Abstract
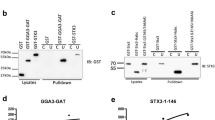
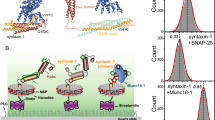
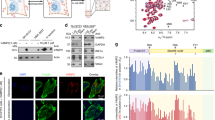
Syntaxins and Sec1/munc18 proteins are central to intracellular membrane fusion. All syntaxins comprise a variable N-terminal region, a conserved SNARE motif that is critical for SNARE complex formation, and a transmembrane region. The N-terminal region of neuronal syntaxin 1A contains a three-helix domain that folds back onto the SNARE motif forming a 'closed' conformation; this conformation is required for munc18-1 binding. We have examined the generality of the structural properties of syntaxins by NMR analysis of Vam3p, a yeast syntaxin essential for vacuolar fusion. Surprisingly, Vam3p also has an N-terminal three-helical domain despite lacking apparent sequence homology with syntaxin 1A in this region. However, Vam3p does not form a closed conformation and its N-terminal domain is not required for binding to the Sec1/munc18 protein Vps33p, suggesting that critical distinctions exist in the mechanisms used by syntaxins to govern different types of membrane fusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Bennett, M.K. & Scheller, R.H. The molecular machinery for secretion is conserved from yeast to neurons. Proc. Natl. Acad. Sci. USA 90, 2559–2563 (1993).
Ferro-Novick, S. & Jahn, R. Vesicle fusion from yeast to man. Nature 370, 191–193 (1994).
Jahn, R. & Südhof, T.C. Membrane fusion and exocytosis. Annu. Rev. Biochem. 68, 863–911 (1999).
Gerst, J.E. SNAREs and SNARE regulators in membrane fusion and exocytosis. Cell Mol. Life Sci. 55,707–734 (1999).
Bock, J.B. & Scheller, R.H. Protein transport. A fusion of new ideas. Nature 387,133–135 (1997).
Pelham, H.R. SNAREs and the secretory pathway; lessons from yeast. Exp. Cell. Res. 247, 1–8 (1999).
Südhof, T.C. The synaptic vesicle cycle: a cascade of protein-protein interactions. Nature 375, 645–653 (1995).
Bennett, M.K. & Scheller, R.H. A molecular description of synaptic vesicle membrane trafficking. Annu. Rev. Biochem. 63, 63–100 (1994).
Hata, Y., Slaughter, C.A. & Sudhof, T.C. Synaptic vesicle fusion complex contains unc-18 homologue bound to syntaxin. Nature 366, 347–351 (1993).
Sollner, T. et al. A protein assembly-disassembly pathway in vitro that may correspond to sequential steps of synaptic vesicle docking, activation, and fusion. Cell 75, 409–418 (1993).
Hayashi, T. et al. Synaptic vesicle membrane fusion complex: action of clostridial neurotoxins on assembly. EMBO J. 13, 5051–5061 (1994).
Poirier, M.A. et al. The synaptic SNARE complex is a parallel four-stranded helical bundle. Nature Struct. Biol . 5, 765–769 (1998).
Sutton, R.B., Fasshauer, D., Jahn, R. & Brunger, A.T. Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 Å resolution. Nature 395, 347–353 (1998).
Hanson, P.I., Roth, R., Morisaki, H., Jahn, R. & Heuser, J.E. Structure and conformational changes in NSF and its membrane receptor complexes visualized by quick-freeze/deep-etch electron microscopy. Cell 90, 523–535 (1997).
Lin, R.C. & Scheller, R.H. Structural organization of the synaptic exocytosis core complex. Neuron 19, 1087–1094 (1997).
Weber, T. et al. SNAREpins: minimal machinery for membrane fusion. Cell 92, 759–772 (1998).
Fernandez, I. et al. Three-dimensional structure of an evolutionarily conserved N-terminal domain of syntaxin 1A. Cell 94, 841–849 (1998).
Dulubova, I. et al. A conformational switch in syntaxin during exocytosis: role of munc18. EMBO J. 18, 4372–4382 (1999).
Misura, K.M., Scheller, R.H. & Weis, W.I. Three-dimensional structure of the neuronal-Sec1-syntaxin 1a complex. Nature 404, 355–362 (2000).
Fiebig, K.M., Rice, L.M., Pollock, E. & Brunger, A.T. Folding intermediates of SNARE complex assembly. Nature Struct. Biol. 6, 117–123 (1999).
Munson, M., Chen, X., Cocina, A. E., Schultz, S.M. & Hughson, F.M. Interactions with the yeast t-SNARE Sso1p that control SNARE complex assembly. Nature Struct. Biol. 7, 894–902 (2000).
Carr, C.M., Grote, E., Munson, M., Hughson, F.M. & Novick, P.J. Sec1p binds to SNARE complexes and concentrates at sites of secretion. J. Cell. Biol. 146, 333–344 (1999).
Darsow, T., Rieder, S.E. & Emr, S.D. A multispecificity syntaxin homologue, Vam3p, essential for autophagic and biosynthetic protein transport to the vacuole. J. Cell. Biol. 138, 517–529 (1997).
Nichols, B.J., Ungermann, C., Pelham, H.R., Wickner, W.T. & Haas, A. Homotypic vacuolar fusion mediated by t- and v-SNAREs. Nature 387, 199–202 (1997).
Wickner, W. & Haas, A. Yeast homotypic vacuole fusion: a window on organelle trafficking mechanisms. Annu. Rev. Biochem. 69, 247–275 (2000).
Sato, T. K., Rehling, P., Peterson, M.R. & Emr, S.D. Class C Vps protein complex regulates vacuolar SNARE pairing and is required for vesicle docking/fusion. Mol. Cell 6, 661–671 (2000).
Price, A., Seals, D., Wickner, W. & Ungermann, C. The docking step of yeast vacuole fusion requires the transfer of proteins from a cis-SNARE complex to a Rab/Ypt protein. J. Cell Biol. 148, 1231–1238 (2000).
Kee, Y., Lin, R.C., Hsu, S.-C. & Scheller, R.H. Distinct domains of syntaxin are required for synaptic vesicle fusion complex formation and dissociation. Neuron 14, 991–998 (1995).
Fasshauer, D., Bruns, D., Shen, B., Jahn, R. & Brunger, A.T. A structural change occurs upon binding of syntaxin to SNAP-25. J. Biol. Chem. 272, 4582–4590 (1997).
Calakos, N., Bennett, M.K., Peterson, K.E. & Scheller, R.H. Protein-protein interactions contributing to the specificity of intracellular vesicular trafficking. Science 263, 1146–1149 (1994).
Guan, K.L. & Dixon, J.E. Eucaryotic proteins expressed in Escherichia coli: an improved thrombin cleavage and purification procedure of fusion proteins with glutathione S-transferase. Anal. Biochem. 192, 262–267 (1991).
Hakes, D.J. & Dixon, J.E. New vectors for high level expression of recombinant proteins in bacteria. Anal. Biochem . 202, 293–298 (1992).
Ubach, J., Garcia, J., Nittler, M.P., Südhof, T.C. & Rizo, J. Structure of the Janus-faced C2B-domain of rabphilin. Nature Cell Biol. 1, 106–112 (1999).
Zhang, O., Kay, L.E., Olivier, J.P. & Forman-Kay, J. Backbone 1H and 15N resonance assignments of N-terminal SH3 domain of drk in folded and unfolded states using enhanced-sensitivity pulsed field gradient NMR techniques. J. Biomol. NMR 4, 845–858 (1994).
Kay, L.E., Xu, G.Y. & Yamazaki, T. Enhanced-sensitivity triple-resonance spectroscopy with minimal H2O saturation. J. Magn. Reson. A 109, 129–133 (1994).
Muhandiram, D.R. & Kay, L.E. Gradient-enhanced triple-resonance three-dimensional NMR experiments with improved sensitivity. J. Magn. Reson. B 103, 203–216 (1994).
Kay, L.E. Pulsed-field gradient-enhanced three-dimensional NMR experiment for correlating 13Cα/β, 13C′, and 1Hα chemical shifts in uniformly 13C-labeled proteins dissolved in H2O. J. Am. Chem. Soc. 115, 2055–2057 (1993).
Grzesiek, S., Anglister, J. & Bax, A. Correlation of backbone amide and aliphatic side-chain resonances in 13C/15N-enriched proteins by isotropic mixing of 13C magnetization. J. Magn. Reson. B 101, 114–119 (1993).
Kay, L.E., Xu, G.Y., Singer, A.U., Muhandiram, D.R. & Forman-Kay, J.D. A gradient-enhanced HCCH-TOCSY experiment for recording side-chain 1H and 13C correlations in H2O samples of proteins. J. Magn. Reson. B 101, 333–337 (1993).
Kuboniwa, H., Grzesiek, S., Delaglio, F. & Bax, A. Measurement of HN-Ha J couplings in calcium-free calmodulin using new 2D and 3D water-flip-back methods. J. Biomol. NMR 4, 871–878 (1994).
Delaglio, F., Grzesiek, S., Vuister, G.W., Zhu, G., Pfeifer, J. and Bax, A . NMRpipe: a multidimensional spectral processing system based on UNIX pipes. J. Biomol. NMR 6, 277–293 (1995).
Johnson, B.A. & Blevins, R.A. NMRView: a computer program for visualization and analysis of NMR data. J. Biomol. NMR 4, 603–614 (1994).
Brunger, A.T. et al. Crystallography and NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998).
Stack, J.H., Herman, Schu, P.V. & Emr, S.D. A membrane-associated complex containing the Vps15 protein kinase and the Vps34 PI 3-kinase is essential for protein sorting to the yeast lysosome-like vacuole. EMBO J. 12, 2195–2204 (1993).
Robinson, J.S., Klionsky, D.J., Banta, L.M. & Emr, S.D. Protein sorting in Saccharomyces cerevisiae: isolation of mutants defective in the delivery and processing of multiple vacuolar hydrolases. Mol. Cell. Biol. 8, 4936–4948 (1988).
Gietz, R.D., Schiestl, R.H., Willems, A.R. & Woods, R.A. Studies on the transformation of intact yeast cells by LiAc/ss-DNA/PEG procedure. Yeast 11, 355–360 (1995).
Haas, A. A quantitative assay to measure homotypic vacuole fusion in vitro. Methods Cell Sci. 17, 283–294 (1995).
Kraulis, P.J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991).
Nicholls, A. Sharp, K.A. & Honig, B. Protein folding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins 11, 281–296 (1991).
Laskowsky, R.A., MacArthur, M.W., Moss, D.S. & Thornton, J.M. PROCHECK: a program to check stereochemical quality of protein structure coordinates. J. Appl. Crystallogr. 26, 283–291 (1993).
Acknowledgements
We thank Y. Gao for technical assistance, B. Horazdovsky for reagents and fruitful discussions, and L. Kay for providing pulse sequences. This work was supported by an Established Investigator grant from the American Heart Association and by an NIH grant to JR.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dulubova, I., Yamaguchi, T., Wang, Y. et al. Vam3p structure reveals conserved and divergent properties of syntaxins. Nat Struct Mol Biol 8, 258–264 (2001). https://doi.org/10.1038/85012
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85012
This article is cited by
-
The Habc domain of syntaxin 3 is a ubiquitin binding domain
Scientific Reports (2020)
-
Munc18-1 is crucial to overcome the inhibition of synaptic vesicle fusion by αSNAP
Nature Communications (2019)
-
Chaperoning SNARE assembly and disassembly
Nature Reviews Molecular Cell Biology (2016)
-
The SM protein Vps33 and the t-SNARE Habc domain promote fusion pore opening
Nature Structural & Molecular Biology (2010)
-
The late stage of autophagy: cellular events and molecular regulation
Protein & Cell (2010)