Abstract
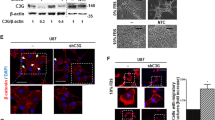
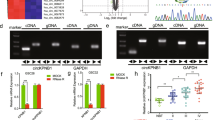
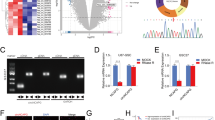
TRIM11 (tripartite motif-containing protein 11), an E3 ubiquitin ligase, is known to be involved in the development of the central nervous system. However, very little is known regarding the role of TRIM11 in cancer biology. Here, we examined the expression profile of TRIM11, along with two stem cell markers CD133 and nestin, in multiple glioma patient specimens, glioma primary cultures derived from tumors taken at surgery and normal neural stem/progenitor cells (NSCs). The oncogenic function of TRIM11 in glioma biology was investigated by knockdown and/or overexpression in vitro and in vivo experiments. Our results showed that TRIM11 expression levels were upregulated in malignant glioma specimens and in high-grade glioma-derived primary cultures, whereas remaining low in glioblastoma multiforme (GBM) stable cell lines, low-grade glioma-derived primary cultures and NSCs. The expression pattern of TRIM11 strongly correlated with that of CD133 and nestin and differentiation status of malignant glioma cells. Knock down of TRIM11 inhibited proliferation, migration and invasion of GBM cells, significantly decreased epidermal growth factor receptor (EGFR) levels and mitogen-activated protein kinase activity, and downregulated HB-EGF (heparin-binding EGF-like growth factor) mRNA levels. Meanwhile, TRIM11 overexpression promoted a stem-like phenotype in vitro (tumorsphere formation) and enhanced glial tumor growth in immunocompromised mice. These findings suggest that TRIM11 might be an indicator of glioma malignancy and has an oncogenic function mediated through the EGFR signaling pathway. TRIM11 overexpression potentially leads to a more aggressive glioma phenotype, along with increased malignant tumor growth and poor survival. Taken together, clarification of the biological function of TRIM11 and pathways it affects may provide novel therapeutic strategies for treating malignant glioma patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gladson CL, Prayson RA, Liu WM . The pathobiology of glioma tumors. Annu Rev Pathol 2010; 5: 33–50.
Wen PY, Kesari S . Malignant gliomas in adults. N Engl J Med 2008; 359: 492–507.
von Deimling A, Korshunov A, Hartmann C . The next generation of glioma biomarkers: MGMT methylation, BRAF fusions and IDH1 mutations. Brain Pathol 2011; 21: 74–87.
Ma YH, Mentlein R, Knerlich F, Kruse ML, Mehdorn HM, Held-Feindt J . Expression of stem cell markers in human astrocytomas of different WHO grades. J Neurooncol 2008; 86: 31–45.
Zhang M, Song T, Yang L, Chen R, Wu L, Yang Z et al. Nestin and CD133: valuable stem cell-specific markers for determining clinical outcome of glioma patients. J Exp Clin Cancer Res 2008; 27: 85.
Singh S, Clarke I, Terasaki M, Bonn V, Hawkins C, Squire J et al. Identification of a cancer stem cell in human brain tumors. Cancer Res 2003; 63: 5821–5828.
Singh S, Hawkins C, Clarke I, Squire J, Bayani J, Hide T et al. Identification of human brain tumour initiating cells. Nature 2004; 432: 396–401.
Chen J, Li Y, Yu TS, McKay RM, Burns DK, Kernie SG et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 2012; 488: 522–526.
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006; 444: 756–760.
Li Z, Wang H, Eyler CE, Hjelmeland AB, Rich JN . Turning cancer stem cells inside out: an exploration of glioma stem cell signaling pathways. J Biol Chem 2009; 284: 16705–16709.
Dey M, Ulasov IV, Tyler MA, Sonabend AM, Lesniak MS . Cancer stem cells: the final frontier for glioma virotherapy. Stem Cell Rev 2011; 7: 119–129.
Uchida N, Buck DW, He D, Reitsma MJ, Masek M, Phan TV et al. Direct isolation of human central nervous system stem cells. Proc Natl Acad Sci USA 2000; 97: 14720–14725.
Zimmerman L, Parr B, Lendahl U, Cunningham M, McKay R, Gavin B et al. Independent regulatory elements in the nestin gene direct transgene expression to neural stem cells or muscle precursors. Neuron 1994; 12: 11–24.
Gong X, Schwartz PH, Linskey ME, Bota DA . Neural stem/progenitors and glioma stem-like cells have differential sensitivity to chemotherapy. Neurology 2011; 76: 1126–1134.
Schmidinger M, Linzmayer L, Becherer A, Fazeny-Doemer B, Fakhrai N, Prayer D et al. Psychometric- and quality-of-life assessment in long-term glioblastoma survivors. J Neurooncol 2003; 63: 55–61.
Corn BW, Wang M, Fox S, Michalski J, Purdy J, Simpson J et al. Health related quality of life and cognitive status in patients with glioblastoma multiforme receiving escalating doses of conformal three dimensional radiation on RTOG 98-03. J Neurooncol 2009; 95: 247–257.
Reymond A, Meroni G, Fantozzi A, Merla G, Cairo S, Luzi L et al. The tripartite motif family identifies cell compartments. EMBO J 2001; 20: 2140–2151.
Saurin AJ, Borden KL, Boddy MN, Freemont PS . Does this have a familiar RING? Trends Biochem Sci 1996; 21: 208–214.
Borden KL . RING fingers and B-boxes: zinc-binding protein-protein interaction domains. Biochem Cell Biol 1998; 76: 351–358.
Hatakeyama S . TRIM proteins and cancer. Nat Rev Cancer 2011; 11: 792–804.
Niikura T, Hashimoto Y, Tajima H, Ishizaka M, Yamagishi Y, Kawasumi M et al. A tripartite motif protein TRIM11 binds and destabilizes Humanin, a neuroprotective peptide against Alzheimer’s disease-relevant insults. Eur J Neurosci 2003; 17: 1150–1158.
Ishikawa H, Tachikawa H, Miura Y, Takahashi N . TRIM11 binds to and destabilizes a key component of the activator-mediated cofactor complex (ARC105) through the ubiquitin-proteasome system. FEBS Lett 2006; 580: 4784–4792.
Tuoc TC, Stoykova A . Trim11 modulates the function of neurogenic transcription factor Pax6 through ubiquitin-proteosome system. Genes Dev 2008; 22: 1972–1986.
Hong SJ, Chae H, Lardaro T, Hong S, Kim KS . Trim11 increases expression of dopamine beta-hydroxylase gene by interacting with Phox2b. Biochem Biophys Res Commun 2008; 368: 650–655.
Sanai N, Alvarez-Buylla A, Berger MS . Neural stem cells and the origin of gliomas. N Engl J Med 2005; 353: 811–822.
Schwartz PH, Bryant PJ, Fuja TJ, Su H, O'Dowd DK, Klassen H . Isolation and characterization of neural progenitor cells from post-mortem human cortex. J Neurosci Res 2003; 74: 838–851.
Kurihara H, Zama A, Tamura M, Takeda J, Sasaki T, Takeuchi T . Glioma/glioblastoma-specific adenoviral gene expression using the nestin gene regulator. Gene Ther 2000; 7: 686–693.
Griguer CE, Oliva CR, Gobin E, Marcorelles P, Benos DJ, Lancaster JR et al. CD133 is a marker of bioenergetic stress in human glioma. PLoS One 2008; 3: e3655.
Zhou XD, Wang XY, Qu FJ, Zhong YH, Lu XD, Zhao P et al. Detection of cancer stem cells from the C6 glioma cell line. J Int Med Res 2009; 37: 503–510.
Lim DA, Cha S, Mayo MC, Chen MH, Keles E, VandenBerg S et al. Relationship of glioblastoma multiforme to neural stem cell regions predicts invasive and multifocal tumor phenotype. Neuro Oncol 2007; 9: 424–429.
Rao RD, Uhm JH, Krishnan S, James CD . Genetic and signaling pathway alterations in glioblastoma: relevance to novel targeted therapies. Front Biosci 2003; 8: e270–e280.
Roberts PJ, Der CJ . Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 2007; 26: 3291–3310.
Schlessinger J . Cell signaling by receptor tyrosine kinases. Cell 2000; 103: 211–225.
Eramo A, Ricci-Vitiani L, Zeuner A, Pallini R, Lotti F, Sette G et al. Chemotherapy resistance of glioblastoma stem cells. Cell Death Differ 2006; 13: 1238–1241.
Zhao C, Deng W, Gage FH . Mechanisms and functional implications of adult neurogenesis. Cell 2008; 132: 645–660.
Kordes C, Sawitza I, Muller-Marbach A, Ale-Agha N, Keitel V, Klonowski-Stumpe H et al. CD133+ hepatic stellate cells are progenitor cells. Biochem Biophys Res Commun 2007; 352: 410–417.
Sherman JH, Redpath GT, Redick JA, Purow BW, Laws ER, Jane JA et al. A novel fixative for immunofluorescence staining of CD133-positive glioblastoma stem cells. J Neurosci Methods 2011; 198: 99–102.
Fredens K, Dahl R, Venge P . An immunofluorescent method for a specific demonstration of granulocytes and some of their proteins (ECP and CCP). Histochemistry 1986; 84: 247–250.
Bigner SH, Humphrey PA, Wong AJ, Vogelstein B, Mark J, Friedman HS et al. Characterization of the epidermal growth factor receptor in human glioma cell lines and xenografts. Cancer Res 1990; 50: 8017–8022.
Raab G, Klagsbrun M . Heparin-binding EGF-like growth factor. Biochim Biophys Acta 1997; 1333: F179–F199.
Suo Z, Risberg B, Karlsson MG, Villman K, Skovlund E, Nesland JM . The expression of EGFR family ligands in breast carcinomas. Int J Surg Pathol 2002; 10: 91–99.
Tarbe N, Losch S, Burtscher H, Jarsch M, Weidle UH . Identification of rat pancreatic carcinoma genes associated with lymphogenous metastasis. Anticancer Res 2002; 22: 2015–2027.
Mishima K, Higashiyama S, Asai A, Yamaoka K, Nagashima Y, Taniguchi N et al. Heparin-binding epidermal growth factor-like growth factor stimulates mitogenic signaling and is highly expressed in human malignant gliomas. Acta Neuropathol 1998; 96: 322–328.
McCarthy SA, Samuels ML, Pritchard CA, Abraham JA, McMahon M . Rapid induction of heparin-binding epidermal growth factor/diphtheria toxin receptor expression by Raf and Ras oncogenes. Genes Dev 1995; 9: 1953–1964.
Ongusaha PP, Kwak JC, Zwible AJ, Macip S, Higashiyama S, Taniguchi N et al. HB-EGF is a potent inducer of tumor growth and angiogenesis. Cancer Res 2004; 64: 5283–5290.
Pistollato F, Chen H-L, Rood BR, Zhang H-Z, D'Avella D, Denaro L et al. Hypoxia and HIF1alpha repress the differentiative effects of BMPs in high-grade glioma. Stem Cells 2009; 27: 7–17.
Acknowledgements
We thank Xing Gong for the preparation of glioma primary cultures, Dr Ronald C Kim for the neuropathological diagnosis of tumor samples and Dr Abhishek Chaturbedi for the organization of the tumor list used in the study. This work was supported by research funds donated by Ralph and Suzanne Stern and the Community Foundation of Jewish Federation. This study was also supported, in part, by start-up funds to DAB from the University of California, Irvine and the UCI Cancer Center Award Number P30CA062203 from the National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Di, K., Linskey, M. & Bota, D. TRIM11 is overexpressed in high-grade gliomas and promotes proliferation, invasion, migration and glial tumor growth. Oncogene 32, 5038–5047 (2013). https://doi.org/10.1038/onc.2012.531
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.531
Keywords
This article is cited by
-
Tripartite motif-containing protein 11 (TRIM11): a novel weapon against Alzheimer’s disease
Signal Transduction and Targeted Therapy (2024)
-
TRIM8: a double-edged sword in glioblastoma with the power to heal or hurt
Cellular & Molecular Biology Letters (2023)
-
TRIM11, a new target of p53, facilitates the migration and invasion of nasopharyngeal carcinoma cells
Molecular Biology Reports (2023)
-
Therapeutic, diagnostic and prognostic values of TRIM proteins in prostate cancer
Pharmacological Reports (2023)
-
Expression of tripartite motif-containing 44 and its prognostic and clinicopathological value in human malignancies:a meta-analysis
BMC Cancer (2020)