Abstract
Objective
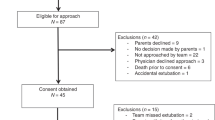
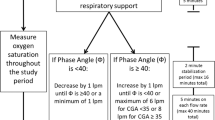
To measure tidal volume delivery during nasal intermittent positive pressure ventilation with two nasal interfaces: infant cannula and nasal prongs.
Study design
A single-center crossover study of neonates with mild respiratory distress. Fifteen preterm neonates were randomized to initial interface of infant cannula or nasal prongs and monitored on a sequence of pressure settings first on the initial interface, then repeated on the alternate interface. We compared relative tidal volumes between the two interfaces with two-way repeated measures ANOVA during three breath types: synchronized (I), patient effort without ventilator breaths (II), and ventilator breaths without patient effort (III). Clinical trial #NCT04326270.
Results
Type III breaths delivered no significant tidal volume. No significant difference was measured in relative tidal volume delivery between the interfaces when breath types were matched.
Conclusions
Nasal intermittent positive pressure ventilation delivers neither clinically nor statistically significant tidal volume with either infant cannula or nasal prongs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data from the results of this study are available from the corresponding author on reasonable request.
References
Miller JD, Carlo WA. Pulmonary complications of mechanical ventilation in neonates. Clin Perinatol. 2008;35:273–81, x-xi.
The STOP-ROP Multicenter Study Group. Supplemental therapeutic oxygen for prethreshold retinopathy of prematurity (STOP-ROP), a randomized, controlled trial. I: Primary outcomes. Pediatrics. 2000;105:295–310.
Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;163:1723–9.
Lemyre B, Davis PG, De Paoli AG, Kirpalani H. Nasal intermittent positive pressure ventilation (NIPPV) versus nasal continuous positive airway pressure (NCPAP) for preterm neonates after extubation. Cochrane Database Syst Rev. 2017;2:CD003212.
Meneses J, Bhandari V, Alves JG. Nasal intermittent positive-pressure ventilation vs. nasal continuous positive airway pressure for preterm infants with respiratory distress syndrome. Arch Pediatr Adolesc Med. 2012;166:372–6.
Rojas-Reyes MX, Lozano JM, Solà I, Soll R Overview of ventilation strategies for the early management of intubated preterm infants. Cochrane Database Syst Rev. 2015, Issue 4. Art. No.: CD011663. https://doi.org/10.1002/14651858.CD011663.
Dumpa V, Northrup V, Bhandari V. Type and timing of ventilation in the first post-natal week is associated with bronchopulmonary dysplasia/death. Am J Perinatol. 2011;28:321–30.
Bhandari V, Gavino RG, Nedrelow JH, Pallela P, Salvador A, Ehrenkranz RA, et al. A randomized controlled trial of synchronized nasal intermittent positive pressure ventilation in RDS. J Perinatol. 2007;27:697–703.
Iyer NP, Chatburn R. Evaluation of a nasal cannula in noninvasive ventilation using a lung simulator. Respir Care. 2015;60:508–12.
Mukerji A, Belik J. Neonatal nasal intermittent positive pressure ventilation efficacy and lung pressure transmission. J Perinatol. 2015;35:716–9.
Owen LS, Morley CJ, Dawson JA, Davis PG. Effects of non-synchronised nasal intermittent positive pressure ventilation on spontaneous breathing in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2011;96:F422–F428.
Silveira CST, Leonardi KM, Melo AP, Zaia JE, Brunherotti MAA. Response of preterm infants to 2 noninvasive ventilatory support systems: nasal CPAP and nasal intermittent positive-pressure ventilation. Respir Care. 2015;60:1772–6.
Kirpalani H, Millar D, Lemyre B, Yoder BA, Chiu A, Roberts RS, et al. A trial comparing noninvasive ventilation strategies in preterm infants. N. Engl J Med. 2013;369:611–20.
Owen LS, Morley CJ, Davis PG. Pressure variation during ventilator generated nasal intermittent positive pressure ventilation in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2010;95:F359–F364.
Matlock DN, Bai S, Weisner MD, Comtois N, Beck J, Sinderby C, et al. Tidal Volume Transmission During Non-Synchronized Nasal Intermittent Positive Pressure Ventilation via RAM® Cannula. J Perinatol. 2019;39:723–9.
Robertson NJ, McCarthy LS, Hamilton PA, Moss ALH. Nasal deformities resulting from flow driver continuous positive airway pressure. Arch Dis Child Fetal Neonatal Ed. 1996;75:F209–F212.
Yong SC, Chen SJ, Boo NY. Incidence of nasal trauma associated with nasal prong versus nasal mask during continuous positive airway pressure treatment in very low birthweight infants: a randomised control study. Arch Dis Child Fetal Neonatal Ed. 2005;90:F480–F483.
Fischer C, Bertelle V, Hohlfield J, Guex MF, Diaw CS, Tolsa JF. Nasal trauma due to continuous positive airway pressure in neonates. Arch Dis Child Fetal Neonatal Ed. 2010;95:F447–F451.
Sinderby C, Beck J. Neurally adjusted ventilatory assist in non-invasive ventilation. Minerva Anestesiol. 2013;79:915–25.
Ulm LN, Hamvas A, Ferkol TW, Rodriguez OM, Cleveland CM, Linneman LA, et al. Sources of methodological variability in phase angles from respiratory inductance plethysmography in preterm infants. Ann Am Thorac Soc. 2014;11:753–60.
Gerdes JS, Sivieri EM, Abbasi S. Factors influencing delivered mean airway pressure during nasal CPAP with the RAM cannula. Pediatr Pulmonol. 2016;51:60–69.
Neotech Products. https://www.neotechproducts.com/product/neotech-ram-cannula/#. Accessed April 6, 2018.
Sivieri EM, Gerdes JS, Abbasi S. Effect of HFNC flow rate, cannula size, and nares diameter on generated airway pressures: an in vitro study. Pediatr Pulmonol. 2013;48:506–14.
Reddy SP, Fisher S, White DB, Stein H. Pilot study to compare two nasal airway interfaces in neonates on non-invasive neurally adjusted ventilatory assist. Neonatal Intensive Care. 2015;28:40–42.
Bailes SA, Firestone KS, Dunn DK, McNinch NL, Brown MF, Volsko TA. Evaluating the effect of flow and interface type on pressures delivered with bubble CPAP in a simulated model. Respir Care. 2016;61:333–9.
Hochwald O, Borenstein-Levin L, Dinur G, Jubran H, Littner Y, Breuer M, et al. The effect of changing respiratory rate settingds on CO2 levels during nasal intermittent positive pressure ventilation (NIPPV) in premature infants. J Perinatol. 2023;43:305–10.
Namdevb S, Tarafdar O, Fusch G, Beck J, Mukerji A. Pressure transmission and electrical diaphragm activity in preterm infants during nasal intermittent positive pressure ventilation – an exploratory prospective physiological study. J Perinatol. 2023;43:1004–6.
Acknowledgements
The authors would like to thank Arkansas Children’s Hospital and the University of Arkansas for Medical Sciences for the provision of a supportive environment in which to conduct research. We would also like to thank the nursing and respiratory therapy staff for their willingness to assist with instrumentation and other practical matters when needed. We thank Mr. Norm Comtois for his invaluable assistance with data analysis. Finally, we thank the families who allowed us the privilege of enlisting their babies in our pursual of improved practices in neonatal critical care.
Funding
This research study was funded by a Postdoctoral Fellowship Grant Award from Arkansas Children’s Research Institute.
Author information
Authors and Affiliations
Contributions
ALL, DNM, MDW, JB, CS, and SEC contributed to the conception and design of this study. ALL, DNM, and SEC consented and enrolled study patients and collected data from the trials. ALL, DNM, BS, MDW, JB, CS, and SEC contributed to data analysis and interpretation. The manuscript was composed by ALL, with critical revisions by DNM, BS, MDW, JB, CS, and SEC.
Corresponding author
Ethics declarations
Competing interests
JB, CS have secured patents for original inventions of technology for neural control of mechanical ventilation. These patents are designated to the academic institutions where the inventions were created, and the license for the patents is owned by Maquet Critical Care®. Commercial use of this technology may result in future financial gain to JB and CS via royalty payments. Neurovent Research Inc.® (NVR) is a research and development company owned by JB and CS which manufactures equipment and catheters for research studies, and there is a consultant contract between NVR and Maquet Critical Care®. ALL, DNM, BS, and SEC have no disclosures.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lynch, A.L., Matlock, D.N., Akmyradov, C. et al. Tidal volume delivery during nasal intermittent positive pressure ventilation: infant cannula vs. nasal continuous positive airway pressure prongs. J Perinatol 44, 244–249 (2024). https://doi.org/10.1038/s41372-023-01846-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-023-01846-7