociceptors by prostaglandin E2 in the rat. Neurosci. Lett. 267, 105–108 (1999).
Manning, D. C., Raja, S. N., Meyer, R. A. & Campbell, J. N. Pain and hyperalgesia after intradermal injection of bradykinin in humans. Clin. Pharmacol. Ther. 50, 721–729 (1991).
Mizumura, K. Natural history of nociceptor sensitization — the search for peripheral mechanism of hyperalgesia. Pain Rev. 5, 59–82 (1998).
Flechtenmacher, J. et al. Bradykinin-receptor-inhibition — a therapeutic option in osteoarthritis? Osteoarthr. Cartil. 12, S137 Abs. P332 (2004).
Saito, T. & Koshino, T. Distribution of neuropeptides in synovium of the knee with osteoarthritis. Clin. Orthop. 376, 172–182 (2000).
Vilensky, J. A. & Cook, J. A. Neurogenic acceleration of osteoarthritis. Curr. Opin. Rheumatol. 10, 251–255 (1998).
Hurley, M. V., Scott, D. L., Rees, J. & Newham, D. J. Sensorimotor changes and functional performance in patients with knee osteoarthritis. Ann. Rheum. Dis. 56, 641–648 (1997).
Baker, K. & McAlindon, T. Exercise for knee osteoarthritis. Curr. Opin. Rheumatol. 12, 456–463 (2000).
Slemenda, C. et al. Quadriceps weakness and osteoarthritis of the knee. Ann. Int. Med. 127, 97–104 (1997).
Becker, R., Berth, A., Nehring, M. & Awiszus, F. Neuromuscular quadriceps dysfunction prior to osteoarthritis of the knee. J. Orthop. Res. 22, 768–773 (2004).
O'Connor, B. L. & Vilensky, J. A. Peripheral and central nervous system mechanisms of joint protection. Am. J. Orthop. 32, 330–336 (2003).
Sharma, L., Dunlop, D. D., Cahue, S., Song, J. & Hayes, K. W. Quadriceps strength and osteoarthritis progression in malaligned and lax knees. Ann. Intern. Med. 138, 613–619 (2003).
Creamer, P., Hunt, M. & Dieppe, P. Pain mechanisms in osteoarthritis of the knee: effect of intraarticular anesthetic. J. Rheumatol. 23, 1031–1036 (1996).
Schaible, H. G., Ebersberger, A., Von Banchet, G. S. Mechanisms of pain in arthritis. Ann. NY Acad. Sci. 966, 343–354 (2002).
Kidd, B. L. & Urban, L. A. Mechanisms of inflammatory pain. Br. J. Anaesth. 87, 3–11 (2001).
US Department of Health and Human Services, Food and Drug Administration. Innovation and Stagnation: Challenge and Opportunity on the Critical Path to New Medical Products [online], <http://www.who.int/intellectualproperty/documents/en/FDAproposals.pdf> (2004).
Butcher, E. C., Berg, E. L. & Kunkel, E. J. Systems biology in drug discovery. Nature Biotechnology 22, 1253–1259 (2004).
Bonnet, C. S. & Walsh, D. A. Osteoarthritis, angiogenesis and inflammation. Rheumatology (Oxford) 44, 7–16 (2005). An interesting recent review illustrating the relevance of angiogenic processes to the pathophysiology of OA.
Walsh, D. A. Angiogenesis in osteoarthritis and spondylosis: successful repair with undesirable outcomes. Curr. Opin. Rheumatol. 16, 609–615 (2004).
Balint, G. & Szebenyi, B. Hereditary disorders mimicking and/or causing premature osteoarthritis. Baillieres Best. Pract. Res. Clin. Rheumatol. 14, 219–250 (2000).
Dequeker, J., Boonen, N., Aerssens, J. & Westhovens, R. Inverse relationship osteoarthritis — osteoporosis: what is the evidence? What are the consequences? Br. J. Rheumatol. 35, 813–820 (1996).
Lark, M. W. & Blake, S. M. Development of novel therapeutics to treat joint diseases — what can be learned from other diseases? Curr. Opin. Pharmacol. 4, 273–275 (2004).
Aspden, R. M., Scheven, B. A. A. & Hutchison, J. D. Osteoarthritis as a systemic disorder including stromal cell differentiation and lipid metabolism. Lancet 357, 1118–1120 (2001). One of the first publications suggesting a direct relationship between OA and metabolic disorders.
Toda, Y. et al. Change in body fat, but not body weight or metabolic correlates of obesity, is related to symptomatic relief of obese patients with knee osteoarthritis after weight control program. J. Rheumatol. 25, 2181–2186 (1998).
Gimble, J. M. & Nuttall, M. E. Bone and fat. Endocrine 23, 183–188 (2004)
Loeser, R. F. Systemic and local regulation of articular cartilage metabolism: where does leptin fit in the puzzle? Arthritis Rheum. 48, 3009–3012 (2003)
Yagami, K. et al. A human chondrogenic cell line retains multi-potency that differentiates into osteoblasts and adipocytes. Bone 34, 648–655 (2004).
Henroitin, Y., Sanchez, C. & Reginster, J. Y. The inhibition of metalloproteinases to treat osteoarthritis: reality and new perspectives. Expert Opin. Ther. Patents 12, 29–43 (2002).
Skiles, J. W., Gonella, N. C. & Jeng, A. Y. The design, structure and clinical update of small metalloproteinase inhibitors. Curr. Med. Chem. 11, 2911–2977 (2004).
Schnitzer, T. J. et al. TARGET Study Group. Comparison of lumiracoxib with naproxen and ibuprofen in the Therapeutic Arthritis Research and Gastrointestinal Event Trial (TARGET), reduction in ulcer complications: randomised controlled trial. Lancet. 364, 665–674 (2004).
Brune, K. Safety of anti-inflammatory treatment — new ways of thinking. Rheumatology (Oxford). 43 S16–S20 (2004).
Ding, C & Cicuttini, F. Licofelone (Merckle). IDrugs 6, 802–808 (2003).
Wallace, J. L. & Del Soldato, P. The therapeutic potential of NO-NSAIDs. Fundam. Clin. Pharmacol. 17, 11–20 (2003).
Whittle, B. J. Cyclooxygenase and nitric oxide systems in the gut as therapeutic targets for safer anti-inflammatory drugs. Curr. Opin. Pharmacol. 4, 538–545 (2004).
Schnitzer, T. J., Kivitz, A. J., Lipetz, S., Sanders, N. & Hee, A. A Phase II study of the efficacy and safety of AZD3582, a CINOD, in subjects with osteoarthritis of the knee. Eur. League Against Rheumatism 2004 Conference Abs OP0016 [online], <http://mcic3.textor.com/cgi-bin/mc/printabs.pl?APP=eular2004SCIE-abstract&TEMPLATE=&keyf=0094&showHide=show&client=> (2004)
Garnero, P. et al. Uncoupling of type II collagen synthesis and degradation predicts progression of joint damage in patients with knee osteoarthritis. Arthritis Rheum. 46, 2613–2624 (2002).
Cerejo, R. et al. The association between cartilage activity measured in serum and progression of knee osteoarthritis in patients with and without evidence of generalized disease. Arthritis Rheum. 46, S144 (2002).
Sharif, M. et al. Suggestion of nonlinear or phasic progression of knee osteoarthritis based on measurements of serum cartilage oligomeric matrix protein levels over five years. Arthritis Rheum. 50, 2479–2488 (2004).
Kola, I. & Landis, J. Can the pharmaceutical industry reduce attrition rates? Nature Rev. Drug Discovery 3, 711–715 (2004).
Acknowledgements
We wish to thank J. Pietsch and B. Schölkens for their valuable input to the preparation of this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors are employees of Aventis Pharma Deutschland GmbH, a company of the Sanofi-Aventis group which is developing drugs that target osteoarthritis.
Related links
Related links
DATABASES
Entrez Gene
OMIM
FURTHER INFORMATION
Glossary
- SYNOVITIS
-
Inflammation of the synovium, the tissue that produces joint-lubricating fluid.
- LEPTIN
-
A polypeptide hormone ligand related to the family of interleukin-6 cytokines encoded by the obese (ob) gene and secreted by adipocytes.
- ENDOCHONDRAL OSSIFICATION
-
A type of bone formation that occurs by replacement of hyaline cartilage.
- EBURNATION
-
Spikes of granulation and fibrous tissue reach the joint surface. The tissue undergoes endochondral ossification and penetrates the thinning cartilage, eventually exposing smooth, dense bone on the articular surface.
Rights and permissions
About this article
Cite this article
Wieland, H., Michaelis, M., Kirschbaum, B. et al. Osteoarthritis — an untreatable disease?. Nat Rev Drug Discov 4, 331–344 (2005). https://doi.org/10.1038/nrd1693
Issue Date:
DOI: https://doi.org/10.1038/nrd1693
This article is cited by
-
Functional Assessment of Human Articular Cartilage Using Second Harmonic Generation (SHG) Imaging: A Feasibility Study
Annals of Biomedical Engineering (2024)
-
Downregulation of lncRNA NEAT1 interacts with miR-374b-5p/PGAP1 axis to aggravate the development of osteoarthritis
Journal of Orthopaedic Surgery and Research (2023)
-
Fructose-bisphosphatase1 (FBP1) alleviates experimental osteoarthritis by regulating Protein crumbs homolog 3 (CRB3)
Arthritis Research & Therapy (2023)
-
MYL3 protects chondrocytes from senescence by inhibiting clathrin-mediated endocytosis and activating of Notch signaling
Nature Communications (2023)
-
Animal Models of Osteoarthritis: Updated Models and Outcome Measures 2016–2023
Regenerative Engineering and Translational Medicine (2023)