Abstract
The management of patients with systemic sclerosis (SSc) can be challenging because disease-associated damage is often difficult to reverse and curative therapies are not yet available. Early identification and appropriate monitoring of patients with SSc is, therefore, critical so that active disease can be controlled and tissue damage prevented or delayed. However, early diagnosis of SSc is often difficult because the early clinical stages of the disease can be very similar to that of other autoimmune conditions. Screening for major organ manifestations of SSc, particularly interstitial lung disease, pulmonary hypertension, renal involvement and cardiac disease is a priority because involvement of these organs is associated with shorter life expectancies and early intervention might prevent disease progression. The prevention and management of digital ischaemia is also important as appropriate therapy often prevents substantial morbidity and functional loss. Treating gastrointestinal dysmotility can usually be managed using proton pump inhibitors and promotility agents, although in severe cases total parenteral nutrition is required. Calcinosis in patients with SSc is another common challenge that requires appropriate disease management and pain control. Each of these topics, which are relevant to both physicians and patients with SSc, are reviewed in further detail herein.
Key Points
-
Screening and identification of risk factors for internal organ manifestations of systemic sclerosis (SSc) is important and should be done soon after diagnosis and regularly thereafter
-
Autoantibody testing can be used to identify patients who are at increased risk of specific scleroderma-related complications; these patients should be closely monitored for early detection of such complications
-
Interstitial lung disease and pulmonary hypertension are serious and often deadly complications of scleroderma; patients with these complications should receive aggressive treatment early in the disease process
-
In patients with scleroderma, gastrointestinal disease can be severe and targeted intervention with gastrointestinal antacids is usually required
-
Promotility agents can also improve symptoms of gastrointestinal disease but in severe cases, total parenteral nutrition might be required
-
Scleroderma is a complex multi-organ disease and the management of affected patients often requires coordinated efforts with experienced specialists in other fields
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
LeRoy, E. C. & Medsger, T. A. Jr. Criteria for the classification of early systemic sclerosis. J. Rheumatol. 28, 1573–1576 (2001).
Basu, D. & Reveille, J. D. Anti-scl-70. Autoimmunity 38, 65–72 (2005).
Emilie, S. et al. Anti-RNA polymerase III antibodies are associated with scleroderma renal crisis in a French cohort. Scand. J. Rheum. 40, 404–406 (2011).
Faucher, B. P. et al. Low prevalence of anti-RNA polymerase III antibodies in a French scleroderma population: anti-RNA polymerase III scleroderma. Eur. J. Intern. Med. 21, 114–117 (2010).
Nikpour, M. et al. Prevalence, correlates and clinical usefulness of antibodies to RNA polymerase III in systemic sclerosis: a cross-sectional analysis of data from an Australian cohort. Arthritis Res. Ther. 13, R211 (2011).
Nihtyanova, S. I., Parker, J. C., Black, C. M., Bunn, C. C. & Denton, C. P. A longitudinal study of anti-RNA polymerase III antibody levels in systemic sclerosis. Rheumatology (Oxford) 48, 1218–1221 (2009).
Shanmugam, V. K. & Steen, V. D. Renal disease in scleroderma: an update on evaluation, risk stratification, pathogenesis and management. Curr. Opin. Rheumatol. 24, 669–676 (2012).
Steen, V. D. Scleroderma renal crisis. Rheum. Dis. Clin. North Am. 29, 315–333 (2003).
Greidinger, E. L. et al. African-American race and antibodies to topoisomerase I are associated with increased severity of scleroderma lung disease. Chest 114, 801–807 (1998).
Steen, V. D. The many faces of scleroderma. Rheum. Dis. Clin. North Am. 34, 1–15 (2008).
Steen, V. D., Lucas, M., Fertig, N. & Medsger, T. A. Jr. Pulmonary arterial hypertension and severe pulmonary fibrosis in systemic sclerosis patients with a nucleolar antibody. J. Rheumatol. 34, 2230–2235 (2007).
Khanna, D. et al. Clinical course of lung physiology in patients with scleroderma and interstitial lung disease: analysis of the Scleroderma Lung Study Placebo Group. Arthritis Rheum. 63, 3078–3085 (2011).
Perera, A. et al. Clinical subsets, skin thickness progression rate, and serum antibody levels in systemic sclerosis patients with anti-topoisomerase I antibody. Arthritis Rheum. 56, 2740–2746 (2007).
Merkel, P. A. et al. Patterns and predictors of change in outcome measures in clinical trials in scleroderma: an individual patient meta-analysis of 629 subjects with diffuse cutaneous systemic sclerosis. Arthritis Rheum. 64, 3420–3429 (2012).
Clements, P. J. et al. Skin thickness score as a predictor and correlate of outcome in systemic sclerosis: high-dose versus low-dose penicillamine trial. Arthritis Rheum. 43, 2445–2454 (2000).
Tashkin, D. P. et al. Cyclophosphamide versus placebo in scleroderma lung disease. N. Engl. J. Med. 354, 2655–2666 (2006).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
Derk, C. T. et al. A prospective open-label study of mycophenolate mofetil for the treatment of diffuse systemic sclerosis. Rheumatology (Oxford) 48, 1595–1599 (2009).
Le, E. N., Wigley, F. M., Shah, A. A., Boin, F. & Hummers, L. K. Long-term experience of mycophenolate mofetil for treatment of diffuse cutaneous systemic sclerosis. Ann. Rheum. Dis. 70, 1104–1107 (2011).
Koutroumpas, A., Ziogas, A., Alexiou, I., Barouta, G. & Sakkas, L. I. Mycophenolate mofetil in systemic sclerosis-associated interstitial lung disease. Clin. Rheumatol. 29, 1167–1168 (2010).
Gerbino, A. J., Goss, C. H. & Molitor, J. A. Effect of mycophenolate mofetil on pulmonary function in scleroderma-associated interstitial lung disease. Chest 133, 455–460 (2008).
Zamora, A. C. et al. Use of mycophenolate mofetil to treat scleroderma-associated interstitial lung disease. Respir. Med. 102, 150–155 (2008).
Simeon-Aznar, C. P. et al. Effect of mycophenolate sodium in scleroderma-related interstitial lung disease. Clin. Rheumatol. 30, 1393–1398 (2011).
van den Hoogen, F. H. et al. Comparison of methotrexate with placebo in the treatment of systemic sclerosis: a 24 week randomized double-blind trial, followed by a 24 week observational trial. Br. J. Rheumatol. 35, 364–372 (1996).
Pope, J. E. et al. A randomized, controlled trial of methotrexate versus placebo in early diffuse scleroderma. Arthritis Rheum. 44, 1351–1358 (2001).
Johnson, S. R., Feldman, B. M., Pope, J. E. & Tomlinson, G. A. Shifting our thinking about uncommon disease trials: the case of methotrexate in scleroderma. J. Rheumatol. 36, 323–329 (2009).
Spiera, R. F. et al. Imatinib mesylate (Gleevec) in the treatment of diffuse cutaneous systemic sclerosis: results of a 1-year, phase IIa, single-arm, open-label clinical trial. Ann. Rheum. Dis. 70, 1003–1009 (2011).
Khanna, D. et al. A one-year, phase I/IIa, open-label pilot trial of imatinib mesylate in the treatment of systemic sclerosis-associated active interstitial lung disease. Arthritis Rheum. 63, 3540–3546 (2011).
Iwamoto, N., Distler, J. H. & Distler, O. Tyrosine kinase inhibitors in the treatment of systemic sclerosis: from animal models to clinical trials. Curr. Rheumatol. Rep. 13, 21–27 (2011).
Distler, O. et al., A multi-center, open-label, proof of concept study of imatinib mesylate demonstrates no benefit for the treatment of fibrosis in patients with early diffuse systemic sclerosis [abstract]. Arthritis Rheum. 62 (Suppl. 10), 560 (2010).
Pope, J. D. et al. Imatinib in active diffuse cutaneous systemic sclerosis: results of a six-month, randomized, double-blind, placebo-controlled, proof-of-concept pilot study at a single center. Arthritis Rheum. 63, 3547–3551 (2011).
van Laar, J. M., Farge, D. & Tyndall, A. Autologous Stem Cell Transplantation International Scleroderma (ASTIS) trial: hope on the horizon for patients with severe systemic sclerosis. Ann. Rheum. Dis. 64, 1515 (2005).
Tyndall, A. Successes and failures of stem cell transplantation in autoimmune diseases. Hematology Am. Soc. Hematol. Educ. Program 2011, 280–284 (2011).
Burt, R. K. et al. Autologous non-myeloablative haemopoietic stem-cell transplantation compared with pulse cyclophosphamide once per month for systemic sclerosis (ASSIST): an open-label, randomised phase 2 trial. Lancet 378, 498–506 (2011).
Tyndall, A. Stem cells: HSCT for systemic sclerosis—swallows and summers. Nat. Rev. Rheumatol. 7, 624–626 (2011).
Steen, V. D. Severe restrictive lung disease in systemic sclerosis. Arthritis Rheum. 37, 1238–1239 (1994).
Le Pavec, J. Scleroderma lung disease. Clin. Rev. Allergy Immunol. 40, 104–116 (2011).
Bussone, G. & Mouthon, L. Interstitial lung disease in systemic sclerosis. Autoimmun. Rev. 10, 248–255 (2011).
Launay, D. et al. High resolution computed tomography in fibrosing alveolitis associated with systemic sclerosis. J. Rheumatol. 33, 1789–1801 (2006).
Roth, M. D. et al. Predicting treatment outcomes and responder subsets in scleroderma-related interstitial lung disease. Arthritis Rheum. 63, 2797–2808 (2011).
Mittoo, S. et al. Long term effects of cyclophosphamide treatment on lung function and survival in scleroderma patients with interstitial lung disease. Open Rheumatol. J. 5, 1–6 (2011).
Chang, B., Schachna, L., White, B., Wigley, F. M. & Wise, R. A. Natural history of mild-moderate pulmonary hypertension and the risk factors for severe pulmonary hypertension in scleroderma. J. Rheumatol. 33, 269–274 (2006).
Cox, S. R. et al. Isolated pulmonary hypertension in scleroderma. Intern. Med. J. 35, 28–33 (2005).
Campo, A., Mathai, S. C., Girgis, R. E. & Hassoun, P. M. Is pulmonary hypertension really a late complication of systemic sclerosis? Chest 138, 461–462 (2010).
Allanore, Y. et al. High N-terminal pro-brain natriuretic peptide levels and low diffusing capacity for carbon monoxide as independent predictors of the occurrence of precapillary pulmonary arterial hypertension in patients with systemic sclerosis. Arthritis Rheum. 58, 284–291 (2008).
Steen, V. & Medsger, T. A. Jr. Predictors of isolated pulmonary hypertension in patients with systemic sclerosis and limited cutaneous involvement. Arthritis Rheum. 48, 516–522 (2003).
Shah, A. A., Wigley, F. M. & Hummers, L. K. Telangiectases in scleroderma: a potential clinical marker of pulmonary arterial hypertension. J. Rheumatol. 37, 98–104 (2010).
Manno, R. L., Wigley, F. M., Gelber, A. C. & Hummers, L. K. Late-age onset systemic sclerosis. J. Rheumatol. 38, 1317–1325 (2011).
Mukerjee, D. et al. Echocardiography and pulmonary function as screening tests for pulmonary arterial hypertension in systemic sclerosis. Rheumatology (Oxford) 43, 461–466 (2004).
Hachulla, E. et al. Early detection of pulmonary arterial hypertension in systemic sclerosis: a French nationwide prospective multicenter study. Arthritis Rheum. 52, 3792–3800 (2005).
Dimitroulas, T. et al. Significance of serum uric acid in pulmonary hypertension due to systemic sclerosis: a pilot study. Rheumatol. Int. 31, 263–267 (2011).
Mathai, S. C. et al. Disproportionate elevation of N-terminal pro-brain natriuretic peptide in scleroderma-related pulmonary hypertension. Eur. Respir. J. 35, 95–104 (2010).
Saggar, R. et al. Exercise-induced pulmonary hypertension associated with systemic sclerosis: four distinct entities. Arthritis Rheum. 62, 3741–3750 (2010).
Mathai, S. C. et al. Survival in pulmonary hypertension associated with the scleroderma spectrum of diseases: impact of interstitial lung disease. Arthritis Rheum. 60, 569–577 (2009).
Launay, D. et al. Clinical characteristics and survival in systemic sclerosis-related pulmonary hypertension associated with interstitial lung disease. Chest 140, 1016–1024 (2011).
Trad, S. et al. Pulmonary arterial hypertension is a major mortality factor in diffuse systemic sclerosis, independent of interstitial lung disease. Arthritis Rheum. 54, 184–191 (2006).
Walker, K. M., Pope, J., participating members of the Scleroderma Clinical Trials Consortium (SCTC) & Canadian Scleroderma Research Group (CSRG). Treatment of systemic sclerosis complications: what to use when first-line treatment fails—a consensus of systemic sclerosis experts. Semin. Arthritis Rheum. 42, 42–55 (2012).
Humbert, M. et al. Screening for pulmonary arterial hypertension in patients with systemic sclerosis: clinical characteristics at diagnosis and long-term survival. Arthritis Rheum. 63, 3522–3530 (2011).
Avouac, J. et al. European League Against Rheumatism (EULAR) Scleroderma Trial and Research group (EUSTAR) recommendations for the treatment of systemic sclerosis: methods of elaboration and results of systematic literature research. Ann. Rheum. Dis. 68, 629–634 (2009).
Schachna, L. et al. Lung transplantation in scleroderma compared with idiopathic pulmonary fibrosis and idiopathic pulmonary arterial hypertension. Arthritis Rheum. 54, 3954–3961 (2006).
Saggar, R. et al. Systemic sclerosis and bilateral lung transplantation: a single centre experience. Eur. Respir. J. 36, 893–900 (2010).
Shitrit, D. et al. Lung transplantation in patients with scleroderma: case series, review of the literature, and criteria for transplantation. Clin. Transplant. 23, 178–183 (2009).
Kubo, M. et al. Lung transplantation in patients with scleroderma. J. Heart Lung Transplant. 20, 174–175 (2001).
Hoppo, T. et al. Antireflux surgery preserves lung function in patients with gastroesophageal reflux disease and end-stage lung disease before and after lung transplantation. Arch. Surg. 146, 1041–1047 (2011).
Davis, R. D. et al. Improved lung allograft function after fundoplication in patients with gastroesophageal reflux disease undergoing lung transplantation. J. Thorac. Cardiovasc. Surg. 125, 533–542 (2003).
Robertson, A. G. et al. Lung transplantation, gastroesophageal reflux, and fundoplication. Ann. Thorac. Surg. 89, 653–660 (2010).
Cantu, E. 3rd & Davis, R. D. Jr. J. Maxwell Chamberlain Memorial Paper. Early fundoplication prevents chronic allograft dysfunction in patients with gastroesophageal reflux disease. Ann. Thorac. Surg. 78, 1142–1151 (2004).
Hartwig, M. G., Appel, J. Z. & Davis, R. D. Antireflux surgery in the setting of lung transplantation: strategies for treating gastroesophageal reflux disease in a high-risk population. Thorac. Surg. Clin. 15, 417–427 (2005).
Hassoun, P. M. Pulmonary arterial hypertension complicating connective tissue diseases. Semin. Respir. Crit. Care Med. 30, 429–439 (2009).
Denton, C. P. et al. Demographic, clinical and antibody characteristics of patients with digital ulcers in systemic sclerosis: data from the DUO Registry. Ann. Rheum. Dis. 71, 718–721 (2012).
McMahan, Z. H. & Wigley, F. M. Raynaud's phenomenon and digital ischaemia: a practical approach to risk stratification, diagnosis and management. Int. J. Clin. Rheumatol. 5, 355–370 (2010).
Hasegawa, M., Nagai, Y., Tamura, A. & Ishikawa, O. Arteriographic evaluation of vascular changes of the extremities in patients with systemic sclerosis. Br. J. Dermatol. 155, 1159–1164 (2006).
Taylor, M. H., McFadden, J. A., Bolster, M. B. & Silver, R. M. Ulner artery involvement in systemic sclerosis (scleroderma). J. Rheumatol. 29, 102–106 (2002).
Shenoy, P. D. et al. Efficacy of tadalafil in secondary Raynaud's phenomenon resistant to vasodilator therapy: a double-blind randomized cross-over trial. Rheumatology (Oxford) 49, 2420–2428 (2010).
Fries, R., Shariat, K., von Wilmowsky, H. & Bohm, M. Sildenafil in the treatment of Raynaud's phenomenon resistant to vasodilatory therapy. Circulation 112, 2980–2985 (2005).
Matucci-Cerinic, M. et al. Bosentan treatment of digital ulcers related to systemic sclerosis: results from the RAPIDS-2 randomised, double-blind, placebo-controlled trial. Ann. Rheum. Dis. 70, 32–38 (2011).
Korn, J. H. et al. Digital ulcers in systemic sclerosis: prevention by treatment with bosentan, an oral endothelin receptor antagonist. Arthritis Rheum. 50, 3985–3993 (2004).
Dziadzio, M. et al. Losartan therapy for Raynaud's phenomenon and scleroderma: clinical and biochemical findings in a fifteen-week, randomized, parallel-group, controlled trial. Arthritis Rheum. 42, 2646–2655 (1999).
Abou-Raya, A., Abou-Raya, S. & Helmii, M. Statins: potentially useful in therapy of systemic sclerosis-related Raynaud's phenomenon and digital ulcers. J. Rheumatol. 35, 1801–1808 (2008).
Kuwana, M. et al. Increase in circulating endothelial precursors by atorvastatin in patients with systemic sclerosis. Arthritis Rheum. 54, 1946–1951 (2006).
Furukawa, S. et al. Protective effect of pravastatin on vascular endothelium in patients with systemic sclerosis: a pilot study. Ann Rheum. Dis. 65, 1118–1120 (2006).
Derk, C. T. & Jimenez, S. A. Statins and the vasculopathy of systemic sclerosis: potential therapeutic agents? Autoimmun. Rev. 5, 25–32 (2006).
Herrick, A. L. Contemporary management of Raynaud's phenomenon and digital ischaemic complications. Curr. Opin. Rheumatol. 23, 555–561 (2011).
Milio, G. et al. Iloprost treatment in patients with Raynaud's phenomenon secondary to systemic sclerosis and the quality of life: a new therapeutic protocol. Rheumatology (Oxford) 45, 999–1004 (2006).
Shapiro, S. & Hills, N. S. Transition from IV to subcutaneous prostacyclin: premature withdrawal? Chest 132, 741–743 (2007).
Pope, J. et al. Iloprost and cisaprost for Raynaud's phenomenon in progressive systemic sclerosis. Cochrane Database of Systematic Reviews, Issue 2. Art. No.: CD000953. http://dx.doi.org/10.1002/14651858.CD000953.
Wigley, F. M. et al. Intravenous iloprost infusion in patients with Raynaud phenomenon secondary to systemic sclerosis. A multicenter, placebo-controlled, double-blind study. Ann. Intern. Med. 120, 199–206 (1994).
Kawald, A., Burmester, G. R., Huscher, D., Sunderkotter, C. & Riemekasten, G. Low versus high-dose iloprost therapy over 21 days in patients with secondary Raynaud's phenomenon and systemic sclerosis: a randomized, open, single-center study. J. Rheumatol. 35, 1830–1837 (2008).
Wigley, F. M. et al. Oral iloprost treatment in patients with Raynaud's phenomenon secondary to systemic sclerosis: a multicenter, placebo-controlled, double-blind study. Arthritis Rheum. 41, 670–677 (1998).
Vayssairat, M. Preventive effect of an oral prostacyclin analog, beraprost sodium, on digital necrosis in systemic sclerosis. French Microcirculation Society Multicenter Group for the Study of Vascular Acrosyndromes. J. Rheumatol. 26, 2173–2178 (1999).
Ruch, D. S., Holden, M., Smith, B. P., Smith, T. L. & Koman, L. A. Periarterial sympathectomy in scleroderma patients: intermediate-term follow-up. J. Hand Surg. Am. 27, 258–264 (2002).
Vonk, M. C. et al. Oesophageal dilatation on high-resolution computed tomography scan of the lungs as a sign of scleroderma. Ann. Rheum. Dis. 67, 1317–1321 (2008).
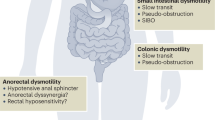
Sjogren, R. W. Gastrointestinal motility disorders in scleroderma. Arthritis Rheum. 37, 1265–1282 (1994).
Sallam, H., McNearney, T. A. & Chen, J. D. Systematic review: pathophysiology and management of gastrointestinal dysmotility in systemic sclerosis (scleroderma). Aliment. Pharmacol. Ther. 23, 691–712 (2006).
Mercado, U., Arroyo de Anda, R., Avendano, L., Araiza-Casillas, R. & Avendano-Reyes, M. Metoclopramide response in patients with early diffuse systemic sclerosis. Effects on esophageal motility abnormalities. Clin. Exp. Rheumatol. 23, 685–688 (2005).
Wollheim, F. A. & Akesson, A. Management of intestinal involvement in systemic sclerosis. J. Clin. Rheumatol. 13, 116–118 (2007).
Tonini, M. et al. Review article: clinical implications of enteric and central D2 receptor blockade by antidopaminergic gastrointestinal prokinetics. Aliment. Pharmacol. Ther. 19, 379–390 (2004).
Heer, M. et al. Diabetic gastroparesis: treatment with domperidone—a double-blind, placebo-controlled trial. Digestion 27, 214–217 (1983).
Reddymasu, S. C., Soycan, I. & McCallum, R. W. Domperidone: review of pharmacology and clinical applications in gastroenterology. Am. J. Gastroenterol. 109, 2036–2045 (2007).
Sugumar, A., Singh, A. & Pasricha, P. J. A systematic review of the efficacy of domperidone for the treatment of diabetic gastroparesis. Clin. Gastroenterol. Hepatol. 6, 726–733 (2008).
Parkman, H. P. et al. Domperidone treatment for gastroparesis: demographic and pharmacogenetic characterization of clinical efficacy and side-effects. Dig. Dis. Sci. 56, 115–124 (2011).
Smith, D. S. & Ferris, C. D. Current concepts in diabetic gastroparesis. Drugs 63, 1339–1358 (2003).
Dhir, R. & Richter, J. E. Erythromycin in the short and long-term control of dyspepsia symptoms in patients with gastroparesis. J. Clin. Gastroenterol. 38, 237–242 (2004).
Perlemuter, G. et al. Octreotide treatment of chronic intestinal pseudo-obstruction secondary to connective tissue diseases. Arthritis Rheum. 42, 1545–1549 (1999).
Fynne, L. et al. Gastrointestinal transit in patients with systemic sclerosis. Scand. J. Gastroenterol. 46, 1187–1193 (2011).
Marie, I., Ducrotte, P., Denis, P., Menard, J. F. & Levesque, H. Small intestinal bacterial overgrowth in systemic sclerosis. Rheumatology (Oxford) 48, 1314–1319 (2009).
Parodi, A. et al. Small intestinal bacterial overgrowth in patients suffering from scleroderma: clinical effectiveness of its eradication. Am. J. Gastroenterol. 103, 1257–1262 (2008).
Corazza, G. R. et al. The diagnosis of small bowel bacterial overgrowth. Reliability of jejunal culture and inadequacy of breath hydrogen testing. Gastroenterology 98, 302–309 (1990).
Bures, J. et al. Small intestinal bacterial overgrowth syndrome. World J. Gastroenterol. 16, 2978–2990 (2010).
Simren, M. & Stotzer, P. O. Use and abuse of hydrogen breath tests. Gut 55, 297–303 (2006).
Lauritano. E. C. et al. Antibiotic therapy in small intestinal bacterial overgrowth: rifaximin versus metronidazole. Eur. Rev. Med. Pharmacol. Sci. 13, 111–116 (2009).
Harrison, E., Herrick, A. L., McLaughlin, J. T. & Lal, S. Malnutrition in systemic sclerosis. Rheumatology (Oxford) 51, 1747–1756 (2012).
Jawa, H., Fernandes, G., Saqui, O. & Allard, J. P. Home parenteral nutrition in patients with systemic sclerosis: a retrospective review of 12 cases. J. Rheumatol. 39, 1004–1007 (2012).
Baron, M., Hudson, M., Steele, R. & Canadian Scleroderma Research Group. Malnutrition is common in systemic sclerosis: results from the Canadian scleroderma research group database. J. Rheumatol. 36, 2737–2743 (2009).
Baron, M., Hudson, M., Steele, R. & Canadian Scleroderma Research Group (CSRG). Is serum albumin a marker of malnutrition in chronic disease? The scleroderma paradigm. J. Am. Coll. Nutr. 29, 144–151 (2010).
Brown, M., Teubner, A., Shaffer, J. & Herrick, A. L. Home parenteral nutrition—an effective and safe long-term therapy for systemic sclerosis-related intestinal failure. Rheumatology (Oxford) 47, 176–179 (2008).
Boulman, N., Slobodin, G., Rozenbaum, M. & Rosner, I. Calcinosis in rheumatic diseases. Semin. Arthritis Rheum. 34, 805–812 (2005).
Bair, B. & Fivenson, D. A novel treatment for ulcerative calcinosis cutis. J. Drugs Dermatol. 10, 1042–1044 (2011).
Auriemma, M. et al. Treatment of cutaneous calciphylaxis with sodium thiosulfate: two case reports and a review of the literature. Am. J. Clin. Dermatol. 12, 339–346 (2011).
Matsuoka, Y., Miyajima, S. & Okada, N. A case of calcinosis universalis successfully treated with low-dose warfarin. J. Dermatol. 25, 716–720 (1998).
Torralba, T. P., Li-Yu, J. & Navarra, S. T. Successful use of diltiazem in calcinosis caused by connective tissue disease. J. Clin. Rheumatol. 5, 74–78 (1999).
Palmieri, G. M. et al. Treatment of calcinosis with diltiazem. Arthritis Rheum. 38, 1646–1654 (1995).
Robertson, L. P., Marshall, R. W. & Hickling, P. Treatment of cutaneous calcinosis in limited systemic sclerosis with minocycline. Ann. Rheum. Dis. 62, 267–269 (2003).
Hazen, P. G., Walker, A. E., Carney, J. F. & Stewart, J. J. Cutaneous calcinosis of scleroderma. Successful treatment with intralesional adrenal steroids. Arch. Dermatol. 118, 366–367 (1982).
Ambler, G. R., Chaitow, J., Rogers, M., McDonald, D. W. & Ouvrier, R. A. Rapid improvement of calcinosis in juvenile dermatomyositis with alendronate therapy. J. Rheumatol. 32, 1837–1839 (2005).
Slimani, S., Abdessemed, A., Haddouche, A. & Ladjouze-Rezig, A. Complete resolution of universal calcinosis in a patient with juvenile dermatomyositis using pamidronate. Joint Bone Spine 77, 70–72 (2010).
Nakamura, H. et al. Efficacy of probenecid for a patient with juvenile dermatomyositis complicated with calcinosis. J. Rheumatol. 33, 1691–1693 (2006).
Eddy, M. C., Leelawattana, R., McAlister, W. H. & Whyte, M. P. Calcinosis universalis complicating juvenile dermatomyositis: resolution during probenecid therapy. J. Clin. Endocrinol. Metab. 82, 3536–3542 (1997).
Nakagawa, T. & Takaiwa, T. Calcinosis cutis in juvenile dermatomyositis responsive to aluminum hydroxide treatment. J. Dermatol. 20, 558–560 (1993).
Penate, Y. et al. Calcinosis cutis associated with amyopathic dermatomyositis: response to intravenous immunoglobulin. J. Am. Acad. Dermatol. 60, 1076–1077 (2009).
Daoussis, D., Antonopoulos, I., Liossis, S. N., Yiannopoulos, G. & Andonopoulos, A. P. Treatment of systemic sclerosis-associated calcinosis: a case report of rituximab-induced regression of CREST-related calcinosis and review of the literature. Semin. Arthritis Rheum. 41, 822–829 (2012).
Sultan-Bichat, N. et al. Treatment of calcinosis cutis by extracorporeal shock-wave lithotripsy. J. Am. Acad. Dermatol. 66, 424–429 (2012).
Shetty, S., Moore, T. L., Jackson, S., Brettle, D. & Herrick, A. L. A pilot study of acetic acid iontophoresis and ultrasound in the treatment of systemic sclerosis-related calcinosis. Rheumatology (Oxford) 44, 536–538 (2005).
Wieder, D. L. Treatment of traumatic myositis ossificans with acetic acid iontophoresis. Phys. Ther. 72, 133–137 (1992).
Abdallah-Lotf, M., Grasland, A., Vinceneux, P. & Sigal-Grinberg, M. Regression of cutis calcinosis with diltiazem in adult dermatomyositis. Eur. J. Dermatol. 15, 102–104 (2005).
Acknowledgements
The authors would like to thank the Division of Rheumatology at Johns Hopkins and the Johns Hopkins Scleroderma Center for their ongoing support.
Author information
Authors and Affiliations
Contributions
Both authors researched the data for the article, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
McMahan, Z., Hummers, L. Systemic sclerosis—challenges for clinical practice. Nat Rev Rheumatol 9, 90–100 (2013). https://doi.org/10.1038/nrrheum.2012.191
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2012.191
This article is cited by
-
Immune Checkpoint Inhibitors and Scleroderma: Data from the European Pharmacovigilance Database
Drugs - Real World Outcomes (2023)
-
Progress in Understanding, Diagnosing, and Managing Cardiac Complications of Systemic Sclerosis
Current Rheumatology Reports (2019)
-
Targeting of dermal myofibroblasts through death receptor 5 arrests fibrosis in mouse models of scleroderma
Nature Communications (2019)
-
Current management of the gastrointestinal complications of systemic sclerosis
Nature Reviews Gastroenterology & Hepatology (2016)
-
Management of Gastrointestinal Involvement in Scleroderma
Current Treatment Options in Rheumatology (2015)