Abstract
Thyroid dysfunction is common in the general population, and mild or subclinical forms can be present in more than 10% of individuals aged >80 years. The diagnosis of abnormal thyroid hormone concentrations in people aged >60 years poses a challenge, as the clinical presentation of thyroid dysfunction is usually nonspecific, and ageing is associated with a number of physiological changes that can affect thyroid function test results. Furthermore, the presence of acute or chronic nonthyroidal illnesses and the use of medications that interfere with thyroid function tests are common confounders in the determination of thyroid status in the elderly. Early diagnosis and treatment of overt thyroid dysfunction is crucial in this population in view of the marked effects of abnormal circulating thyroid hormone levels on a number of organ systems, including the heart, the skeleton and the neurological system. The clinical significance of mild thyroid overactivity and underactivity remains uncertain, and the need for treatment of subclinical thyroid dysfunction is much debated. A number of large epidemiological studies have identified associations between mild thyroid dysfunction and short-term as well as long-term adverse outcomes, and a small but increasing number of randomized controlled intervention studies have been reported. Guidelines recommend treatment of thyroid dysfunction on the basis of the degree of abnormal serum TSH concentrations, patient age and associated comorbidities. This Review describes the current evidence on the prevalence, diagnosis, management and long-term consequences of thyroid dysfunction in the elderly.
Key Points
-
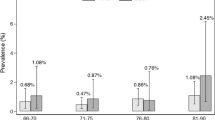
Large epidemiological studies confirm an increased prevalence of thyroid dysfunction in elderly people; subclinical or mild thyroid dysfunction is more prevalent than overt forms of thyroid hormone excess or deficiency
-
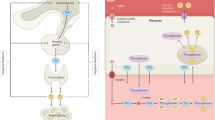
Diagnosis of thyroid dysfunction is difficult in elderly individuals given its sometimes asymptomatic clinical presentation and the effects of a number of physiological changes on the biochemical evaluation of thyroid function
-
Overt hyperthyroidism is associated with significantly increased risks of cardiovascular disease, osteoporosis and mortality, especially in the elderly population
-
Treatment of subclinical hyperthyroidism is warranted when serum TSH concentrations are undetectable owing to important associations with cardiovascular disease and reduced BMD
-
Biological end points of hyperthyroidism correlate with serum free T4 levels; risk of atrial fibrillation and fracture susceptibility are increased with serum free T4 concentrations at the upper limit of the reference range
-
Studies on the long-term consequences of hypothyroidism on cardiovascular disease risk are inconsistent, but treatment of serum TSH concentrations >10 mIU/l with levothyroxine monotherapy is recommended in most guidelines
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Christensen, K., Doblhammer, G., Rau, R. & Vaupel, J. W. Ageing populations: the challenges ahead. Lancet 374, 1196–1208 (2009).
Crimmins, E. M. Trends in the health of the elderly. Annu. Rev. Public Health 25, 79–98 (2004).
Manton, K. G. Recent declines in chronic disability in the elderly U. S. population: risk factors and future dynamics. Annu. Rev. Public Health 29, 91–113 (2008).
Tunbridge, W. M. et al. The spectrum of thyroid disease in a community: the Whickham survey. Clin. Endocrinol. (Oxf.) 7, 481–493 (1977).
Vanderpump, M. P. et al. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin. Endocrinol. (Oxf.) 43, 55–68 (1995).
Biondi, B. & Cooper, D. S. The clinical significance of subclinical thyroid dysfunction. Endocr. Rev. 29, 76–131 (2008).
Cooper, D. S. & Biondi, B. Subclinical thyroid disease. Lancet 379, 1142–1154 (2012).
Franklyn, J. A. The Thyroid—too much and too little across the ages. The consequences of subclinical thyroid dysfunction. Clin. Endocrinol. (Oxf.) 78, 1–8 (2013).
Roberts, C. G. & Ladenson, P. W. Hypothyroidism. Lancet 363, 793–803 (2004).
Garber, J. R. et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid 22, 1200–1235 (2012).
Sawin, C. T., Castelli, W. P., Hershman, J. M., McNamara, P. & Bacharach, P. The aging thyroid. Thyroid deficiency in the Framingham Study. Arch. Intern. Med. 145, 1386–1388 (1985).
Hollowell, J. G. et al. Serum TSH, T(4), and thyroid antibodies in the United States population 1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J. Clin. Endocrinol. Metab. 87, 489–499 (2002).
Canaris, G. J., Manowitz, N. R., Mayor, G. & Ridgway, E. C. The Colorado thyroid disease prevalence study. Arch. Intern. Med. 160, 526–534 (2000).
Bagchi, N., Brown, T. R. & Parish, R. F. Thyroid dysfunction in adults over age 55 years. A study in an urban US community. Arch. Intern. Med. 150, 785–787 (1990).
Bemben, D. A. et al. Thyroid disease in the elderly. Part 1. Prevalence of undiagnosed hypothyroidism. J. Fam. Pract. 38, 577–582 (1994).
Parle, J. V., Franklyn, J. A., Cross, K. W., Jones, S. C. & Sheppard, M. C. Prevalence and follow-up of abnormal thyrotrophin (TSH) concentrations in the elderly in the United Kingdom. Clin. Endocrinol. (Oxf.) 34, 77–83 (1991).
Szabolcs, I. et al. Comparative screening for thyroid disorders in old age in areas of iodine deficiency, long-term iodine prophylaxis and abundant iodine intake. Clin. Endocrinol. (Oxf.) 47, 87–92 (1997).
Wilson, S. et al. Prevalence of subclinical thyroid dysfunction and its relation to socioeconomic deprivation in the elderly: a community-based cross-sectional survey. J. Clin. Endocrinol. Metab. 91, 4809–4816 (2006).
Surks, M. I. & Hollowell, J. G. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 92, 4575–4582 (2007).
Surks, M. I. & Boucai, L. Age- and race-based serum thyrotropin reference limits. J. Clin. Endocrinol. Metab. 95, 496–502 (2010).
Golden, S. H., Robinson, K. A., Saldanha, I., Anton, B. & Ladenson, P. W. Clinical review: Prevalence and incidence of endocrine and metabolic disorders in the United States: a comprehensive review. J. Clin. Endocrinol. Metab. 94, 1853–1878 (2009).
Flynn, R. W., Macdonald, T. M., Morris, A. D., Jung, R. T. & Leese, G. P. The thyroid epidemiology, audit, and research study: thyroid dysfunction in the general population. J. Clin. Endocrinol. Metab. 89, 3879–3884 (2004).
Wilson, S. et al. Prevalence of subclinical thyroid dysfunction and its relation to socioeconomic deprivation in the elderly: a community-based cross-sectional survey. J. Clin. Endocrinol. Metab. 91, 4809–4816 (2006).
Cappola, A. R. et al. Thyroid status, cardiovascular risk, and mortality in older adults. JAMA 295, 1033–1041 (2006).
Vadiveloo, T., Donnan, P. T., Cochrane, L. & Leese, G. P. The Thyroid Epidemiology, Audit, and Research Study (TEARS): the natural history of endogenous subclinical hyperthyroidism. J. Clin. Endocrinol. Metab. 96, E1–E8 (2011).
Laurberg, P. et al. Iodine intake as a determinant of thyroid disorders in populations. Best Pract. Res. Clin. Endocrinol. Metab. 24, 13–27 (2010).
Laurberg, P. et al. Iodine intake and the pattern of thyroid disorders: a comparative epidemiological study of thyroid abnormalities in the elderly in Iceland and in Jutland, Denmark. J. Clin. Endocrinol. Metab. 83, 765–769 (1998).
Mahne, A. et al. Assessment of age-related morphological and functional changes of selected structures of the head and neck by computed tomography, magnetic resonance imaging, and positron emission tomography. Semin. Nucl. Med. 37, 88–102 (2007).
Mortensen, J. D., Woolner, L. B. & Bennett, W. A. Gross and microscopic findings in clinically normal thyroid glands. J. Clin. Endocrinol. Metab. 15, 1270–1280 (1955).
Gerber, D. Thyroid weights and iodized salt prophylaxis: a comparative study from autopsy material from the Institute of Pathology, University of Zurich [German]. Schweiz. Med. Wochenschr. 110, 2010–2017 (1980).
van den Beld, A. W., Visser, T. J., Feelders, R. A., Grobbee, D. E. & Lamberts, S. W. Thyroid hormone concentrations, disease, physical function, and mortality in elderly men. J. Clin. Endocrinol. Metab. 90, 6403–6409 (2005).
van, C. A. et al. Decreased basal and stimulated thyrotropin secretion in healthy elderly men. J. Clin. Endocrinol. Metab. 69, 177–185 (1989).
Mariotti, S. et al. Complex alteration of thyroid function in healthy centenarians. J. Clin. Endocrinol. Metab. 77, 1130–1134 (1993).
Lewis, G. F., Alessi, C. A., Imperial, J. G. & Refetoff, S. Low serum free thyroxine index in ambulating elderly is due to a resetting of the threshold of thyrotropin feedback suppression. J. Clin. Endocrinol. Metab. 73, 843–849 (1991).
Barreca, T., Franceschini, R., Messina, V., Bottaro, L. & Rolandi, E. 24-hour thyroid-stimulating hormone secretory pattern in elderly men. Gerontology 31, 119–123 (1985).
van, C. A. et al. Neuroendocrine rhythms and sleep in aging men. Am. J. Physiol. 260, E651–E661 (1991).
Gan, E. H. & Pearce, S. H. Clinical review: The thyroid in mind: cognitive function and low thyrotropin in older people. J. Clin. Endocrinol. Metab. 97, 3438–3449 (2012).
Gregerman, R. I., Gaffney, G. W., Shock, N. W. & Crowder, S. E. Thyroxine turnover in euthyroid man with special reference to changes with age. J. Clin. Invest. 41, 2065–2074 (1962).
Atzmon, G., Barzilai, N., Hollowell, J. G., Surks, M. I. & Gabriely, I. Extreme longevity is associated with increased serum thyrotropin. J. Clin. Endocrinol. Metab. 94, 1251–1254 (2009).
Atzmon, G., Barzilai, N., Surks, M. I. & Gabriely, I. Genetic predisposition to elevated serum thyrotropin is associated with exceptional longevity. J. Clin. Endocrinol. Metab. 94, 4768–4775 (2009).
Pappa, T. A., Vagenakis, A. G. & Alevizaki, M. The nonthyroidal illness syndrome in the non-critically ill patient. Eur. J. Clin. Invest. 41, 212–220 (2011).
Warner, M. H. & Beckett, G. J. Mechanisms behind the non-thyroidal illness syndrome: an update. J. Endocrinol. 205, 1–13 (2010).
Tognini, S. et al. Non-thyroidal illness syndrome and short-term survival in a hospitalised older population. Age Ageing 39, 46–50 (2010).
Adler, S. M. & Wartofsky, L. The nonthyroidal illness syndrome. Endocrinol. Metab. Clin. North Am. 36, 657–672, vi (2007).
Fliers, E., Alkemade, A. & Wiersinga, W. M. The hypothalamic–pituitary-thyroid axis in critical illness. Best Pract. Res. Clin. Endocrinol. Metab. 15, 453–464 (2001).
Fliers, E., Alkemade, A., Wiersinga, W. M. & Swaab, D. F. Hypothalamic thyroid hormone feedback in health and disease. Prog. Brain Res. 153, 189–207 (2006).
Fliers, E., Guldenaar, S. E., Wiersinga, W. M. & Swaab, D. F. Decreased hypothalamic thyrotropin-releasing hormone gene expression in patients with nonthyroidal illness. J. Clin. Endocrinol. Metab. 82, 4032–4036 (1997).
Donini, L. M. et al. Malnutrition in elderly: social and economic determinants. J. Nutr. Health Aging 17, 9–15 (2013).
Morley, J. E. Undernutrition in older adults. Fam. Pract. 29 (Suppl. 1), i89–i93 (2012).
Kundra, P., Burman, K. D. The effect of medications on thyroid function tests. Med. Clin. North Am. 96, 283–295 (2012).
Mitrou, P., Raptis, S. A. & Dimitriadis, G. Thyroid disease in older people. Maturitas 70, 5–9 (2011).
Trivalle, C. et al. Differences in the signs and symptoms of hyperthyroidism in older and younger patients. J. Am. Geriatr. Soc. 44, 50–53 (1996).
Mooradian, A. D. Asymptomatic hyperthyroidism in older adults: is it a distinct clinical and laboratory entity? Drugs Aging 25, 371–380 (2008).
Mariotti, S., Franceschi, C., Cossarizza, A. & Pinchera, A. The aging thyroid. Endocr. Rev. 16, 686–715 (1995).
Peeters, R. P. Thyroid hormones and aging. Hormones (Athens) 7, 28–35 (2008).
Kramer, C. K., von, M. D., Kritz-Silverstein, D. & Barrett-Connor, E. Treated hypothyroidism, cognitive function, and depressed mood in old age: the Rancho Bernardo Study. Eur. J. Endocrinol. 161, 917–921 (2009).
Doucet, J. et al. Does age play a role in clinical presentation of hypothyroidism? J. Am. Geriatr. Soc. 42, 984–986 (1994).
de Jongh, R. T. et al. Endogenous subclinical thyroid disorders, physical and cognitive function, depression, and mortality in older individuals. Eur. J. Endocrinol. 165, 545–554 (2011).
Roberts, L. M. et al. Is subclinical thyroid dysfunction in the elderly associated with depression or cognitive dysfunction? Ann. Intern. Med. 145, 573–581 (2006).
Simonsick, E. M. et al. Subclinical hypothyroidism and functional mobility in older adults. Arch. Intern. Med. 169, 2011–2017 (2009).
Nordyke, R. A., Gilbert, F. I. Jr & Harada, A. S. Graves' disease. Influence of age on clinical findings. Arch. Intern. Med. 148, 626–631 (1988).
Boelaert, K., Torlinska, B., Holder, R. L. & Franklyn, J. A. Older individuals with hyperthyroidism present with a paucity of symptoms and signs: a large cross-sectional study. J. Clin. Endocrinol. Metab. 95, 2715–2726 (2010).
Boelaert, K. et al. Prevalence and relative risk of other autoimmune diseases in individuals with autoimmune thyroid disease. Am. J. Med. 123, 183–189 (2010).
Broadley, S. A., Deans, J., Sawcer, S. J., Clayton, D. & Compston, D. A. Autoimmune disease in first-degree relatives of patients with multiple sclerosis. A UK survey. Brain 123, 1102–1111 (2000).
Walker, D. J., Griffiths, M. & Griffiths, I. D. Occurrence of autoimmune diseases and autoantibodies in multicase rheumatoid arthritis families. Ann. Rheum. Dis. 45, 323–326 (1986).
Kanaya, A. M. et al. Association between thyroid dysfunction and total cholesterol level in an older biracial population: the health, aging and body composition study. Arch. Intern. Med. 162, 773–779 (2002).
Desai, J. et al. Hypothyroidism after sunitinib treatment for patients with gastrointestinal stromal tumors. Ann. Intern. Med. 145, 660–664 (2006).
Kappers, M. H. et al. Sunitinib-induced hypothyroidism is due to induction of type 3 deiodinase activity and thyroidal capillary regression. J. Clin. Endocrinol. Metab. 96, 3087–3094 (2011).
Surks, M. I. et al. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA 291, 228–238 (2004).
Diez, J. J., Iglesias, P. & Burman, K. D. Spontaneous normalization of thyrotropin concentrations in patients with subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 90, 4124–4127 (2005).
Somwaru, L. L., Rariy, C. M., Arnold, A. M. & Cappola, A. R. The natural history of subclinical hypothyroidism in the elderly: the cardiovascular health study. J. Clin. Endocrinol. Metab. 97, 1962–1969 (2012).
Huber, G. et al. Prospective study of the spontaneous course of subclinical hypothyroidism: prognostic value of thyrotropin, thyroid reserve, and thyroid antibodies. J. Clin. Endocrinol. Metab. 87, 3221–3226 (2002).
Somwaru, L. L., Arnold, A. M., Joshi, N., Fried, L. P. & Cappola, A. R. High frequency of and factors associated with thyroid hormone over-replacement and under-replacement in men and women aged 65 and over. J. Clin. Endocrinol. Metab. 94, 1342–1345 (2009).
Parle, J. V., Franklyn, J. A., Cross, K. W., Jones, S. R. & Sheppard, M. C. Thyroxine prescription in the community: serum thyroid stimulating hormone level assays as an indicator of undertreatment or overtreatment. Br. J. Gen. Pract. 43, 107–109 (1993).
Franklyn, J. A. & Boelaert, K. Thyrotoxicosis. Lancet 379, 1155–1166 (2012).
Abraham-Nordling, M. et al. Incidence of hyperthyroidism in Stockholm, Sweden, 2003–2005. Eur. J. Endocrinol. 158, 823–827 (2008).
Nyirenda, M. J. et al. Thyroid disease and increased cardiovascular risk. Thyroid 15, 718–724 (2005).
Cohen-Lehman, J., Dahl, P., Danzi, S. & Klein, I. Effects of amiodarone therapy on thyroid function. Nat. Rev. Endocrinol. 6, 34–41 (2010).
Basaria, S. & Cooper, D. S. Amiodarone and the thyroid. Am. J. Med. 118, 706–714 (2005).
Yiu, K. H. et al. Amiodarone-induced thyrotoxicosis is a predictor of adverse cardiovascular outcome. J. Clin. Endocrinol. Metab. 94, 109–114 (2009).
O'Sullivan, A. J., Lewis, M., Diamond, T. Amiodarone-induced thyrotoxicosis: left ventricular dysfunction is associated with increased mortality. Eur. J. Endocrinol. 154, 533–536 (2006).
Meyerovitch, J. et al. Serum thyrotropin measurements in the community: five-year follow-up in a large network of primary care physicians. Arch. Intern. Med. 167, 1533–1538 (2007).
Lindeman, R. D. et al. Subclinical hypothyroidism in a biethnic, urban community. J. Am. Geriatr. Soc. 47, 703–709 (1999).
Almeida, O. P. et al. Thyroid hormones and depression: the Health in Men study. Am. J. Geriatr. Psychiatry 19, 763–770 (2011).
Parle, J. et al. A randomized controlled trial of the effect of thyroxine replacement on cognitive function in community-living elderly individuals with subclinical hypothyroidism: the Birmingham Elderly Thyroid study. J. Clin. Endocrinol. Metab. 95, 3623–3632 (2010).
Cappola, A. R. & Ladenson, P. W. Hypothyroidism and atherosclerosis. J. Clin. Endocrinol. Metab. 88, 2438–2444 (2003).
Danese, M. D., Ladenson, P. W., Meinert, C. L. & Powe, N. R. Clinical review 115: effect of thyroxine therapy on serum lipoproteins in patients with mild thyroid failure: a quantitative review of the literature. J. Clin. Endocrinol. Metab. 85, 2993–3001 (2000).
Tognini, S. et al. Age and gender substantially influence the relationship between thyroid status and the lipoprotein profile: results from a large cross-sectional study. Thyroid 22, 1096–1103 (2012).
Liu, D. et al. A cross-sectional survey of relationship between serum TSH level and blood pressure. J. Hum. Hypertens. 24, 134–138 (2010).
Maratou, E. et al. Studies of insulin resistance in patients with clinical and subclinical hypothyroidism. Eur. J. Endocrinol. 160, 785–790 (2009).
Knudsen, N. et al. Small differences in thyroid function may be important for body mass index and the occurrence of obesity in the population. J. Clin. Endocrinol. Metab. 90, 4019–4024 (2005).
Owen, P. J., Sabit, R. & Lazarus, J. H. Thyroid disease and vascular function. Thyroid 17, 519–524 (2007).
Rodondi, N. et al. Subclinical thyroid dysfunction, cardiac function, and the risk of heart failure. The Cardiovascular Health study. J. Am. Coll. Cardiol. 52, 1152–1159 (2008).
Rodondi, N. et al. Subclinical hypothyroidism and the risk of heart failure, other cardiovascular events, and death. Arch. Intern. Med. 165, 2460–2466 (2005).
Nanchen, D. et al. Subclinical thyroid dysfunction and the risk of heart failure in older persons at high cardiovascular risk. J. Clin. Endocrinol. Metab. 97, 852–861 (2012).
Gussekloo, J. et al. Thyroid status, disability and cognitive function, and survival in old age. JAMA 292, 2591–2599 (2004).
Hyland, K. A., Arnold, A. M., Lee, J. S. & Cappola, A. R. Persistent subclinical hypothyroidism and cardiovascular risk in the elderly: the Cardiovascular Health Study. J. Clin. Endocrinol. Metab. http://dx.doi.org/10.1210/jc.2012-2180.
McQuade, C. et al. Hypothyroidism and moderate subclinical hypothyroidism are associated with increased all-cause mortality independent of coronary heart disease risk factors: a PreCIS database study. Thyroid 21, 837–843 (2011).
Razvi, S., Weaver, J. U., Vanderpump, M. P. & Pearce, S. H. The incidence of ischemic heart disease and mortality in people with subclinical hypothyroidism: reanalysis of the Whickham Survey cohort. J. Clin. Endocrinol. Metab. 95, 1734–1740 (2010).
Boekholdt, S. M. et al. Initial thyroid status and cardiovascular risk factors: the EPIC-Norfolk prospective population study. Clin. Endocrinol. (Oxf.) 72, 404–410 (2010).
Rodondi, N. et al. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA 304, 1365–1374 (2010).
Osman, F., Franklyn, J. A., Holder, R. L., Sheppard, M. C. & Gammage, M. D. Cardiovascular manifestations of hyperthyroidism before and after antithyroid therapy: a matched case–control study. J. Am. Coll. Cardiol. 49, 71–81 (2007).
Frost, L., Vestergaard, P. & Mosekilde, L. Hyperthyroidism and risk of atrial fibrillation or flutter: a population-based study. Arch. Intern. Med. 164, 1675–1678 (2004).
Sawin, C. T. et al. Low serum thyrotropin concentrations as a risk factor for atrial fibrillation in older persons. N. Engl. J. Med. 331, 1249–1252 (1994).
Gammage, M. D. et al. Association between serum free thyroxine concentration and atrial fibrillation. Arch. Intern. Med. 167, 928–934 (2007).
Klein, I. & Danzi, S. Thyroid disease and the heart. Circulation 116, 1725–1735 (2007).
Siu, C. W., Yeung, C. Y., Lau, C. P., Kung, A. W. & Tse, H. F. Incidence, clinical characteristics and outcome of congestive heart failure as the initial presentation in patients with primary hyperthyroidism. Heart 93, 483–487 (2007).
Franklyn, J. A., Maisonneuve, P., Sheppard, M. C., Betteridge, J. & Boyle, P. Mortality after the treatment of hyperthyroidism with radioactive iodine. N. Engl. J. Med. 338, 712–718 (1998).
Franklyn, J. A., Sheppard, M. C. & Maisonneuve, P. Thyroid function and mortality in patients treated for hyperthyroidism. JAMA 294, 71–80 (2005).
Metso, S. et al. Increased cardiovascular and cancer mortality after radioiodine treatment for hyperthyroidism. J. Clin. Endocrinol. Metab. 92, 2190–2196 (2007).
Bauer, D. C., Rodondi, N., Stone, K. L. & Hillier, T. A. Thyroid hormone use, hyperthyroidism and mortality in older women. Am. J. Med. 120, 343–349 (2007).
Parle, J. V., Maisonneuve, P., Sheppard, M. C., Boyle, P. & Franklyn, J. A. Prediction of all-cause and cardiovascular mortality in elderly people from one low serum thyrotropin result: a 10-year cohort study. Lancet 358, 861–865 (2001).
Singh, S. et al. Impact of subclinical thyroid disorders on coronary heart disease, cardiovascular and all-cause mortality: a meta-analysis. Int. J. Cardiol. 125, 41–48 (2008).
Volzke, H., Schwahn, C., Wallaschofski, H. & Dorr, M. Review: The association of thyroid dysfunction with all-cause and circulatory mortality: is there a causal relationship? J. Clin. Endocrinol. Metab. 92, 2421–2429 (2007).
Ochs, N. et al. Meta-analysis: subclinical thyroid dysfunction and the risk for coronary heart disease and mortality. Ann. Intern. Med. 148, 832–845 (2008).
Haentjens, P., Van, M. A., Poppe, K. & Velkeniers, B. Subclinical thyroid dysfunction and mortality: an estimate of relative and absolute excess all-cause mortality based on time-to-event data from cohort studies. Eur. J. Endocrinol. 159, 329–341 (2008).
Yang, L. B. et al. Subclinical hyperthyroidism and the risk of cardiovascular events and all-cause mortality: an updated meta-analysis of cohort studies. Eur. J. Endocrinol. 167, 75–84 (2012).
Collet, T. H. et al. Subclinical hyperthyroidism and the risk of coronary heart disease and mortality. Arch. Intern. Med. 172, 799–809 (2012).
Vestergaard, P. & Mosekilde, L. Hyperthyroidism, bone mineral, and fracture risk--a meta-analysis. Thyroid 13, 585–593 (2003).
Foldes, J. et al. Bone mineral density in patients with endogenous subclinical hyperthyroidism: is this thyroid status a risk factor for osteoporosis? Clin. Endocrinol. (Oxf.) 39, 521–527 (1993).
Mudde, A. H., Houben, A. J. & Nieuwenhuijzen Kruseman, A. C. Bone metabolism during anti-thyroid drug treatment of endogenous subclinical hyperthyroidism. Clin. Endocrinol. (Oxf.) 41, 421–424 (1994).
Faber, J. & Galloe, A. M. Changes in bone mass during prolonged subclinical hyperthyroidism due to L-thyroxine treatment: a meta-analysis. Eur. J. Endocrinol. 130, 350–356 (1994).
Uzzan, B. et al. Effects on bone mass of long term treatment with thyroid hormones: a meta-analysis. J. Clin. Endocrinol. Metab. 81, 4278–4289 (1996).
Bauer, D. C., Ettinger, B., Nevitt, M. C. & Stone, K. L. Risk for fracture in women with low serum levels of thyroid-stimulating hormone. Ann. Intern. Med. 134, 561–568 (2001).
Lee, J. S. et al. Subclinical thyroid dysfunction and incident hip fracture in older adults. Arch. Intern. Med. 170, 1876–1883 (2010).
Murphy, E. et al. Thyroid function within the upper normal range is associated with reduced bone mineral density and an increased risk of nonvertebral fractures in healthy euthyroid postmenopausal women. J. Clin. Endocrinol. Metab. 95, 3173–3181 (2010).
van der Deure, W. M. et al. Effects of serum TSH and FT4 levels and the TSHR-Asp727Glu polymorphism on bone: the Rotterdam Study. Clin. Endocrinol. (Oxf.) 68, 175–181 (2008).
Lin, J. D. et al. The relationship between thyroid function and bone mineral density in euthyroid healthy individuals in Taiwan. Endocr. Res. 36, 1–8 (2011).
Yeap, B. B. et al. Higher free thyroxine levels are associated with frailty in older men: the Health In Men Study. Clin. Endocrinol. (Oxf.) 76, 741–748 (2012).
Ceresini, G. et al. Mild thyroid hormone excess is associated with a decreased physical function in elderly men. Aging Male 14, 213–219 (2011).
Yeap, B. B. et al. Higher free thyroxine levels predict increased incidence of dementia in older men: the health in men study. J. Clin. Endocrinol. Metab. 97, E2230–E2237 (2012).
Meier, C. et al. TSH-controlled L-thyroxine therapy reduces cholesterol levels and clinical symptoms in subclinical hypothyroidism: a double blind, placebo-controlled trial (Basel Thyroid Study). J. Clin. Endocrinol. Metab. 86, 4860–4866 (2001).
Monzani, F. et al. Effect of levothyroxine replacement on lipid profile and intima-media thickness in subclinical hypothyroidism: a double-blind, placebo- controlled study. J. Clin. Endocrinol. Metab. 89, 2099–2106 (2004).
Razvi, S. et al. The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J. Clin. Endocrinol. Metab. 92, 1715–1723 (2007).
Bahn Chair, R. S. et al. Hyperthyroidism and other causes of thyrotoxicosis: management guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Thyroid 21, 593–646 (2011).
Díez, J. J. Hyperthyroidism in patients older than 55 years: an analysis of the etiology and management. Gerontology 49, 316–323 (2003).
Takáts, K. I. et al. The efficacy of long term thyrostatic treatment in elderly patients with toxic nodular goitre compared to radioiodine therapy with different doses. Exp. Clin. Endocrinol. Diabetes 107, 70–74 (1999).
Chen, D. Y., Jing, J., Schneider, P. F. & Chen, T. H. Comparison of the long-term efficacy of low dose 131I versus antithyroid drugs in the treatment of hyperthyroidism. Nucl. Med. Commun. 30, 160–168 (2009).
Allahabadia, A. et al. Age and gender predict the outcome of treatment for Graves' hyperthyroidism. J. Clin. Endocrinol. Metab. 85, 1038–1042 (2000).
Auer, J. et al. Subclinical hyperthyroidism as a risk factor for atrial fibrillation. Am. Heart J. 142, 838–842 (2001).
Faber, J. et al. Normalization of serum thyrotrophin by means of radioiodine treatment in subclinical hyperthyroidism: effect on bone loss in postmenopausal women. Clin. Endocrinol. (Oxf.) 48, 285–290 (1998).
Sgarbi, J. A., Villaca, F. G., Garbeline, B., Villar, H. E. & Romaldini, J. H. The effects of early antithyroid therapy for endogenous subclinical hyperthyroidism in clinical and heart abnormalities. J. Clin. Endocrinol. Metab. 88, 1672–1677 (2003).
Santini, F. et al. Lean body mass is a major determinant of levothyroxine dosage in the treatment of thyroid diseases. J. Clin. Endocrinol. Metab. 90, 124–127 (2005).
Rosenbaum, R. L. & Barzel, U. S. Levothyroxine replacement dose for primary hypothyroidism decreases with age. Ann. Intern. Med. 96, 53–55 (1982).
Sawin, C. T., Herman, T., Molitch, M. E., London, M. H. & Kramer, S. M. Aging and the thyroid. Decreased requirement for thyroid hormone in older hypothyroid patients. Am. J. Med. 75, 206–209 (1983).
Devdhar, M., Drooger, R., Pehlivanova, M., Singh, G. & Jonklaas, J. Levothyroxine replacement doses are affected by gender and weight, but not age. Thyroid 21, 821–827 (2011).
Roos, A., Linn-Rasker, S. P., van Domburg, R. T., Tijssen, J. P. & Berghout, A. The starting dose of levothyroxine in primary hypothyroidism treatment: a prospective, randomized, double-blind trial. Arch. Intern. Med. 165, 1714–1720 (2005).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Boelaert, K. Thyroid dysfunction in the elderly. Nat Rev Endocrinol 9, 194–204 (2013). https://doi.org/10.1038/nrendo.2013.30
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2013.30
This article is cited by
-
Low levels of free triiodothyronine are associated with risk of cognitive impairment in older euthyroid adults
Scientific Reports (2023)
-
A systematic review and meta-analysis investigating the relationship between metabolic syndrome and the incidence of thyroid diseases
Endocrine (2023)
-
Impatto dell’utilizzo di valori di riferimento età e sesso-specifici per TSH e FT4 sulla diagnosi di disfunzione tiroidea subclinica: risultati di uno studio multicentrico in Giappone
L'Endocrinologo (2023)
-
Low T3 syndrome as a predictor of poor outcomes in patients with follicular lymphoma
Annals of Hematology (2023)
-
Clinical profile of 80-year-old and older thyroid eye disease patients
Graefe's Archive for Clinical and Experimental Ophthalmology (2022)