Abstract
Cardiovascular related deaths account for over 40% of the excess mortality in Turner syndrome (TS). Hypertension, a modifiable risk factor for both aortic dilatation and dissection, is more commonly encountered in TS during childhood and adolescence. Treatment of hypertension is currently recommended beyond the age of 16 years in TS to help reduce the risk of aortic dissection. This study aims to determine the prevalence of hypertension in paediatric patients with TS and explore the associated methodologies of blood pressure evaluation reported in these studies. Three online databases were searched (Medline, Embase and Web of Science) for literature which reported a prevalence, or allowed calculation of prevalence, of hypertension in patients with TS who were 18 years of age or younger. Seventeen studies which met the primary eligibility criteria, with a total of 1948 patients, were included. The estimated pooled prevalence of hypertension in children and adolescents with TS was 16% (95% CI: 8.9–24.6%). There was significant heterogeneity detected between the studies. The prevalence of hypertension in those studies which assessed 24-h Ambulatory Blood Pressure Monitoring (ABPM) was 21.1% (95% CI: 15.2–27.6%) compared those which used another method of blood pressure measurement which was 13.5% (95% CI: 5.2–24.4%). Given the impact of hypertension with long-term health outcomes and the reversibility of these same outcomes by addressing abnormal blood pressure, prompt and early diagnosis of hypertension in young girls with TS should be prioritised. We recommend the use of 24-h ABPM in screening for hypertension in the paediatric TS population.
Similar content being viewed by others
Introduction
Turner syndrome (TS) is a chromosomal disorder characterised by an abnormality of partial or complete absence of one of the X chromosomes in a phenotypic female. It affects ~1 in every 2000–2500 live born females. Whilst short stature and primary ovarian failure are characteristics of TS, a variety of health issues can present throughout the lifespan of a person with TS.
Congenital cardiac anomalies are present in ~50% of girls with TS, of which bicuspid aortic valve (20–30%), and coarctation of the aorta (4–12%), are most frequently reported [1, 2]. Aortic dilatation and aortic dissection can be attributed to the excess mortality seen in women with TS. The risk of aortic dissection in women with TS is up to 100-fold higher compared to the general population and is associated with a significantly high risk of mortality [3]. Aortic dissection in TS occurs at a much younger age in the second and third decade compared to non-TS related aortic dissection [4]. Whilst congenital anomalies in particular bicuspid aortic valves contribute to this excess risk of aortic dissection, non-congenital circulatory disease is also thought to play a role [5, 6]. Hypertension is often observed in girls and women with TS which is an important factor contributing to aortic dilatation, leading to aortic dissection [7]. Studies in adults with TS document a frequency of hypertension of up to 50% [8]. Of concern is that published research reports a frequency of hypertension in children and adolescents with TS of up to 40% [9,10,11] However, published studies in growing children with TS use a variety of different methods of assessment of blood pressure; and with many studies of small sample size.
The 2016 international clinical practice guideline for management of TS recommend annual monitoring of blood pressure and initiation of anti-hypertensives if hypertension is diagnosed in girls aged 16 years or older [9]. Clarifying the prevalence of hypertension in children and adolescents with TS is important to guide development of clinical pathways on monitoring and management of this important health issue in children and adolescents with TS. Both European and American paediatric guidelines (non-TS specific) recommend ambulatory blood pressure measurement (ABPM) before starting anti-hypertensive treatment in children [12]. The method of assessment for diagnosis of hypertension in children and adolescents with TS was not specified in the international clinical practice guideline for TS [9]. Therefore, when assessing the prevalence of hypertension in a paediatric population, it is also important to consider the method of blood pressure measurement.
The objective of this systematic review and meta-analysis is to determine the prevalence of hypertension in paediatric patients with TS who are 18 years of age or younger and explore the associated methodologies of blood pressure evaluation reported in these studies on the prevalence.
Methods
The systematic review of the literature was performed per the Preferred Reporting Items for Systematic Reviews and Meta-analysis guidelines [13].
Eligibility criteria
All studies which reported a prevalence or allowed calculation of prevalence of hypertension in children and adolescents with TS who were 18 years of age or younger were included in this review. We included studies reporting on all methods of blood pressure assessment and all definitions of hypertension which fulfilled the primary eligibility criteria. In papers which included adult subjects >18 years, if group data for subjects ≤18 years was able to be evaluated independently, this was included in our review. In longitudinal studies or studies which assessed any treatment in TS, only baseline blood pressure and hypertension data was used for purposes of our analysis. Papers that only provided group comparison of blood pressure to a control population were excluded. No publication date restrictions were imposed. Case reports, unpublished manuscripts, review articles and conference abstracts were excluded. Only studies reported in the English language were included in this review.
Information sources, search strategy and selection process
Two researchers (SM and AC) independently carried out the literature search in May 2021. Studies were identified by searching three online databases: Medline, Embase and Web of Science. Medline and Embase were searched through the Ovid interface. Broad search terms were used, which included the key terms ‘Turner Syndrome’ or ‘Turners’ and ‘Hypertension’ or ‘Blood pressure’. A decision was made not to include the terms ‘Paediatric’ or ‘Children’ in the search strategy to achieve a comprehensive search. Full details of the searches carried out can be found in the Supplementary Information provided.
Duplicates identified during the initial search were first removed. The title, abstract and keywords of the remaining studies were then independently screened by the two researchers (SM and AC). In addition, reference lists of relevant review articles identified were manually searched for any papers which met the eligibility criteria. After the potentially relevant studies were identified, full-text records of these studies were sought and then independently reviewed. All studies which then met the eligibility criteria above were included. Any disagreements were reviewed by a third researcher (AM) to reach a consensus.
Data collection progress and data items
A standardised data extraction proforma was designed and the relevant data was extracted independently from half the included studies by SM and the other half by AC. The extracted data were then crossed checked by the other reviewer to confirm accuracy of the data. The collected data included the title and authors of the study, year of publication, study design, age of subjects, sample size, method of blood pressure measurement, definition of hypertension and prevalence of hypertension. If more than one method of blood pressure assessment was included in the study, the prevalence data of hypertension were sought and reported for each method.
Study risk of bias assessment
A risk of bias tool designed for systematic reviews of prevalence studies was used [14]. The tool included ten domains which assessed both external and internal validity. We also provided a numerical score of the risk of bias based on an adapted tool as described by Ciona et al. [15]. A numerical score of ‘1’ was given if risk of bias was deemed low or ‘0’ if the risk was deemed high. A maximal numerical score out of 10 could be assigned for each paper which then classified the overall risk of study bias as low (>8), moderate [6,7,8] or high (≤5). If there was insufficient information in a paper to permit judgement for a particular domain, a score of ‘0’ was attributed to that item.
External validity items included: assessment of study’s target population, the sampling frame, assessment of random selection and non-response bias. Internal validity items included: Data collection (was the same mode used for all subjects and was the data collected directly from the subjects), assessment of case definition, the reliability and validity of the study instrument, the prevalence period and the appropriateness of numerator and denominator of the parameter of interest.
The risk of bias assessment was carried out independently by two researchers (SM and AC). Any disagreements were reviewed by a third researcher (AM) to reach a consensus.
Statistics
The primary outcome of this review was the pooled prevalence of hypertension in children and adolescents TS 18 years or younger. For each included study, the prevalence of hypertension stated within the study was included in the meta-analysis. For studies that reported more than one prevalence as more than one method of assessment of blood pressure was used in the same group of subjects, only one prevalence was included in the meta-analysis from that study.
For those studies, we selected the method of blood pressure measurement in the following preferential order: (1) 24-h ABPM, (2) Mercury sphygmomanometer, (3) Oscillometric device, (4) Data collection from clinical records, (5) Self-reported questionnaire.
Meta-analysis was conducted using a random-effects model. As some studies reported extremely low prevalence, we applied the Freeman-Tukey double arscine variance stabilising transformation to the raw prevalence data. The transformed summary proportions were then converted back to proportion estimates for reporting. These were then reported as prevalence percentages with 95% confidence intervals. The between-study variance heterogeneity of effect size estimates across the studies was assessed using the Q-test and the I2 statistic. It was assumed that an I2 of 25%, 50%, and 75% indicates low, medium, and large heterogeneity, respectively. Visual inspection of a forest plot was also carried out as well as identifying any outliers using leave-one-out diagnostic tests. Subgroup analysis was carried out using a mixed-effects model, by first fitting two separate random-effects models within each subgroup and then combining the estimated statistics from each model using a fixed-effect model to compare the two transformed proportions. A funnel plot was used to investigate publication bias with visual inspection and the Egger test to assess plot asymmetry. All analyses were performed using R Statistical Software (v4.1.3; R Core Team 2022) using the metafor (v3.0-2) and meta (v5.2-0) packages [16,17,18].
Results
Study selection
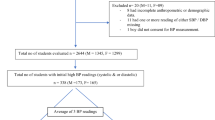
One thousand three hundred and seventy-four studies were identified through our initial searches. Eight hundred and fifty-one remaining studies were then screened using the title, abstract and keywords. A total of 127 full texts were reviewed to assess eligibility criteria. Forty of these were conference abstracts which were excluded.
After reviewing the remaining 87 full texts, 17 studies were included which met primary eligibility criteria. The main reason for exclusion was the inclusion of TS patients >18 years of age or no data to allow for evaluation of prevalence of hypertension in TS. A flowchart detailing this selection process is shown in Fig. 1.
Study characteristics
A total of 1948 children and adolescents with TS were included in the final 17 studies. Sample size in the included studies ranged from 15 to 842 subjects. The included studies were published between 1992–2020. A detailed summary of study characteristics is included in Table 1.
Two out of 17 (11.8%) studies were prospective longitudinal cohort studies [19, 20], four (23.5%) were retrospective cohort studies of clinical evaluation [21,22,23,24], six were prospective case-control studies (35.3%) [25,26,27,28,29,30], four (23.5%) were prospective cross-sectional studies [31,32,33,34] and one (5.9%) was a multicentre randomised dose-response study of growth hormone treatment [35].
Five (29.4%) out of the studies used more than one method of assessment of blood pressure [25, 27, 29, 33, 34]. Five studies reported 24-h ABPM [19, 25, 27, 32, 33]. Four studies used a mercury sphygmomanometer as the method of assessment of blood pressure [20, 25, 28, 33]. Five used an oscillometric device [26, 27, 31, 33, 34]. Three studies used data from clinical records [21,22,23] and two used questionnaires in their assessment [29, 34]. Three studies did not state the method of blood pressure assessment in their methodology [24, 30, 35]. Of the five studies which used more than one method of blood pressure assessment, one study reported only one out of two of their methods of assessment [29].
Five studies did not report the definition of hypertension used in their methodology [20,21,22,23, 35]. In those which assessed ABPM, three studies used a definition of mean ambulatory BP > 95th centile based on AHA recommendations using normative data adapted from Wuhl et al. [25, 27, 33, 36, 37]. One study defined hypertension if the 24-h mean BP exceeded the upper limit of the standard values in >30% of ABPM measurements using 1997 ABPM standard values [32, 38]. One study reported hypertension as determined by height-based standards of ABPM but gave no further detail on this [19]. The remaining studies reported BP assessment using mercury sphygmomanometer, oscillometric device or an undefined method used a definition of hypertension as ≥ the 95th percentile for age, sex, and height [24,25,26,27,28,29,30,31, 33, 34].
Risk of bias in studies
A risk of bias assessment for each included study is shown in Table 2. All included studies were classified to be at moderate (76.5%) or high (23.5%) risk of bias based on the assessment of internal and external validity items. No study included in our review reported a form of random selection or census used to select the sample or provided an analysis comparing responders and non-responders. In three studies, it could not be determined whether blood pressure was collected directly from the subjects as measurements were taken from online health records [21,22,23]. Five studies provided no case definition for hypertension [20,21,22,23, 35] and one study was deemed to have an unacceptable case definition with not enough information provided [19]. Eight studies either did not specify the instrument used for blood pressure assessment or did not provide sufficient information for assessment of reliability and validity of the method blood pressure assessment [21,22,23,24, 29,30,31, 35].
Results of individual studies
The overall prevalence of hypertension of the studies reviewed ranged from 0–44%. The prevalence of hypertension with the associated 95% confidence interval for each individual study is shown in Table 1. In those studies which assessed 24-h ABPM, the prevalence ranged from 13.8–36.4%. In those which used a mercury sphygmomanometer, the prevalence ranged from 4.3–27.6%. Prevalence in studies which used an oscillometric device was 0–44%.
Pooled prevalence estimate
The estimated pooled prevalence of hypertension in children and adolescents with TS using a random-effects model was 16% (95% CI: 8.9–24.6%). This is shown in the Forest plot in Fig. 2. There was significant heterogeneity detected between the studies. I2 was 94.32% and Q-test for heterogeneity was statistically significant at 379.7 (p < 0.0001). A visual inspection of summary proportions leaving out a study at a time revealed no outliers. This was confirmed with a series of leave-1-out diagnostic tests which revealed no influential studies. Funnel plot shown in Fig. 3 demonstrated no asymmetry and p value for Egger’s test was not significant (p = 0.3123), suggesting no publication bias was detected.
Each dot represents a separate study. The double-arscine transformed proportion of children and adolescents with Turner syndrome <18 years with hypertension relative to the total sample size (as a measure of prevalence) in each individual study on the x-axis plotted against its standard error (as a measure of study precision) on the y-axis. Visual inspection suggested no asymmetry.
Subgroup analysis
Subgroup analysis was performed due to significant heterogeneity detected between the studies. This is shown in Table 3. There was a noteworthy difference between subgroups in those studies with a high risk of bias compared with those with a moderate risk of bias. No differences were noted with method of blood pressure assessment or year of publication.
According to method of blood pressure measurement, the prevalence of hypertension in those studies which assessed 24-h ABPM was 21.1% (15.2–27.6). Those which used another method of blood pressure measurement, detailed above, showed a prevalence of 13.5% (5.2–24.4%). According to the risk of bias noted in the studies, the prevalence of hypertension in those studies with a moderate risk of bias was 21.9% (95% CI: 12.7–31.4%). Those with a high risk of bias showed a prevalence of 2.7% (95% CI 0.9–8.6%). This difference was noted to be statistically significant (p = 0.002). The prevalence of hypertension in studies carried out in the last 10 years (after 2012) was 18.4% (95% CI: 9.9–28.7%), compared with those before 2012 which showed a prevalence of 10.7% (95% CI: 0.6–28.4%).
Discussion
This systematic review and meta-analysis showed a 16% prevalence of hypertension in children and adolescents with TS and is notably higher than estimates of childhood hypertension world-wide, thought to be ~4% [39]. Our subgroup analysis identified a prevalence of hypertension of 21.2% in TS in studies which used 24-h ABPM as the method of assessment of blood pressure. Previous studies which have compared blood pressure in children with TS to a control population have also shown higher blood pressures in these girls from a young age [40]. Therefore, greater focus should now be given on early accurate identification and management of hypertension in children and adolescents with TS.
The underlying pathogenesis of hypertension in TS is multifactorial with impaired vagal tone, congenital cardiovascular and renal anomalies, higher cortisol levels, impaired insulin resistance and the metabolic syndrome in the context of obesity all considered to be potential contributing factors [41]. In addition, oestrogen deficiency is felt to contribute in part to this and studies in adult patients have shown a beneficial effect on blood pressure with sex hormone replacement [42, 43]. However, a recent study in young adults with TS showed that despite effective oestrogen replacement, blood pressure increased significantly during adolescence and young adulthood [44]. The cause of this increase in blood pressure early in life in TS is still unknown.
Not only has hypertension been identified as a risk factor for aortic dissection in TS but the end-organ effects of hypertension including cardiovascular disease, cerebrovascular damage and nephropathy are well documented in the general population [45]. Children with TS have also been shown to have additional risk factors for cardiovascular morbidity with vasculopathy detected as early as 9 years of age [46]. Thus, further research in understanding the underlying aetiology of hypertension in young girls with TS may help in the development of effective management strategies.
It is well known that practical difficulties in clinic blood pressure measurement in children can lead to both under- and over- estimation of blood pressure readings [47]. Due to these difficulties, 24-h ABPM gives a more accurate representation of assessment of hypertension in a paediatric population [48]. This is also supported by two studies in our review who identified girls with hypertension with 24-h ABPM that were not picked up by resting BP alone [27, 33]. Our subgroup analysis shows that the pick-up with 24-h ABPM may be higher, with the prevalence of hypertension in those studies that assessed 24-h ABPM being 21.1% compared to 13.5% in those studies which did not assess 24-h ABPM. Studies which have assessed ABPM in a young TS population have also shown a lack of normal diurnal variation with less than the normal 10% reduction in nocturnal systolic blood pressure in up to 57% of patients [10, 11]. Lack of this normal mechanism, termed ‘non-dipper’, has been associated with increased morbidity and is likely to play a part in the end-organ damage, including left ventricular hypertrophy observed in ‘normotensive’ women with TS [49, 50].
The 2016 international clinical practice guideline suggests individuals with TS should have monitoring of blood pressure on an annual basis but no further detail on how this monitoring should be carried out is provided, especially for children and adolescents. Anxiety disorders are common in girls with TS [51]; and clinic measured blood pressure may not allow differentiation between true hypertension and white coat hypertension. Studies which have assessed the natural history of hypertension in a TS population have shown an increased prevalence of hypertension with age [41, 52]. To the best of our knowledge, there are no published longitudinal studies of blood pressure monitoring using 24-h ABPM in children and adolescents with TS. There is evidence in adults (although not specific to the TS population) that aggressive treatment of blood pressure not only stops but can reverse end organ damage [45]. We therefore suggest that 24-h ABPM should be gold standard for monitoring in adolescents with TS from the age of 10–12 years. Early treatment with anti-hypertensives should be initiated in TS girls (even in those <16 years) identified as hypertensive with 24-h ABPM. In normotensive girls identified through ABPM to be non-dippers, closer monitoring of blood pressure should be initiated. Whilst out of the scope of the current review, we also recommend greater focus on more intensive lifestyle modification (physical activity and nutrition) in these young growing girls with abnormal blood pressure. A suggested pathway for monitoring, diagnosis, and treatment of hypertension in children and adolescents with TS in shown in Fig. 4. There is some evidence that the renin-aldosterone-angiotensin system and sympathetic nervous system appear to be altered in TS and therefore we suggest hypertensive agents targeting these systems may have a beneficial role in treatment of hypertension in these patients [42, 53, 54]. However, we acknowledge there is little existing evidence assessing management of hypertension in children and adolescents and further systematic reviews will be required to explore this further.
Our study has a number of limitations which should be considered. The pooled prevalence of hypertension should be interpreted with some caution, considering the large heterogeneity between the studies. Many of the studies included in our review had small sample sizes and all studies had a moderate to high risk of bias. We identified a number of studies which included subjects up to the age of 22 which were not included in our study due to the age restrictions we set at the beginning of the review process to ensure the results were generalisable to a purely paediatric TS population [10, 11, 55]. Whilst these limitations should be considered, it is important to note that this review presents the current published data on the prevalence of this important health condition in a young TS population.
There is a distinct need for more high-quality observational studies assessing the natural history of hypertension in a paediatric TS population with clear reporting of methodology, definitions, and confounding factors. Such studies are needed to develop evidence based clinical guidelines of monitoring and management in young growing children with TS, with the goal of reducing the risk of long-term adverse cardiovascular outcomes including aortic dissection.
Conclusion
To our knowledge, this is the first systematic review and meta-analysis evaluating the prevalence of hypertension in a paediatric TS population. Given the impact of hypertension with long-term health outcomes and the reversibility of these same health risks achieved by addressing abnormal blood pressure, prompt and early diagnosis of hypertension in young girls with TS should be prioritised.
Data availability
Data generated or analysed during this study are included in this published article and its Supplementary Information files. Any further data can be made available from the corresponding author on reasonable request.
References
Ho VB, Bakalov VK, Cooley M, Van PL, Hood MN, Burklow TR, et al. Major vascular anomalies in Turner syndrome: prevalence and magnetic resonance angiographic features. Circulation. 2004;110:1694–700.
Völkl TMK, Degenhardt K, Koch A, Simm D, Dörr HG, Singer H. Cardiovascular anomalies in children and young adults with Ullrich-Turner syndrome the Erlangen experience. Clin Cardiol. 2005;28:88–92.
Bondy CA. Aortic dissection in Turner syndrome. Curr Opin Cardiol. 2008;23:519–26.
Carlson M, Silberbach M. Dissection of the aorta in Turner syndrome: two cases and review of 85 cases in the literature. J Med Genet. 2007;44:745–9.
Schoemaker MJ, Swerdlow AJ, Higgins CD, Wright AF, Jacobs PA. Mortality in women with turner syndrome in Great Britain: a national cohort study. J Clin Endocrinol Metab. 2008;93:4735–42.
Stochholm K, Juul S, Juel K, Naeraa RW, Gravholt CH. Prevalence, incidence, diagnostic delay, and mortality in Turner syndrome. J Clin Endocrinol Metab. 2006;91:3897–902.
De Groote K, Demulier L, De Backer J, De Wolf D, De Schepper J, Tʼsjoen G, et al. Arterial hypertension in Turner syndrome: a review of the literature and a practical approach for diagnosis and treatment. J Hypertens. 2015;33:1342–51.
Elsheikh M, Casadei B, Conway GS, Wass JAH. Hypertension is a major risk factor for aortic root dilatation in women with Turner’s syndrome. Clin Endocrinol. 2001;54:69–73.
Gravholt CH, Andersen NH, Conway GS, Dekkers OM, Geffner ME, Klein KO, et al. Clinical practice guidelines for the care of girls and women with Turner syndrome: proceedings from the 2016 Cincinnati International Turner Syndrome Meeting. Eur J Endocrinol. 2017;177:G1–70.
Nathwani NC, Unwin R, Brook CG, Hindmarsh PC. Blood pressure and Turner syndrome. Clin Endocrinol. 2000;52:363–70.
Nathwani NC, Unwin R, Brook CG, Hindmarsh PC. The influence of renal and cardiovascular abnormalities on blood pressure in Turner syndrome. Clin Endocrinol. 2000;52:371–7.
Lurbe E, Agabiti-Rosei E, Cruickshank JK, Dominiczak A, Erdine S, Hirth A, et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens. 2016;34:1887–920.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Hoy D, Brooks P, Woolf A, Blyth F, March L, Bain C, et al. Assessing risk of bias in prevalence studies: modification of an existing tool and evidence of interrater agreement. J Clin Epidemiol. 2012;65:934–9.
Cioana M, Deng J, Hou M, Nadarajah A, Qiu Y, Chen SSJ, et al. Prevalence of hypertension and albuminuria in pediatric type 2 diabetes: a systematic review and meta-analysis. JAMA Netw Open. 2021;4:e216069.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Soft. 2010;36:1–48.
Wang N. How to conduct a meta-analysis of proportions in R: a comprehensive tutorial. 2018.
Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health. 2019;22:153–60.
McCarthy K, Bondy CA. Turner syndrome in childhood and adolescence. Expert Rev Endocrinol Metab. 2008;3:771–5.
Price DA, Clayton PE, Crowne EH, Roberts CR. Safety and efficacy of human growth hormone treatment in girls with Turner syndrome. Horm Res. 1993;39(Suppl 2):44–8.
Valencia E, Serna LM, Betancur LM, Medina CM, Arango Á. Lipid profile in a group of patients with Turner’s syndrome at Clínica Universitaria Bolivariana in the Medellín city between 2000 and 2009. Colomb Méd. 2011;42:54–60.
Yeşilkaya E, Bereket A, Darendeliler F, Baş F, Poyrazoğlu Ş, Küçükemre Aydın B, et al. Turner syndrome and associated problems in Turkish children: a multicenter study. J Clin Res Pediatr Endocrinol. 2015;7:27–36.
Hamberis AO, Mehta CH, Dornhoffer JR, Meyer TA. Characteristics and progression of hearing loss in children with turner’s syndrome. Laryngoscope. 2020;130:1540–6.
Lebenthal Y, Levy S, Sofrin-Drucker E, Nagelberg N, Weintrob N, Shalitin S, et al. The natural history of metabolic comorbidities in Turner syndrome from childhood to early adulthood: comparison between 45,X monosomy and other karyotypes. Front Endocrinol. 2018;9:27.
Akyürek N, Atabek ME, Eklioglu BS, Alp H. Ambulatory blood pressure and subclinical cardiovascular disease in children with turner syndrome. Pediatr Cardiol. 2014;35:57–62.
An HS, Baek JS, Kim GB, Lee YA, Song MK, Kwon BS, et al. Impaired vascular function of the aorta in adolescents with Turner syndrome. Pediatr Cardiol. 2017;38:20–6.
De Groote K, Devos D, Van Herck K, De Wolf D, Van der Straaten S, Rietzschel E, et al. Increased aortic stiffness in prepubertal girls with Turner syndrome. J Cardiol. 2017;69:201–7.
Akyürek N, Atabek ME, Eklioglu BS, Alp H. The relationship of periaortic fat thickness and cardiovascular risk factors in children with Turner syndrome. Pediatr Cardiol. 2015;36:925–9.
Lopez L, Arheart KL, Colan SD, Stein NS, Lopez-Mitnik G, Lin AE, et al. Turner syndrome is an independent risk factor for aortic dilation in the young. Pediatrics. 2008;121:1622.
Zhang Y, Chen R, Lin X, Yuan X, Yang X. The correlation between serum adipokines levels and metabolic indicators in girls with Turner syndrome. Cytokine. 2019;113:139–43.
Quezada E, Lapidus J, Shaughnessy R, Chen Z, Silberbach M. Aortic dimensions in Turner syndrome. Am J Med Genet A. 2015;167A:2527–32.
Tahhan N, Mura T, Jeandel C, Guillaumont S, De La Villeon G, Vincenti M, et al. Evaluation of cardiac MRI and ambulatory blood pressure monitoring in a pediatric Turner syndrome population. Prog Pediatr Cardiol. 2019;52:18–21.
Fudge EB, Constantacos C, Fudge JC, Davenport M. Improving detection of hypertension in girls with Turner syndrome using ambulatory blood pressure monitoring. HRP. 2014;81:25–31.
Los E, Quezada E, Chen Z, Lapidus J, Silberbach M. Pilot study of blood pressure in girls With Turner syndrome. Hypertension. 2016;68:133–6.
Sas TC, Cromme-Dijkhuis AH, de Muinck Keizer-Schrama SM, Stijnen T, van Teunenbroek A, Drop SL. The effects of long-term growth hormone treatment on cardiac left ventricular dimensions and blood pressure in girls with Turner’s syndrome. Dutch Working Group on Growth Hormone. J Pediatr. 1999;135:470–6.
Wühl E, Witte K, Soergel M, Mehls O, Schaefer F. Distribution of 24-h ambulatory blood pressure in children: normalized reference values and role of body dimensions. J Hypertens. 2002;20:1995–2007.
Urbina E, Alpert B, Flynn J, Hayman L, Harshfield GA, Jacobson M, et al. Ambulatory blood pressure monitoring in children and adolescents: recommendations for standard assessment. Hypertension. 2008;52:433–51.
Soergel M, Kirschstein M, Busch C, Danne T, Gellermann J, Holl R, et al. Oscillometric twenty-four-hour ambulatory blood pressure values in healthy children and adolescents: a multicenter trial including 1141 subjects. J Pediatr. 1997;130:178–84.
Song P, Zhang Y, Yu J, Zha M, Zhu Y, Rahimi K, et al. Global prevalence of hypertension in children: a systematic review and meta-analysis. JAMA Pediatr. 2019;173:1154–63.
O’Gorman CS, Syme C, Lang J, Bradley TJ, Wells GD, Hamilton JK. An evaluation of early cardiometabolic risk factors in children and adolescents with Turner syndrome. Clin Endocrinol. 2013;78:907–13.
Sandahl K, Wen J, Erlandsen M, Andersen NH, Gravholt CH. Natural history of hypertension in Turner syndrome during a 12-year pragmatic interventional study. Hypertension. 2020;76:1608–15.
Gravholt CH, Hansen KW, Erlandsen M, Ebbehøj E, Christiansen JS. Nocturnal hypertension and impaired sympathovagal tone in Turner syndrome. J Hypertens. 2006;24:353–60.
Gravholt CH, Naeraa RW, Nyholm B, Gerdes LU, Christiansen E, Schmitz O, et al. Glucose metabolism, lipid metabolism, and cardiovascular risk factors in adult Turner’s syndrome. The impact of sex hormone replacement. Diabetes Care. 1998;21:1062–70.
Brun S, Cleemann L, Holm K, Salskov G, Erlandsen M, Berglund A, et al. Five-year randomized study demonstrates blood pressure increases in young women with Turner syndrome regardless of estradiol dose. Hypertension. 2019;73:242–8.
Schmieder RE. End organ damage in hypertension. Dtsch Arztebl Int. 2010;107:866–73.
Lawson SA, Urbina EM, Gutmark-Little I, Khoury PR, Gao Z, Backeljauw PF. Vasculopathy in the young Turner syndrome population. J Clin Endocrinol Metab. 2014;99:E2039–45.
Goonasekera CDA, Dillon MJ. Measurement and interpretation of blood pressure. Arch Dis Child. 2000;82:261–5.
Stergiou GS, Boubouchairopoulou N, Kollias A. Accuracy of automated blood pressure measurement in children: evidence, issues, and perspectives. Hypertension. 2017;69:1000–6.
Andersen NH, Hjerrild BE, Sørensen K, Pedersen EM, Stochholm K, Gormsen LC, et al. Subclinical left ventricular dysfunction in normotensive women with Turner’s syndrome. Heart. 2006;92:1516–7.
Sozen AB, Cefle K, Kudat H, Ozturk S, Oflaz H, Akkaya V, et al. Left ventricular thickness is increased in nonhypertensive Turner’s syndrome. Echocardiography. 2009;26:943–9.
Kiliç BG, Ergür AT, Ocal G. Depression, levels of anxiety and self-concept in girls with Turner’s syndrome. J Pediatr Endocrinol Metab. 2005;18:1111–7.
Fiot E, Zénaty D, Boizeau P, Haignere J, Dos Santos S, Léger J. X chromosome gene dosage as a determinant of congenital malformations and of age-related comorbidity risk in patients with Turner syndrome, from childhood to early adulthood. Eur J Endocrinol. 2019;180:397–406.
Virdis R, Cantu MC, Ghizzoni L, Ammenti A, Nori G, Volta C, et al. Blood pressure behaviour and control in Turner syndrome. Clin Exp Hypertens A. 1986;8:787–91.
Zuckerman-Levin N, Zinder O, Greenberg A, Levin M, Jacob G, Hochberg Z. Physiological and catecholamine response to sympathetic stimulation in turner syndrome. Clin Endocrinol. 2006;64:410–5.
Lee SH, Jung JM, Song MS, Choi SJ, Chung WY. Evaluation of cardiovascular anomalies in patients with asymptomatic turner syndrome using multidetector computed tomography. J Korean Med Sci. 2013;28:1169–73.
Author information
Authors and Affiliations
Contributions
AC, SM, AM and SCW contributed to the conception and design of this study; AC and SM carried out the search and identification of relevant studies for the systematic review; AM resolved any disagreements; SM carried out analysis and interpretation of data and drafted the manuscript; SM and AC designed the tables and figures; AM and SCW critically revised the manuscript for important intellectual content and supervised the study. All authors have read and approved the final version of the manuscript and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. AM has full access to the data in the study and final responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McCarrison, S., Carr, A., Wong, S.C. et al. The prevalence of hypertension in paediatric Turner syndrome: a systematic review and meta-analysis. J Hum Hypertens 37, 675–688 (2023). https://doi.org/10.1038/s41371-022-00777-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-022-00777-8
This article is cited by
-
Journal of Human Hypertension special issue on sex and gender differences in hypertension
Journal of Human Hypertension (2023)