Abstract
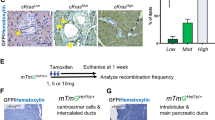
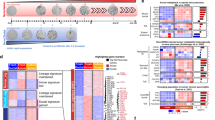
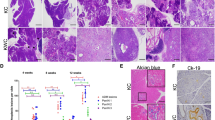
Pancreatic cancer is one of the most lethal malignancies, with virtually all patients eventually succumbing to their disease. Mutations in p53 have been documented in >50% of pancreatic cancers. Owing to the high incidence of p53 mutations in PanIN 3 lesions and pancreatic tumors, we interrogated the comparative ability of adult pancreatic acinar and ductal cells to respond to oncogenic Kras and mutant Tp53R172H using Hnf1b:CreERT2 and Mist1:CreERT2 mice. These studies involved co-activation of a membrane-tethered GFP lineage label, allowing for direct visualization and isolation of cells undergoing Kras and mutant p53 activation. Kras activation in Mist1+ adult acinar cells resulted in brisk PanIN formation, whereas no evidence of pancreatic neoplasia was observed for up to 6 months following Kras activation in Hnf1beta+ adult ductal cells. In contrast to the lack of response to oncogenic Kras alone, simultaneous activation of Kras and mutant p53 in adult ductal epithelium generated invasive PDAC in 75% of mice as early as 2.5 months after tamoxifen administration. These data demonstrate that pancreatic ductal cells, whereas exhibiting relative resistance to oncogenic Kras alone, can serve as an effective cell of origin for pancreatic ductal adenocarcinoma in the setting of gain-of-function mutations in p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Waddell N, Pajic M, Patch AM, Chang DK, Kassahn KS, Bailey P et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature 2015; 518: 495–501.
Kopp JL, von Figura G, Mayes E, Liu FF, Dubois CL, JPt Morris et al. Identification of Sox9-dependent acinar-to-ductal reprogramming as the principal mechanism for initiation of pancreatic ductal adenocarcinoma. Cancer Cell 2012; 22: 737–750.
Habbe N, Shi G, Meguid RA, Fendrich V, Esni F, Chen H et al. Spontaneous induction of murine pancreatic intraepithelial neoplasia (mPanIN) by acinar cell targeting of oncogenic Kras in adult mice. Proc Natl Acad Sci USA 2008; 105: 18913–18918.
Ji B, Tsou L, Wang H, Gaiser S, Chang DZ, Daniluk J et al. Ras activity levels control the development of pancreatic diseases. Gastroenterology 2009; 137: 82 e1–82 e6.
von Figura G, Fukuda A, Roy N, Liku ME, Morris IvJP, Kim GE et al. The chromatin regulator Brg1 suppresses formation of intraductal papillary mucinous neoplasm and pancreatic ductal adenocarcinoma. Nat Cell Biol 2014; 16: 255–267.
Guerra C, Schuhmacher AJ, Canamero M, Grippo PJ, Verdaguer L, Perez-Gallego L et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell 2007; 11: 291–302.
Bailey JM, Alsina J, Rasheed ZA, McAllister FM, Fu YY, Plentz R et al. DCLK1 marks a morphologically distinct subpopulation of cells with stem cell properties in preinvasive pancreatic cancer. Gastroenterology 2014; 146: 245–256.
Solar M, Cardalda C, Houbracken I, Martin M, Maestro MA, De Medts N et al. Pancreatic exocrine duct cells give rise to insulin-producing beta cells during embryogenesis but not after birth. Dev Cell 2009; 17: 849–860.
Strobel O, Dor Y, Alsina J, Stirman A, Lauwers G, Trainor A et al. In vivo lineage tracing defines the role of acinar-to-ductal transdifferentiation in inflammatory ductal metaplasia. Gastroenterology 2007; 133: 1999–2009.
Collisson EA, Trejo CL, Silva JM, Gu S, Korkola JE, Heiser LM et al. A central role for RAF—>MEK—>ERK signaling in the genesis of pancreatic ductal adenocarcinoma. Cancer Discov 2012; 2: 685–693.
Huang H, Daniluk J, Liu Y, Chu J, Li Z, Ji B et al. Oncogenic K-Ras requires activation for enhanced activity. Oncogene 2014; 33: 532–535.
Logsdon CD, Ji B . Ras activity in acinar cells links chronic pancreatitis and pancreatic cancer. Clin Gastroenterol 2009; 7 (11 Suppl): S40–S43.
Morton JP, Timpson P, Karim SA, Ridgway RA, Athineos D, Doyle B et al. Mutant p53 drives metastasis and overcomes growth arrest/senescence in pancreatic cancer. Proc Natl Acad Sci USA 2010; 107: 246–251.
Cooks T, Pateras IS, Tarcic O, Solomon H, Schetter AJ, Wilder S et al. Mutant p53 prolongs NF-kappaB activation and promotes chronic inflammation and inflammation-associated colorectal cancer. Cancer Cell 2013; 23: 634–646.
Coqueret O, Gascan H . Functional interaction of STAT3 transcription factor with the cell cycle inhibitor p21WAF1/CIP1/SDI1. J Biol Chem 2000; 275: 18794–18800.
Weissmueller S, Manchado E, Saborowski M, JPt Morris, Wagenblast E, Davis CA et al. Mutant p53 drives pancreatic cancer metastasis through cell-autonomous PDGF receptor beta signaling. Cell 2014; 157: 382–394.
Bailey JM, Alsina J, Rasheed ZA, McAllister FM, Fu YY, Plentz R et al. DCLK1 marks a morphologically distinct subpopulation of cells with stem cell properties in preinvasive pancreatic cancer. Gastroenterology 146: 245–256.
Siveke JT, Lubeseder-Martellato C, Lee M, Mazur PK, Nakhai H, Radtke F et al. Notch signaling is required for exocrine regeneration after acute pancreatitis. Gastroenterology 2008; 134: 544–555.
Acknowledgements
We acknowledge Anzer Habibulla, Danielle Blake, Mara Swaim, Jeffery Roeser and Qingfeng Zhu for their help in maintaining mouse colonies and genotyping. We thank Jorge Ferrer for providing the Hnf1b mice. This work was supported by NIH P01 CA134292 and R21 CA158898 (to SDL). JMB was supported by an NIH F32CA157044 and an AACR/Pancreatic Cancer Action Network Pathway to Leadership Award. NCJ was supported by an NIH T35 DK007676-22.
Author contributions
Conception and design, and writing the manuscript: JMB and SDL; acquisition of data: JMB, AMH, JA and AM; analysis and interpretation of data (statistical analysis, biostatistics and computational analysis): JMB, SDL, AMH, KJL, MY, CAI-D and AM; Administrative, technical or material support: JMB, AMH, MAP, NCJ.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Bailey, J., Hendley, A., Lafaro, K. et al. p53 mutations cooperate with oncogenic Kras to promote adenocarcinoma from pancreatic ductal cells. Oncogene 35, 4282–4288 (2016). https://doi.org/10.1038/onc.2015.441
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.441
This article is cited by
-
Senescence program and its reprogramming in pancreatic premalignancy
Cell Death & Disease (2023)
-
Intrapancreatic fat, pancreatitis, and pancreatic cancer
Cellular and Molecular Life Sciences (2023)
-
ZMAT1 acts as a tumor suppressor in pancreatic ductal adenocarcinoma by inducing SIRT3/p53 signaling pathway
Journal of Experimental & Clinical Cancer Research (2022)
-
Comprehensive microRNA analysis across genome-edited colorectal cancer organoid models reveals miR-24 as a candidate regulator of cell survival
BMC Genomics (2022)
-
Molecular signaling in pancreatic ductal metaplasia: emerging biomarkers for detection and intervention of early pancreatic cancer
Cellular Oncology (2022)