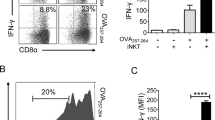
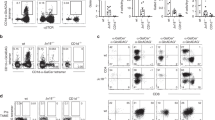
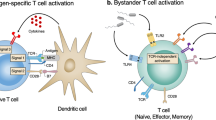
Key Points
-
The principles that underlie the interaction of the type I natural killer T (NKT) cell T cell receptor (TCR) with CD1d are distinct from those that govern the interaction of the TCR of conventional T cells with their peptide–MHC ligands.
-
Type I NKT TCRs consistently adopt a parallel docking mode over the F′-pocket of CD1d that is independent of TCR chain usage or of the antigen that is presented on the CD1d molecule. This indicates that the NKT TCR acts in a manner akin to a pattern-recognition receptor.
-
Within the consensus type I NKT TCR–antigen–CD1d footprint, variations in TCR α- and β-chain usage can determine the levels of CD1d-mediated autoreactivity and the degree of antigen specificity.
-
The type I NKT cell TCR can 'bulldoze' distinct ligands, including β-linked self glycolipid ligands, such that they mimic the conformation of the α-linked antigens, thereby enabling the consensus type I NKT TCR–antigen–CD1d footprint.
-
The affinity of the type I NKT TCR–antigen–CD1d interaction is a good measure of the efficacy of an antigen at stimulating NKT cells, whereas the T helper 1 (TH1)- and TH2-biasing properties of an antigen seem to be more attributable to CD1d loading considerations and the features of the antigen itself.
-
The type II NKT TCR has been shown to dock above the A′-pocket of CD1d. Thus, type II NKT TCRs can interact with antigen–CD1d complexes in a markedly different manner from type I NKT TCRs.
Abstract
Natural killer T (NKT) cells are innate-like T cells that rapidly produce a variety of cytokines following T cell receptor (TCR) activation and can shape the immune response in many different settings. There are two main NKT cell subsets: type I NKT cells are typically characterized by the expression of a semi-invariant TCR, whereas the TCRs expressed by type II NKT cells are more diverse. This Review focuses on the defining features and emerging generalities regarding how NKT cells specifically recognize self, microbial and synthetic lipid-based antigens that are presented by CD1d. Such information is vitally important to better understand, and fully harness, the therapeutic potential of NKT cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Brigl, M. & Brenner, M. B. CD1: antigen presentation and T cell function. Annu. Rev. Immunol. 22, 817–890 (2004).
Mori, L. & De Libero, G. T cells specific for lipid antigens. Immunol. Res. 53, 191–199 (2012).
Bendelac, A., Savage, P. B. & Teyton, L. The biology of NKT cells. Annu. Rev. Immunol. 25, 297–336 (2007).
Godfrey, D. I., MacDonald, H. R., Kronenberg, M., Smyth, M. J. & Van Kaer, L. NKT cells: what's in a name? Nature Rev. Immunol. 4, 231–237 (2004).
Godfrey, D. I. & Kronenberg, M. Going both ways: immune regulation via CD1d-dependent NKT cells. J. Clin. Invest. 114, 1379–1388 (2004).
Matsuda, J. L., Mallevaey, T., Scott-Browne, J. & Gapin, L. CD1d-restricted iNKT cells, the 'Swiss-Army knife' of the immune system. Curr. Opin. Immunol. 20, 358–368 (2008).
Berzins, S. P., Smyth, M. J. & Baxter, A. G. Presumed guilty: natural killer T cell defects and human disease. Nature Rev. Immunol. 11, 131–142 (2011).
Cerundolo, V., Barral, P. & Batista, F. D. Synthetic iNKT cell-agonists as vaccine adjuvants — finding the balance. Curr. Opin. Immunol. 22, 417–424 (2010).
Cerundolo, V., Silk, J. D., Masri, S. H. & Salio, M. Harnessing invariant NKT cells in vaccination strategies. Nature Rev. Immunol. 9, 28–38 (2009).
Godfrey, D. I. et al. Antigen recognition by CD1d-restricted NKT T cell receptors. Semin. Immunol. 22, 61–67 (2010).
Godfrey, D. I. & Rossjohn, J. New ways to turn on NKT cells. J. Exp. Med. 208, 1121–1125 (2011).
Wingender, G. et al. Invariant NKT cells are required for airway inflammation induced by environmental antigens. J. Exp. Med. 208, 1151–1162 (2011).
Venkataswamy, M. M. & Porcelli, S. A. Lipid and glycolipid antigens of CD1d-restricted natural killer T cells. Semin. Immunol. 22, 68–78 (2010).
Dellabona, P., Padovan, E., Casorati, G., Brockhaus, M. & Lanzavecchia, A. An invariant Vα24–JαQ/Vβ11 T cell receptor is expressed in all individuals by clonally expanded CD4−8− T cells. J. Exp. Med. 180, 1171–1176 (1994).
Porcelli, S., Yockey, C. E., Brenner, M. B. & Balk, S. P. Analysis of T cell antigen receptor (TCR) expression by human peripheral blood CD4−8− α/β T cells demonstrates preferential use of several Vβ genes and an invariant TCR α chain. J. Exp. Med. 178, 1–16 (1993).
Lantz, O. & Bendelac, A. An invariant T cell receptor α chain is used by a unique subset of major histocompatibility complex class I-specific CD4+ and CD4−8− T cells in mice and humans. J. Exp. Med. 180, 1097–1106 (1994).
Godfrey, D. I., McCluskey, J. & Rossjohn, J. CD1d antigen presentation: treats for NKT cells. Nature Immunol. 6, 754–756 (2005).
Kjer-Nielsen, L. et al. A structural basis for selection and cross-species reactivity of the semi-invariant NKT cell receptor in CD1d/glycolipid recognition. J. Exp. Med. 203, 661–673 (2006).
Brossay, L. et al. Cd1d-mediated recognition of an α-galactosylceramide by natural killer T cells is highly conserved through mammalian evolution. J. Exp. Med. 188, 1521–1528 (1998).
Cui, J. Q. et al. Requirement for Vα14 NKT cells in IL-12-mediated rejection of tumors. Science 278, 1623–1626 (1997).
Bedel, R. et al. Lower TCR repertoire diversity in Traj18-deficient mice. Nature Immunol. 13, 705–706 (2012).
Matsuda, J. L. et al. Natural killer T cells reactive to a single glycolipid exhibit a highly diverse T cell receptor β repertoire and small clone size. Proc. Natl Acad. Sci. USA 98, 12636–12641 (2001).
Gapin, L. iNKT cell autoreactivity: what is 'self' and how is it recognized? Nature Rev. Immunol. 10, 272–277 (2010).
Kawano, T. et al. CD1d-restricted and TCR-mediated activation of Vα14 NKT cells by glycosylceramides. Science 278, 1626–1629 (1997). This study characterizes α -galactosylceramide as a ligand for type I NKT cells.
Burdin, N. et al. Structural requirements for antigen presentation by mouse CD1. Proc. Natl Acad. Sci. USA 97, 10156–10161 (2000).
Burdin, N. et al. Selective ability of mouse CD1 to present glycolipids: α-galactosylceramide specifically stimulates Vα14+ NK T lymphocytes. J. Immunol. 161, 3271–3281 (1998).
Rhost, S., Sedimbi, S., Kadri, N. & Cardell, S. L. Immunomodulatory type II natural killer T (NKT) lymphocytes in health and disease. Scand. J. Immunol. 76, 246–255 (2012).
Jahng, A. et al. Prevention of autoimmunity by targeting a distinct, noninvariant CD1d-reactive T cell population reactive to sulfatide. J. Exp. Med. 199, 947–957 (2004).
Cardell, S. et al. CD1-restricted CD4+ T cells in major histocompatibility complex class II-deficient mice. J. Exp. Med. 182, 993–1004 (1995).
Arrenberg, P., Halder, R., Dai, Y., Maricic, I. & Kumar, V. Oligoclonality and innate-like features in the TCR repertoire of type II NKT cells reactive to a β-linked self-glycolipid. Proc. Natl Acad. Sci. USA 107, 10984–10989 (2010).
Park, S. H. et al. The mouse CD1d-restricted repertoire is dominated by a few autoreactive T cell receptor families. J. Exp. Med. 193, 893–904 (2001).
Bai, L. et al. The majority of CD1d–sulfatide-specific T cells in human blood use a semiinvariant Vδ1 TCR. Eur. J. Immunol. 42, 2505–2510 (2012).
Gadola, S. D., Dulphy, N., Salio, M. & Cerundolo, V. Vα24-JαQ-independent, CD1d-restricted recognition of α-galactosylceramide by human CD4+ and CD8αβ+ T lymphocytes. J. Immunol. 168, 5514–5520 (2002).
Gadola, S. D. et al. Structure and binding kinetics of three different human CD1d–α-galactosylceramide-specific T cell receptors. J. Exp. Med. 203, 699–710 (2006).
Brigl, M. et al. Conserved and heterogeneous lipid antigen specificities of CD1d-restricted NKT cell receptors. J. Immunol. 176, 3625–3634 (2006).
Constantinides, M. G., Picard, D., Savage, A. K. & Bendelac, A. A. Naive-like population of human CD1d-restricted T cells expressing intermediate levels of promyelocytic leukemia zinc finger. J. Immunol. 187, 309–315 (2011).
Uldrich, A. P. et al. A semi-invariant Vα10+ T cell antigen receptor defines a population of natural killer T cells with distinct glycolipid antigen-recognition properties. Nature Immunol. 12, 616–623 (2011).
Brennan, P. J. et al. Invariant natural killer T cells recognize lipid self antigen induced by microbial danger signals. Nature Immunol. 12, 1202–1211 (2011). This study shows that a particular form of β GlcCer represents a self ligand for the NKT TCR that is upregulated during infection.
Rhost, S. et al. Identification of novel glycolipid ligands activating a sulfatide reactive, CD1d-restricted, type II natural killer T lymphocyte. Eur. J. Immunol. 10 Jul 2012 (doi:10.1002/eji.201142350).
Zeng, Z. et al. Crystal structure of mouse CD1: an MHC-like fold with a large hydrophobic binding groove. Science 277, 339–345 (1997).
Gadola, S. D. et al. Structure of human CD1b with bound ligands at 2.3 Å, a maze for alkyl chains. Nature Immunol. 3, 721–726 (2002).
Zajonc, D. M. et al. Structure and function of a potent agonist for the semi-invariant natural killer T cell receptor. Nature Immunol. 6, 810–818 (2005).
Zajonc, D. M., Elsliger, M. A., Teyton, L. & Wilson, I. A. Crystal structure of CD1a in complex with a sulfatide self antigen at a resolution of 2.15 Å. Nature Immunol. 4, 808–815 (2003).
Scharf, L. et al. The 2.5Å structure of CD1c in complex with a mycobacterial lipid reveals an open groove ideally suited for diverse antigen presentation. Immunity 33, 853–862 (2010).
Koch, M. et al. The crystal structure of human CD1d with and without α-galactosylceramide. Nature Immunol. 6, 819–826 (2005).
Moody, D. B., Zajonc, D. M. & Wilson, I. A. Anatomy of CD1–lipid antigen complexes. Nature Rev. Immunol. 5, 387–399 (2005).
Zajonc, D. M. & Wilson, I. A. Architecture of CD1 proteins. Curr. Top. Microbiol. Immunol. 314, 27–50 (2007).
Salio, M., Silk, J. D. & Cerundolo, V. Recent advances in processing and presentation of CD1 bound lipid antigens. Curr. Opin. Immunol. 22, 81–88 (2010).
Cohen, N. R., Garg, S. & Brenner, M. B. Antigen presentation by CD1: lipids, T cells, and NKT cells in microbial immunity. Adv. Immunol. 102, 1–94 (2009).
Brutkiewicz, R. R. CD1d ligands: the good, the bad, and the ugly. J. Immunol. 177, 769–775 (2006).
Borg, N. A., Kjer-Nielsen, L., McCluskey, J. & Rossjohn, J. Structural insight into natural killer T cell receptor recognition of CD1d. Adv. Exp. Med. Biol. 598, 20–34 (2007).
Borg, N. A. et al. CD1d–lipid-antigen recognition by the semi-invariant NKT T-cell receptor. Nature 448, 44–49 (2007). This study provides the first description of a type I NKT TCR–antigen–CD1d interaction.
Godfrey, D. I., Rossjohn, J. & McCluskey, J. The fidelity, occasional promiscuity, and versatility of T cell receptor recognition. Immunity 28, 304–314 (2008).
Burrows, S. R. et al. Hard wiring of T cell receptor specificity for the major histocompatibility complex is underpinned by TCR adaptability. Proc. Natl Acad. Sci. USA 107, 10608–10613 (2010).
Pellicci, D. G. et al. Differential recognition of CD1d–α-galactosyl ceramide by the Vβ8.2 and Vβ7 semi-invariant NKT T cell receptors. Immunity 31, 47–59 (2009).
Li, Y. et al. The Va14 invariant natural killer T cell TCR forces microbial glycolipids and CD1d into a conserved binding mode. J. Exp. Med. 207, 2383–2393 (2010). This report provided the first insights into how the type I NKT TCR can interact with microbial ligands.
Girardi, E. et al. Unique interplay between sugar and lipid in determining the antigenic potency of bacterial antigens for NKT cells. PLoS Biol. 9, e1001189 (2011).
Wun, K. S. et al. A molecular basis for the exquisite CD1d-restricted antigen specificity and functional responses of natural killer T cells. Immunity 34, 327–339 (2011).
Scott-Browne, J. P. et al. Germline-encoded recognition of diverse glycolipids by natural killer T cells. Nature Immunol. 8, 1105–1113 (2007). This study showed for the first time how the type I NKT TCR can function like a pattern-recognition receptor.
Wun, K. S. et al. A minimal binding footprint on CD1d–glycolipid is a basis for selection of the unique human NKT TCR. J. Exp. Med. 205, 939–949 (2008).
Florence, W. C. et al. Adaptability of the semi-invariant natural killer T-cell receptor towards structurally diverse CD1d-restricted ligands. EMBO J. 28, 3579–3590 (2009).
Patel, O. et al. Vb2 natural killer T cell antigen receptor-mediated recognition of CD1d–glycolipid antigen. Proc. Natl Acad. Sci. USA 108, 19007–19012 (2011).
Joyce, S., Girardi, E. & Zajonc, D. M. NKT cell ligand recognition logic: molecular basis for a synaptic duet and transmission of inflammatory effectors. J. Immunol. 187, 1081–1089 (2011).
Mallevaey, T. & Selvanantham, T. Strategy of lipid recognition by invariant natural killer T cells: 'one for all and all for one'. Immunology 136, 273–282 (2012).
Adams, E. J. & Lopez-Sagaseta, J. The immutable recognition of CD1d. Immunity 34, 281–283 (2011).
Mallevaey, T. et al. T cell receptor CDR2β and CDR3β loops collaborate functionally to shape the iNKT cell repertoire. Immunity 31, 60–71 (2009).
Pellicci, D. G. et al. Recognition of β-linked self glycolipids mediated by natural killer T cell antigen receptors. Nature Immunol. 12, 827–833 (2011).
Mallevaey, T. et al. A molecular basis for NKT cell recognition of CD1d–self-antigen. Immunity 34, 315–326 (2011). This report provided the first structural insights into the basis of CD1d-mediated autoreactivity.
Yu, E. D., Girardi, E., Wang, J. & Zajonc, D. M. Cutting edge: structural basis for the recognition of β-linked glycolipid antigens by invariant NKT cells. J. Immunol. 187, 2079–2083 (2011). Together with reference 67, this study showed that the type I NKT TCR can 'bulldoze' β -linked ligands to enable CD1d engagement.
Lopez-Sagaseta, J., Sibener, L. V., Kung, J. E., Gumperz, J. & Adams, E. J. Lysophospholipid presentation by CD1d and recognition by a human natural killer T-cell receptor. EMBO J. 31, 2047–2059 (2012). This is the first description of how an NKT TCR can recognize lysophospholipids.
Miyamoto, K., Miyake, S. & Yamamura, T. A synthetic glycolipid prevents autoimmune encephalomyelitis by inducing TH2 bias of natural killer T cells. Nature 413, 531–534 (2001).
Schmieg, J., Yang, G., Franck, R. W. & Tsuji, M. Superior protection against malaria and melanoma metastases by a C-glycoside analogue of the natural killer T cell ligand α-galactosylceramide. J. Exp. Med. 198, 1631–1641 (2003).
Aspeslagh, S. et al. Galactose-modified iNKT cell agonists stabilized by an induced fit of CD1d prevent tumour metastasis. EMBO J. 30, 2294–2305 (2011).
Yu, K. O. et al. Modulation of CD1d-restricted NKT cell responses by using N-acyl variants of α-galactosylceramides. Proc. Natl Acad. Sci. USA 102, 3383–3388 (2005).
Patel, O. et al. NKT TCR recognition of CD1d–α-C-galactosylceramide. J. Immunol. 187, 4705–4713 (2011).
Kerzerho, J. et al. Structural and functional characterization of a novel nonglycosidic type I NKT agonist with immunomodulatory properties. J. Immunol. 188, 2254–2265 (2012).
Tyznik, A. J. et al. Glycolipids that elicit IFN-γ-biased responses from natural killer T cells. Chem. Biol. 18, 1620–1630 (2011).
Sloan-Lancaster, J. & Allen, P. M. Altered peptide ligand-induced partial T cell activation: molecular mechanisms and role in T cell biology. Annu. Rev. Immunol. 14, 1–27 (1996).
Raju, R. et al. Synthesis and evaluation of 3′′- and 4′′-deoxy and -fluoro analogs of the immunostimulatory glycolipid, KRN7000. Bioorg. Med. Chem. Lett. 19, 4122–4125 (2009).
Wun, K. S. et al. Human and mouse type I natural killer T-cell antigen receptors exhibit different fine specificities for CD1d–antigen. J. Biol. Chem. 20 Sep 2012 (doi:10.1074/jbc.M112.412320).
Sullivan, B. A. et al. Mechanisms for glycolipid antigen-driven cytokine polarization by Vα14i NKT cells. J. Immunol. 184, 141–153 (2010).
McCarthy, C. et al. The length of lipids bound to human CD1d molecules modulates the affinity of NKT cell TCR and the threshold of NKT cell activation. J. Exp. Med. 204, 1131–1144 (2007).
Im, J. S. et al. Kinetics and cellular site of glycolipid loading control the outcome of natural killer T cell activation. Immunity 30, 888–898 (2009). This study shows how mechanisms pertaining to antigen loading can relate to the efficacy of an antigen in activating NKT cells.
Bai, L. et al. Lysosomal recycling terminates CD1d-mediated presentation of short and polyunsaturated variants of the NKT cell lipid antigen αGalCer. Proc. Natl Acad. Sci. USA 106, 10254–10259 (2009).
Bai, L. et al. Distinct APCs explain the cytokine bias of α-galactosylceramide variants in vivo. J. Immunol. 188, 3053–3061 (2012).
Mattner, J. et al. Exogenous and endogenous glycolipid antigens activate NKT cells during microbial infections. Nature 434, 525–529 (2005).
Sriram, V., Du, W., Gervay-Hague, J. & Brutkiewicz, R. R. Cell wall glycosphingolipids of Sphingomonas paucimobilis are CD1d-specific ligands for NKT cells. Eur. J. Immunol. 35, 1692–1701 (2005).
Kinjo, Y. et al. Invariant natural killer T cells recognize glycolipids from pathogenic Gram-positive bacteria. Nature Immunol. 12, 966–974 (2011).
Kinjo, Y. et al. Natural killer T cells recognize diacylglycerol antigens from pathogenic bacteria. Nature Immunol. 7, 978–986 (2006).
Kinjo, Y. et al. Recognition of bacterial glycosphingolipids by natural killer T cells. Nature 434, 520–525 (2005). This report describes the importance of NKT cells in protective immunity to infection.
Fischer, K. et al. Mycobacterial phosphatidylinositol mannoside is a natural antigen for CD1d-restricted T cells. Proc. Natl Acad. Sci. USA 101, 10685–10690 (2004).
Chang, Y.-J. et al. Influenza infection in suckling mice expands an NKT cell subset that protects against airway hyperreactivity. J. Clin. Invest. 121, 57–69 (2011).
Wu, D. et al. Design of natural killer T cell activators: structure and function of a microbial glycosphingolipid bound to mouse CD1d. Proc. Natl Acad. Sci. USA 103, 3972–3977 (2006).
Wang, J. et al. Lipid binding orientation within CD1d affects recognition of Borrelia burgorferi antigens by NKT cells. Proc. Natl Acad. Sci. USA 107, 1535–1540 (2010).
Zeissig, S. et al. Hepatitis B virus-induced lipid alterations contribute to natural killer T cell-dependent protective immunity. Nature Med. 18, 1060–1068 (2012). This report describes a role for phospholipid-reactive type II NKT cells in promoting immunity to hepatitis B virus.
Brigl, M. et al. Innate and cytokine-driven signals, rather than microbial antigens, dominate in natural killer T cell activation during microbial infection. J. Exp. Med. 208, 1163–1177 (2011).
Chiu, Y. H. et al. Multiple defects in antigen presentation and T cell development by mice expressing cytoplasmic tail-truncated CD1d. Nature Immunol. 3, 55–60 (2002).
Hogquist, K. A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Zhou, D. et al. Lysosomal glycosphingolipid recognition by NKT cells. Science 306, 1786–1789 (2004).
Speak, A. O. et al. Implications for invariant natural killer T cell ligands due to the restricted presence of isoglobotrihexosylceramide in mammals. Proc. Natl Acad. Sci. USA 104, 5971–5976 (2007).
Porubsky, S. et al. Normal development and function of invariant natural killer T cells in mice with isoglobotrihexosylceramide (iGb3) deficiency. Proc. Natl Acad. Sci. USA 104, 5977–5982 (2007).
Christiansen, D. et al. Humans lack iGb3 due to the absence of functional iGb3-synthase: implications for NKT cell development and transplantation. PLoS Biol. 6, e172 (2008).
Porubsky, S. et al. Globosides but not isoglobosides can impact the development of invariant NKT cells and their interaction with dendritic cells. J. Immunol. 189, 3007–3017 (2012).
Facciotti, F. et al. Peroxisome-derived lipids are self antigens that stimulate invariant natural killer T cells in the thymus. Nature Immunol. 13, 474–480 (2012).
Zajonc, D. M., Ainge, G. D., Painter, G. F., Severn, W. B. & Wilson, I. A. Structural characterization of mycobacterial phosphatidylinositol mannoside binding to mouse CD1d. J. Immunol. 177, 4577–4583 (2006).
Zajonc, D. M., Savage, P. B., Bendelac, A., Wilson, I. A. & Teyton, L. Crystal structures of mouse CD1d–iGb3 complex and its cognate Vα14 T cell receptor suggest a model for dual recognition of foreign and self glycolipids. J. Mol. Biol. 377, 1104–1116 (2008).
Macdonald, W. A. et al. T cell allorecognition via molecular mimicry. Immunity 31, 897–908 (2009).
Archbold, J. K. et al. Natural micropolymorphism in human leukocyte antigens provides a basis for genetic control of antigen recognition. J. Exp. Med. 206, 209–219 (2009).
Tynan, F. E. et al. A T cell receptor flattens a bulged antigenic peptide presented by a major histocompatibility complex class I molecule. Nature Immunol. 8, 268–276 (2007).
Gumperz, J. E. et al. Murine CD1d-restricted T cell recognition of cellular lipids. Immunity 12, 211–221 (2000).
Matulis, G. et al. Innate-like control of human iNKT cell autoreactivity via the hypervariable CDR3β loop. PLoS Biol. 8, e1000402 (2010).
Muindi, K. et al. Activation state and intracellular trafficking contribute to the repertoire of endogenous glycosphingolipids presented by CD1d. Proc. Natl Acad. Sci. USA 107, 3052–3057 (2010).
Blomqvist, M. et al. Multiple tissue-specific isoforms of sulfatide activate CD1d-restricted type II NKT cells. Eur. J. Immunol. 39, 1726–1735 (2009).
Chang, D. H. et al. Inflammation associated lysophospholipids as ligands for CD1d restricted T cells in human cancer. Blood 112, 1308–1316 (2008).
Fox, L. M. et al. Recognition of lyso-phospholipids by human natural killer T lymphocytes. PLoS Biol. 7, e1000228 (2009).
Pei, B. et al. Diverse endogenous antigens for mouse NKT cells: self-antigens that are not glycosphingolipids. J. Immunol. 186, 1348–1360 (2011).
Berzofsky, J. A. & Terabe, M. NKT cells in tumor immunity: opposing subsets define a new immunoregulatory axis. J. Immunol. 180, 3627–3635 (2008).
Patel, O. et al. Recognition of CD1d–sulfatide mediated by a type II natural killer T cell antigen receptor. Nature Immunol. 13, 857–863 (2012).
Girardi, E. et al. Type II natural killer T cells use features of both innate-like and conventional T cells to recognize sulfatide self antigens. Nature Immunol. 13, 851–856 (2012). Together with reference 118, this study provided the first description of a type II NKT TCR–antigen–CD1d interaction.
Monzon-Casanova, E. et al. CD1d expression in Paneth cells and rat exocrine pancreas revealed by novel monoclonal antibodies which differentially affect NKT cell activation. PLoS ONE 5, e13089 (2010).
Krogsgaard, M. et al. Agonist/endogenous peptide–MHC heterodimers drive T cell activation and sensitivity. Nature 434, 238–243 (2005).
Agea, E. et al. Human CD1-restricted T cell recognition of lipids from pollens. J. Exp. Med. 202, 295–308 (2005).
Umetsu, D. T. & DeKruyff, R. H. Natural killer T cells are important in the pathogenesis of asthma: the many pathways to asthma. J. Allergy Clin. Immunol. 125, 975–979 (2010).
Braun, N. A., Covarrubias, R. & Major, A. S. Natural killer T cells and atherosclerosis: form and function meet pathogenesis. J. Innate Immun. 2, 316–324 (2010).
Kunii, N. et al. Combination therapy of in vitro-expanded natural killer T cells and α-galactosylceramide-pulsed antigen-presenting cells in patients with recurrent head and neck carcinoma. Cancer Sci. 100, 1092–1098 (2009).
Yamasaki, K. et al. Induction of NKT cell-specific immune responses in cancer tissues after NKT cell-targeted adoptive immunotherapy. Clin. Immunol. 138, 255–265 (2011).
Reantragoon, R. et al. Structural insight into MR1-mediated recognition of the mucosal associated invariant T cell receptor. J. Exp. Med. 209, 761–774 (2012).
Treiner, E. et al. Selection of evolutionarily conserved mucosal-associated invariant T cells by MR1. Nature 422, 164–169 (2003).
Hooper, L. V., Littman, D. R. & Macpherson, A. J. Interactions between the microbiota and the immune system. Science 336, 1268–1273 (2012).
López-Sagaseta, J., Kung, J. E., Savage, P. B., Gumperz, J. & Adams, E. J. The molecular basis for recognition of CD1d/α-galactosylceramide by a human non-Vα24 T cell receptor. PLoS Biol. 10, e1001412 (2012).
Kjer-Nielsen, L. et al. MR1 presents microbial vitamin B metabolites to MAIT cells. Nature 10 Oct 2012 (doi:10.1038/nature11605).
Acknowledgements
This work was supported by the US National Institutes of Health (grants AI090450 and AI092108), Australian National Health and Medical Research Council (NHMRC) program and project grants, Cancer Council Victoria and the Australian Research Council (ARC). D.I.G. was supported by an NHMRC Senior Principal Research Fellowship; J.R. was supported by an NHMRC Australia Fellowship. O.P. was supported by an ARC Future Fellowship.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- γδ T cells
-
T cells express either a T cell receptor (TCR) composed of α- and β-subunits (αβTCR) or a TCR composed of γ- and δ-subunits (γδTCR). The majority (more than 90%) of human T cells express αβ TCRs, which mainly recognize antigenic peptides bound to conventional MHC class I or II molecules. T cells that express γδTCRs are less abundant, and the ligands for these receptors are less well characterized.
- Altered peptide ligands
-
(APLs). Peptides that are analogues of an original antigenic peptide. They commonly have amino acid substitutions at residues that make contact with the T cell receptor (TCR). TCR engagement by these APLs usually leads to partial or incomplete T cell activation. Some APLs (antagonists) can specifically antagonize and inhibit T cell activation by the wild-type antigenic peptide.
- Peroxisome
-
An indispensable cytoplasmic organelle that has essential roles in antioxidant defence, cholesterol and bile-acid synthesis, eicosanoid metabolism and the β- and ω-oxidation of long-chain and very-long-chain fatty acids.
- Mucosa-associated invariant T cells
-
(MAIT cells). A population of innate-like lymphocytes. MAIT cells express an evolutionarily conserved invariant T cell receptor and are selected by the MHC class I-related molecule MR1. They are abundant in human blood, in the intestinal mucosa and in mesenteric lymph nodes, and they can produce interferon-γ in response to various bacterial infections.
Rights and permissions
About this article
Cite this article
Rossjohn, J., Pellicci, D., Patel, O. et al. Recognition of CD1d-restricted antigens by natural killer T cells. Nat Rev Immunol 12, 845–857 (2012). https://doi.org/10.1038/nri3328
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3328
This article is cited by
-
New immunotherapy approaches for colorectal cancer: focusing on CAR-T cell, BiTE, and oncolytic viruses
Cell Communication and Signaling (2024)
-
Markers and makers of NKT17 cells
Experimental & Molecular Medicine (2023)
-
CD1-mediated immune responses in mucosal tissues: molecular mechanisms underlying lipid antigen presentation system
Experimental & Molecular Medicine (2023)
-
Histone demethylases in the regulation of immunity and inflammation
Cell Death Discovery (2023)
-
The cytokine receptor DR3 identifies and promotes the activation of thymic NKT17 cells
Cellular and Molecular Life Sciences (2023)