Key Points
-
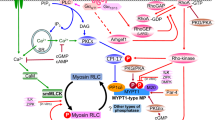
The regulation of intracellular Ca2+ is key to cardiac function. A large fraction of Ca2+ is released from the sarcoplasmic reticulum (SR) to initiate contraction and is pumped back into the SR to initiate relaxation by the sarco(endo)plasmic reticulum Ca2+-ATPase SERCA2a, the activity of which is regulated by phospholamban (PLN).
-
The inhibitory interaction between SERCA2a and PLN, as deduced from biochemical studies and structural modelling, involves sites of interaction that are located in the transmembrane and cytosolic domains of the two proteins. The interactions are disrupted by Ca2+ binding to SERCA2a, which brings about vast conformational changes that force PLN out of its transmembrane binding site, or by phosphorylation of PLN. Here the structural basis for disruption of the inhibited complex is less clear.
-
The ablation of PLN in mice prevents SERCA2a inhibition and enhances cardiac contractility by increasing the SR Ca2+ store. The ablation of PLN also reverses heart failure in some cardiomyopathic animal models, indicating the possibility of therapeutic approaches.
-
The overexpression of PLN in mouse heart depresses cardiac function and proves that only ∼40% of SERCA pumps are normally regulated by PLN in mouse heart.
-
The superinhibition of SERCA by specific PLN mutants impairs cardiac function and leads to cardiac remodelling and early death if the effects of the mutation cannot be reversed by β-agonists.
-
In human and animal models of heart failure, the PLN–SERCA inhibited complex increases. Interventions that diminish the PLN–SERCA complex have been beneficial in some mouse models of heart failure.
-
Specific human PLN mutations cause dilated cardiomyopathy: one mutation functions as a chronic SERCA2a inhibitor, whereas another destabilizes PLN, resulting in a PLN-null phenotype. So, for correct human cardiac function, there is a fine balance between excess inhibition of SERCA2a by PLN and no inhibition.
Abstract
Heart failure is a major cause of death and disability. Impairments in blood circulation that accompany heart failure can be traced, in part, to alterations in the activity of the sarcoplasmic reticulum Ca2+ pump that are induced by its interactions with phospholamban, a reversible inhibitor. If phospholamban becomes superinhibitory or chronically inhibitory, contractility is diminished, inducing dilated cardiomyopathy in mice and humans. In mice, phospholamban seems to encumber an otherwise healthy heart, but humans with a phospholamban-null genotype develop early-onset dilated cardiomyopathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kass, D. A., Hare, J. M. & Georgakopoulos, D. Murine cardiac function: a cautionary tail. Circ. Res. 82, 519–522 (1998).
Levitzki, A. From epinephrine to cyclic AMP. Science 241, 800–806 (1988).
Bers, D. M. Calcium and cardiac rhythms: physiological and pathophysiological. Circ. Res. 90, 14–17 (2002).
Simmerman, H. K. & Jones, L. R. Phospholamban: protein structure, mechanism of action, and role in cardiac function. Physiol. Rev. 78, 921–947 (1998).
Toyoshima, C. et al. Modeling of the inhibitory interaction of phospholamban with the Ca2+ ATPase. Proc. Natl Acad. Sci USA 100, 467–472 (2003). Structural modelling shows that grooves in the transmembrane and cytosolic domains of SERCA1a provide high-affinity binding sites for PLN in the E 2 , but not the E 1 .2Ca2+ conformation.
Kimura, Y., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Phospholamban inhibitory function is activated by depolymerization. J. Biol. Chem. 272, 15061–15064 (1997). Scanning mutagenesis of the PLN transmembrane helix shows two faces: one interacts with SERCA, so that mutants remain pentameric but lose inhibitory function; the other interacts with PLN, so that mutants become monomeric and gain inhibitory function. It is deduced that the PLN monomer is the active inhibitory species.
Zhai, J. et al. Cardiac-specific overexpression of a superinhibitory pentameric phospholamban mutant enhances inhibition of cardiac function in vivo. J. Biol. Chem. 275, 10538–10544 (2000). Superinhibition of SERCA2a by a PLN mutant results in dilated cardiomyopathy in a mouse model.
Zvaritch, E. et al. The transgenic expression of highly inhibitory monomeric forms of phospholamban in mouse heart impairs cardiac contractility. J. Biol. Chem. 275, 14985–14991 (2000).
Haghighi, K. et al. Superinhibition of sarcoplasmic reticulum function by phospholamban induces cardiac contractile failure. J. Biol. Chem. 276, 24145–24152 (2001).
Luo, W. et al. Targeted ablation of the phospholamban gene is associated with markedly enhanced myocardial contractility and loss of β-agonist stimulation. Circ. Res. 75, 401–409 (1994). The first clear evidence of the important role of PLN in the regulation of cardiac contractility in a whole animal.
Kadambi, V. J. et al. Cardiac-specific overexpression of phospholamban alters calcium kinetics and resultant cardiomyocyte mechanics in transgenic mice. J. Clin. Invest. 97, 533–539 (1996). Only about 40% of SERCA2a molecules are inhibited by PLN under normal conditions, even though PLN is in molar excess over SERCA2a.
Koss, K. L. & Kranias, E. G. Phospholamban: a prominent regulator of myocardial contractility. Circ. Res. 79, 1059–1063 (1996).
Minamisawa, S. et al. Chronic phospholamban–sarcoplasmic reticulum calcium ATPase interaction is the critical calcium cycling defect in dilated cardiomyopathy. Cell 99, 313–322 (1999). PLN ablation can reverse heart failure in a cardiomyopathic mouse model that is induced by a mutation in a structural protein.
He, H. et al. Effects of mutant and antisense RNA of phospholamban on SR Ca2+-ATPase activity and cardiac myocyte contractility. Circulation 100, 974–980 (1999).
Hoshijima, M. et al. Chronic suppression of heart-failure progression by a pseudophosphorylated mutant of phospholamban via in vivo cardiac rAAV gene delivery. Nature Med. 8, 864–871 (2002).
Schmitt, J. P. et al. Dilated cardiomyopathy and heart failure caused by a mutation in phospholamban. Science 299, 1410–1413 (2003). A human PLN mutation that is associated with heart failure in man inhibits PKA, leading to a lack of PLN phosphorylation and chronic inhibition of SERCA2a.
Haghighi, K. et al. Human phospholamban null results in lethal dilated cardiomyopathy: critical difference between mouse and man. J. Clin. Invest. 111, 869–876 (2003). In contrast to the beneficial effects of PLN ablation in mice, the absence of PLN in man might be associated with heart failure.
Kirchberger, M. A., Tada, M., Repke, D. I. & Katz, A. M. Cyclic adenosine 3′,5′-monophosphate-dependent protein kinase stimulation of calcium uptake by canine cardiac microsomes. J. Mol. Cell. Cardiol. 4, 673–680 (1972).
Katz, A. M. Discovery of phospholamban. A personal history. Ann. NY Acad. Sci. 853, 9–19 (1998).
Tada, M., Kirchberger, M. A. & Katz, A. M. Phosphorylation of a 22,000-dalton component of the cardiac sarcoplasmic reticulum by adenosine 3′:5′-monophosphate-dependent protein kinase. J. Biol. Chem. 250, 2640–2647 (1975). Identification of PLN as a crucial player in β-adrenergic stimulation of the heart.
La Raia, P. J. & Morkin, E. Adenosine 3′,5′-monophosphate dependent membrane phosphorylation: a possible mechanism for the control of microsomal calcium transport in heart muscle. Circ. Res. 35, 298–306 (1974).
Cantilina, T., Sagara, Y., Inesi, G. & Jones, L. R. Comparative studies of cardiac and skeletal sarcoplasmic reticulum ATPases. Effect of a phospholamban antibody on enzyme activation by Ca2+. J. Biol. Chem. 268, 17018–17025 (1993).
Hughes, G., Starling, A. P., Sharma, R. P., East, J. M. & Lee, A. G. An investigation of the mechanism of inhibition of the Ca2+-ATPase by phospholamban. Biochem. J. 318, 973–979 (1996).
Kranias, E. G. & Solaro, R. J. Phosphorylation of troponin I and phospholamban during catecholamine stimulation of rabbit heart. Nature 298, 182–184 (1982). PLN is phosphorylated when elevated levels of catecholamines stimulate contractility of intact hearts.
Wegener, A. D., Simmerman, H. K., Lindemann, J. P. & Jones, L. R. Phospholamban phosphorylation in intact ventricles. Phosphorylation of serine 16 and threonine 17 in response to β-adrenergic stimulation. J. Biol. Chem. 264, 11468–11474 (1989).
Talosi, L., Edes, I. & Kranias, E. G. Intracellular mechanisms mediating reversal of β-adrenergic stimulation in intact beating hearts. Am. J. Physiol. 264, H791–H797 (1993).
Lindemann, J. P., Jones, L. R., Hathaway, D. R., Henry, B. G. & Watanabe, A. M. β-adrenergic stimulation of phospholamban phosphorylation and Ca2+-ATPase activity in guinea pig ventricles. J. Biol. Chem. 258, 464–471 (1983).
Garvey, J. L., Kranias, E. G. & Solaro, R. J. Phosphorylation of C-protein, troponin I and phospholamban in isolated rabbit hearts. Biochem. J. 249, 709–714 (1988).
Mundina-Weilenmann, C., Vittone, L., Ortale, M., de Cingolani, G. C. & Mattiazzi, A. Immunodetection of phosphorylation sites gives new insights into the mechanisms underlying phospholamban phosphorylation in the intact heart. J. Biol. Chem. 271, 33561–33567 (1996).
Fujii, J. et al. Complete complementary DNA-derived amino acid sequence of canine cardiac phospholamban. J. Clin. Invest. 79, 301–304 (1987).
Odermatt, A. et al. Characterization of the gene encoding human sarcolipin (SLN), a proteolipid associated with SERCA1: absence of structural mutations in five patients with Brody disease. Genomics 45, 541–553 (1997).
Odermatt, A. et al. Sarcolipin regulates the activity of SERCA1, the fast-twitch skeletal muscle sarcoplasmic reticulum Ca2+-ATPase. J. Biol. Chem. 273, 12360–12369 (1998).
Mortishire-Smith, R. J. et al. Solution structure of the cytoplasmic domain of phopholamban: phosphorylation leads to a local perturbation in secondary structure. Biochemistry 34, 7603–7613 (1995).
Pollesello, P., Annila, A. & Ovaska, M. Structure of the 1–36 amino-terminal fragment of human phospholamban by nuclear magnetic resonance and modeling of the phospholamban pentamer. Biophys. J. 76, 1784–1795 (1999).
Lamberth, S. et al. NMR structure of phospholamban. Helvetica Chimica Acta 83, 2141–2152 (2000).
Mascioni, A., Karim, C., Barany, G., Thomas, D. D. & Veglia, G. Structure and orientation of sarcolipin in lipid environments. Biochemistry 41, 475–482 (2002).
James, P., Inui, M., Tada, M., Chiesi, M. & Carafoli, E. Nature and site of phospholamban regulation of the Ca2+ pump of sarcoplasmic reticulum. Nature 342, 90–92 (1989).
Toyoshima, C. & Nomura, H. Structural changes in the calcium pump accompanying the dissociation of calcium. Nature 418, 605–611 (2002).
Toyofuku, T., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Identification of regions in the Ca2+-ATPase of sarcoplasmic reticulum that affect functional association with phospholamban. J. Biol. Chem. 268, 2809–2815 (1993).
Asahi, M., Kimura, Y., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Transmembrane helix m6 in sarco(endo)plasmic reticulum Ca2+-ATPase forms a functional interaction site with phospholamban. Evidence for physical interactions at other sites. J. Biol. Chem. 274, 32855–32862 (1999).
Toyofuku, T., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Amino acids Lys-Asp-Asp-Lys-Pro-Val402 in the Ca2+-ATPase of cardiac sarcoplasmic reticulum are critical for functional association with phospholamban. J. Biol. Chem. 269, 22929–22932 (1994).
Toyofuku, T., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Amino acids Glu2 to Ile18 in the cytoplasmic domain of phospholamban are essential for functional association with the Ca2+-ATPase of sarcoplasmic reticulum. J. Biol. Chem. 269, 3088–3094 (1994).
Kimura, Y., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Phospholamban regulates the Ca2+-ATPase through intramembrane interactions. J. Biol. Chem. 271, 21726–21731 (1996).
Arkin, I. T. et al. Structural organization of the pentameric transmembrane α-helices of phospholamban, a cardiac ion channel. EMBO J. 13, 4757–4764 (1994).
Simmerman, H. K., Kobayashi, Y. M., Autry, J. M. & Jones, L. R. A leucine zipper stabilizes the pentameric membrane domain of phospholamban and forms a coiled-coil pore structure. J. Biol. Chem. 271, 5941–5946 (1996).
Adams, P. D., Arkin, I. T., Engelman, D. M. & Brunger, A. T. Computational searching and mutagenesis suggest a structure for the pentameric transmembrane domain of phospholamban. Nature Struct. Biol. 2, 154–162 (1995).
Autry, J. M. & Jones, L. R. Functional co-expression of the canine cardiac Ca2+ pump and phospholamban in Spodoptera frugiperda (Sf21) cells reveals new insights on ATPase regulation. J. Biol. Chem. 272, 15872–15880 (1997).
Kimura, Y., Asahi, M., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Phospholamban domain Ib mutations influence functional interactions with the Ca2+-ATPase isoform of cardiac sarcoplasmic reticulum. J. Biol. Chem. 273, 14238–14241 (1998).
MacLennan, D. H., Rice, W. J. & Green, N. M. The mechanism of Ca2+ transport by sarco(endo)plasmic reticulum Ca2+-ATPases. J. Biol. Chem. 272, 28815–28818 (1997).
Toyoshima, C., Nakasako, M., Nomura, H. & Ogawa, H. Crystal structure of the calcium pump of sarcoplasmic reticulum at 2.6 å resolution. Nature 405, 647–655 (2000).
Knighton, D. R. et al. Structure of a peptide inhibitor bound to the catalytic subunit of cyclic adenosine monophosphate-dependent protein kinase. Science 253, 414–420 (1991).
Hutter, M. C. et al. A structural model of the complex formed by phospholamban and the calcium pump of sarcoplasmic reticulum obtained by molecular mechanics. Chembiochem. 3, 1200–1208 (2002).
Asahi, M., McKenna, E., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Physical interactions between phospholamban and sarco(endo)plasmic reticulum Ca2+-ATPases are dissociated by elevated Ca2+, but not by phospholamban phosphorylation, vanadate, or thapsigargin, and are enhanced by ATP. J. Biol. Chem. 275, 15034–15038 (2000).
Luo, W. et al. Phospholamban gene dosage effects in the mammalian heart. Circ. Res. 78, 839–847 (1996).
Wolska, B. M., Stojanovic, M. O., Luo, W., Kranias, E. G. & Solaro, R. J. Effect of ablation of phospholamban on dynamics of cardiac myocyte contraction and intracellular Ca2+. Am. J. Physiol. 271, C391–C397 (1996).
Li, L., Chu, G., Kranias, E. G. & Bers, D. M. Cardiac myocyte calcium transport in phospholamban knockout mouse: relaxation and endogenous CaMKII effects. Am. J. Physiol. 274, H1335–H1347 (1998).
Lorenz, J. N. & Kranias, E. G. Regulatory effects of phospholamban on cardiac function in intact mice. Am. J. Physiol. 273, H2826–H2831 (1997).
Kiss, E. et al. β-adrenergic regulation of cAMP and protein phosphorylation in phospholamban-knockout mouse hearts. Am. J. Physiol. 272, H785–H790 (1997).
Santana, L. F., Gomez, A. M., Kranias, E. G. & Lederer, W. J. Amount of calcium in the sarcoplasmic reticulum: influence on excitation–contraction coupling in heart muscle. Heart Vessels Suppl. 12, 44–49 (1997).
Masaki, H., Sato, Y., Luo, W., Kranias, E. G. & Yatani, A. Phospholamban deficiency alters inactivation kinetics of L-type Ca2+ channels in mouse ventricular myocytes. Am. J. Physiol. 272, H606–H612 (1997).
Chu, G. et al. Compensatory mechanisms associated with the hyperdynamic function of phospholamban-deficient mouse hearts. Circ. Res. 79, 1064–1076. (1996).
Desai, K. H., Schauble, E., Luo, W., Kranias, E. & Bernstein, D. Phospholamban deficiency does not compromise exercise capacity. Am. J. Physiol. 276, H1172–H1177 (1999).
Slack, J. P. et al. The enhanced contractility of the phospholamban-deficient mouse heart persists with aging. J. Mol. Cell. Cardiol. 33, 1031–1040 (2001).
Kiriazis, H. et al. Hypertrophy and functional alterations in hyperdynamic phospholamban-knockout mouse hearts under chronic aortic stenosis. Cardiovasc. Res. 53, 372–381 (2002).
Cross, H. R., Kranias, E. G., Murphy, E. & Steenbergen, C. Ablation of PLB exacerbates ischemic injury to a lesser extent in female than male mice: protective role of NO. Am. J. Physiol. Heart Circ. Physiol. 284, H683–H690 (2003).
Lalli, J., Harrer, J. M., Luo, W., Kranias, E. G. & Paul, R. J. Targeted ablation of the phospholamban gene is associated with a marked decrease in sensitivity in aortic smooth muscle. Circ. Res. 80, 506–513 (1997).
Slack, J. P., Grupp, I. L., Luo, W. & Kranias, E. G. Phospholamban ablation enhances relaxation in the murine soleus. Am. J. Physiol. 273, C1–C6 (1997).
Nobe, K., Sutliff, R. L., Kranias, E. G. & Paul, R. J. Phospholamban regulation of bladder contractility: evidence from gene-altered mouse models. J. Physiol. 535, 867–878 (2001).
Sutliff, R. L., Hoying, J. B., Kadambi, V. J., Kranias, E. G. & Paul, R. J. Phospholamban is present in endothelial cells and modulates endothelium-dependent relaxation. Evidence from phospholamban gene-ablated mice. Circ. Res. 84, 360–364 (1999).
Dash, R. et al. Interactions between phospholamban and β-adrenergic drive may lead to cardiomyopathy and early mortality. Circulation 103, 889–896 (2001).
Dash, R., Frank, K. F., Carr, A. N., Moravec, C. S. & Kranias, E. G. Gender influences on sarcoplasmic reticulum Ca2+-handling in failing human myocardium. J. Mol. Cell. Cardiol. 33, 1345–1353 (2001).
Colyer, J. & Wang, J. H. Dependence of cardiac sarcoplasmic reticulum calcium pump activity on the phosphorylation status of phospholamban. J. Biol. Chem. 266, 17486–17493 (1991).
Brittsan, A. G., Carr, A. N., Schmidt, A. G. & Kranias, E. G. Maximal inhibition of SERCA2 Ca2+ affinity by phospholamban in transgenic hearts overexpressing a non-phosphorylatable form of phospholamban. J. Biol. Chem. 275, 12129–12135 (2000).
Schmidt, A. G. et al. Structural and functional implications of the phospholamban hinge domain: impaired SR Ca2+ uptake as a primary cause of heart failure. Cardiovasc. Res. 56, 248–259 (2002).
Luo, W. et al. Transgenic approaches to define the functional role of dual site phospholamban phosphorylation. J. Biol. Chem. 273, 4734–4739 (1998).
Chu, G. et al. A single site (Ser16) phosphorylation in phospholamban is sufficient in mediating its maximal cardiac responses to β-agonists. J. Biol. Chem. 275, 38938–38943 (2000).
Vittone, L., Mundina-Weilenmann, C., Said, M., Ferrero, P. & Mattiazzi, A. Time course and mechanisms of phosphorylation of phospholamban residues in ischemia-reperfused rat hearts. Dissociation of phospholamban phosphorylation pathways. J. Mol. Cell. Cardiol. 34, 39–50 (2002).
Hagemann, D. et al. Frequency-encoding Thr17 phospholamban phosphorylation is independent of Ser16 phosphorylation in cardiac myocytes. J. Biol. Chem. 275, 22532–22536 (2000).
Beuckelmann, D. J., Nabauer, M. & Erdmann, E. Intracellular calcium handling in isolated ventricular myocytes from patients with terminal heart failure. Circulation 85, 1046–1055 (1992).
Dipla, K., Mattiello, J. A., Margulies, K. B., Jeevanandam, V. & Houser, S. R. The sarcoplasmic reticulum and the Na+/Ca2+ exchanger both contribute to the Ca2+ transient of failing human ventricular myocytes. Circ. Res. 84, 435–444 (1999).
Hasenfuss, G. et al. Relation between myocardial function and expression of sarcoplasmic reticulum Ca2+-ATPase in failing and nonfailing human myocardium. Circ. Res. 75, 434–442 (1994).
Schwinger, R. H. et al. Unchanged protein levels of SERCA II and phospholamban but reduced Ca2+ uptake and Ca2+-ATPase activity of cardiac sarcoplasmic reticulum from dilated cardiomyopathy patients compared with patients with nonfailing hearts. Circulation 92, 3220–3228 (1995).
Frank, K., Bolck, B., Bavendiek, U. & Schwinger, R. H. Frequency dependent force generation correlates with sarcoplasmic calcium ATPase activity in human myocardium. Basic Res. Cardiol. 93, 405–411 (1998).
Movsesian, M. A., Karimi, M., Green, K. & Jones, L. R. Ca2+-transporting ATPase, phospholamban, and calsequestrin levels in nonfailing and failing human myocardium. Circulation 90, 653–657 (1994).
Meyer, M. et al. Alterations of sarcoplasmic reticulum proteins in failing human dilated cardiomyopathy. Circulation 92, 778–784 (1995).
Schwinger, R. H. et al. Reduced Ca2+-sensitivity of SERCA2a in failing human myocardium due to reduced serine-16 phospholamban phosphorylation. J. Mol. Cell. Cardiol. 31, 479–491 (1999).
Giordano, F. J. et al. Adenovirus-mediated gene transfer reconstitutes depressed sarcoplasmic reticulum Ca2+-ATPase levels and shortens prolonged cardiac myocyte Ca2+ transients. Circulation 96, 400–403 (1997).
del Monte, F. et al. Restoration of contractile function in isolated cardiomyocytes from failing human hearts by gene transfer of SERCA2a. Circulation 100, 2308–2311 (1999).
Miyamoto, M. I. et al. Adenoviral gene transfer of SERCA2a improves left-ventricular function in aortic-banded rats in transition to heart failure. Proc. Natl Acad. Sci. USA 97, 793–798 (2000).
del Monte, F. et al. Improvement in survival and cardiac metabolism after gene transfer of sarcoplasmic reticulum Ca2+-ATPase in a rat model of heart failure. Circulation 104, 1424–1429 (2001).
He, H. et al. Overexpression of the rat sarcoplasmic reticulum Ca2+ ATPase gene in the heart of transgenic mice accelerates calcium transients and cardiac relaxation. J. Clin. Invest. 100, 380–389 (1997).
Baker, D. L. et al. Targeted overexpression of the sarcoplasmic reticulum Ca2+-ATPase increases cardiac contractility in transgenic mouse hearts. Circ. Res. 83, 1205–1214 (1998).
Eizema, K. et al. Adenovirus-based phospholamban antisense expression as a novel approach to improve cardiac contractile dysfunction: comparison of a constitutive viral versus an endothelin-1-responsive cardiac promoter. Circulation 101, 2193–2199 (2000).
del Monte, F., Harding, S. E., Dec, G. W., Gwathmey, J. K. & Hajjar, R. J. Targeting phospholamban by gene transfer in human heart failure. Circulation 105, 904–907 (2002).
Sato, Y. et al. Rescue of contractile parameters and myocyte hypertrophy in calsequestrin overexpressing myocardium by phospholamban ablation. J. Biol. Chem. 276, 9392–9399 (2001).
Freeman, K. et al. Alterations in cardiac adrenergic signaling and calcium cycling differentially affect the progression of cardiomyopathy. J. Clin. Invest. 107, 967–974 (2001).
Song, Q. et al. Rescue of cardiomyocyte dysfunction by phospholamban ablation does not prevent ventricular failure in genetic hypertrophy. J. Clin. Invest. 111, 859–867 (2003).
Fink, M. A. et al. AKAP-mediated targeting of protein kinase a regulates contractility in cardiac myocytes. Circ. Res. 88, 291–297 (2001).
Pieske, B. et al. Functional effects of endothelin and regulation of endothelin receptors in isolated human nonfailing and failing myocardium. Circulation 99, 1802–1809 (1999).
MacLennan, D. H. & Reithmeier, R. A. Ion tamers. Nature Struct. Biol. 5, 409–411 (1998).
Zot, A. S. & Potter, J. D. Structural aspects of troponin–tropomyosin regulation of skeletal muscle contraction. Annu. Rev. Biophys. Biophys. Chem. 16, 535–559 (1987).
MacLennan, D. H. Ca2+ signalling and muscle disease. Eur. J. Biochem. 267, 5291–5297 (2000).
Loke, J. & MacLennan, D. H. Malignant hyperthermia and central core disease: disorders of Ca2+ release channels. Am. J. Med. 104, 470–486 (1998).
Priori, S. G. et al. Mutations in the cardiac ryanodine receptor gene (hRyR2) underlie catecholaminergic polymorphic ventricular tachycardia. Circulation 103, 196–200 (2001).
Laitinen, P. J. et al. Mutations of the cardiac ryanodine receptor (RyR2) gene in familial polymorphic ventricular tachycardia. Circulation 103, 485–490 (2001).
Postma, A. V. et al. Absence of calsequestrin 2 causes severe forms of catecholaminergic polymorphic ventricular tachycardia. Circ. Res. 91, e21–e26 (2002).
Sakuntabhai, A. et al. Mutations in ATP2A2, encoding a Ca2+ pump, cause Darier disease. Nature Genet. 21, 271–277 (1999).
Wawrzynow, A. et al. Sarcolipin, the 'proteolipid' of skeletal muscle sarcoplasmic reticulum, is a unique, amphipathic, 31-residue peptide. Arch. Biochem. Biophys. 298, 620–623 (1992).
Gayan-Ramirez, G., Vanzeir, L., Wuytack, F. & Decramer, M. Corticosteroids decrease mRNA levels of SERCA pumps, whereas they increase sarcolipin mRNA in the rat diaphragm. J. Physiol. 524, 387–397 (2000).
Minamisawa, S. et al. Atrial-specific expression of sarcolipin is regulated during development and hypertrophic remodeling. J. Biol. Chem. 278, 9570–9575 (2003).
Tupling, A. R., Asahi, M. & MacLennan, D. H. Sarcolipin overexpression in rat slow twitch muscle inhibits sarcoplasmic reticulum Ca2+ uptake and impairs contractile function. J. Biol. Chem. 277, 44740–44746 (2002).
Asahi, M., Kurzydlowski, K., Tada, M. & MacLennan, D. H. Sarcolipin inhibits polymerization of phospholamban to induce superinhibition of sarco(endo)plasmic reticulum Ca2+-ATPases (SERCAs). J. Biol. Chem. 277, 26725–26728 (2002).
Asahi, M. et al. Sarcolipin regulates sarco(endo)plasmic reticulum Ca2+-ATPase (SERCA1a) by binding to transmembrane helices alone or in association with phospholamban. Proc. Natl Acad. Sci. USA 100, 5040–5045 (2003).
de Meis, L. & Vianna, A. L. Energy interconversion by the Ca2+-dependent ATPase of the sarcoplasmic reticulum. Annu. Rev. Biochem. 48, 275–293 (1979).
Stokes, D. L. & Green, N. M. Structure and function of the calcium pump. Annu. Rev. Biophys. Biomol. Struct. 32, 445–468 (2003).
Cornea, R. L., Jones, L. R., Autry, J. M. & Thomas, D. D. Mutation and phosphorylation change the oligomeric structure of phospholamban in lipid bilayers. Biochemistry 36, 2960–2967 (1997).
Acknowledgements
We are grateful to D. Bers, A. Gramolini, K. Haghighi, G. Inesi and C. Toyoshima for their helpful comments on this manuscript. The original studies described in this review were supported by grants to D.H.M. from the Heart and Stroke Foundation of Ontario, the Canadian Institutes for Health Research and the Canadian Genetic Diseases Network of Centres of Excellence, and to E.G.K. by grants from the National Institutes of Health (USA).
Author information
Authors and Affiliations
Corresponding author
Glossary
- CARDIAC RESERVE
-
The maximum percentage that the cardiac output can increase above normal — the ability of the heart to adjust rapidly to demands placed on it.
- β-AGONIST
-
A molecule that activates β-adrenergic receptors.
- β-ADRENERGIC STIMULATION
-
The ligand- or agonist-dependent activation of β-adrenergic receptors and subsequent signalling events.
- SARCOPLASMIC RETICULUM
-
(SR). An organellar membrane system that encases each myofibril within a muscle cell. Its essential components are a Ca2+-ATPase (Ca2+ pump), lumenal Ca2+-sequestering proteins and a Ca2+-release channel.
- SARCO(ENDO)PLASMIC RETICULUM Ca2+-ATPASE
-
(SERCA). A pump that is located in sarcoplasmic or endoplasmic reticulum membranes that couples ATP hydrolysis to the transport of Ca2+ from cytosolic to lumenal spaces.
- RYANODINE RECEPTOR
-
(RyR). A Ca2+-release channel that is located in the membrane of the sarcoplasmic and the endoplasmic reticulum that is regulated by protein–protein interactions with the dihydropyridine receptor and by a series of ligands, including Ca2+ itself.
- DIHYDROPYRIDINE RECEPTOR
-
(DHPR). A slow, or L-type, voltage-dependent Ca2+-entry channel that is located in the plasma membrane. DHPRs require a membrane potential that is greater than −30 mV for activation, and they are commonly found in neurons, neuroendocrine cells and muscle cells.
- PLASMA-MEMBRANE Ca2+-ATPASE
-
(PMCA). A plasma-membrane pump that couples ATP hydrolysis to the transport of Ca2+ from cytosolic to extracellular spaces.
- Na+/Ca2+ EXCHANGER
-
(NCX). A plasma-membrane enzyme that exchanges three moles of Na+ for one mole of Ca2+ either inward or outward, depending on ionic gradients across the membrane.
- LUSITROPIC
-
Affecting cardiac relaxation.
- INOTROPIC
-
Affecting the force of cardiac contractions.
- CARDIOMYOPATHY
-
A disease of the heart muscle.
- VMAX
-
The maximal rate of enzymatic activity.
- FAST-TWITCH SKELETAL MUSCLE
-
A rapidly contracting and relaxing muscle, such as the extensor digitorum longus, which is primarily involved in bodily movement.
- SLOW-TWITCH SKELETAL MUSCLE
-
A slowly contracting muscle, such as the soleus, with major involvement in posture maintenance.
- SOLEUS
-
A predominantly slow-twitch muscle in the leg.
- G PROTEIN
-
A heterotrimeric, guanyl nucleotide-binding protein that transmits signals from ligand-activated receptors to effector molecules.
- LOD SCORE
-
The log10 of the odds of linkage between genotype and phenotype versus non-linkage.
Rights and permissions
About this article
Cite this article
MacLennan, D., Kranias, E. Phospholamban: a crucial regulator of cardiac contractility. Nat Rev Mol Cell Biol 4, 566–577 (2003). https://doi.org/10.1038/nrm1151
Issue Date:
DOI: https://doi.org/10.1038/nrm1151
This article is cited by
-
The interplay of inflammation, exosomes and Ca2+ dynamics in diabetic cardiomyopathy
Cardiovascular Diabetology (2023)
-
Small open reading frames: a comparative genetics approach to validation
BMC Genomics (2023)
-
Identification of cell-biologic mechanisms of coronary artery spasm and its ex vivo diagnosis using peripheral blood-derived iPSCs
Biomaterials Research (2023)
-
Skeletal muscle overexpression of sAnk1.5 in transgenic mice does not predispose to type 2 diabetes
Scientific Reports (2023)
-
Phosphoinositides and intracellular calcium signaling: novel insights into phosphoinositides and calcium coupling as negative regulators of cellular signaling
Experimental & Molecular Medicine (2023)