Abstract
Study design
Pre–post intervention.
Objectives
-
1.
To test whether replacement of oral anticholinergic (AC) agents with mirabegron for neurogenic lower urinary tract dysfunction (NLUTD) yields improved cognitive function in older persons with spinal cord injury (SCI).
-
2.
To test whether mirabegron is safe and as efficacious as AC.
Setting
USA.
Methods
Pilot study: Twenty older (>60 y/o) persons with SCI taking chronic (>6 months) AC medication for NLUTD were enrolled. All participants were first studied on AC at baseline then switched to mirabegron for 6 months. Primary outcomes were cognitive tests of (1) executive function (TEXAS, SDMT); (2) attention (SCWT); and (3) memory (SLUMS and WMS-IV Story A/B). Secondary outcomes assessed efficacy and safety including Neurogenic Bladder Symptom Score (NBSS), bladder diary, neurogenic bowel dysfunction (NBD) survey, heart rate (HR), electrocardiogram (EKG), and mean arterial pressure (MAP).
Results
When switching from AC to mirabegron for NLUTD, older persons with SCI exhibited statistically significant improvements in immediate Story A recall (p = 0.01), delayed story A and B recall (p = 0.01, 0.004), and in TEXAS (p = 0.04). Three subscores within NBSS significantly improved (p = 0.001) and the frequency of incontinence decreased (p = 0.03) on mirabegron. NBD, HR, MAP, and EKGs were unchanged.
Conclusions
Older persons with SCI on AC for NLUTD demonstrated improved short-term and delayed memory (WMS-IV Story A/B) as well as executive function (TEXAS) when switched to mirabegron. Efficacy of mirabegron for NLUTD symptoms was superior to AC with no adverse effects on bowel or cardiovascular function.
Sponsorship
Claude D. Pepper Older Americans Independence Center.
Similar content being viewed by others
Introduction
Approximately 285,000 persons in the United States are living with a spinal cord injury (SCI) with an incidence of 17,500 persons annually [1]. Post-SCI loss of supraspinal bladder innervation impairs urine storage and evacuation resulting in ‘neurogenic lower urinary tract dysfunction‘ (NLUTD). As a result of NLUTD, up to 81% of persons with SCI experience urinary incontinence, urinary frequency, and frequent urinary tract infections (UTIs) [2, 3]. Treatment of NLUTD often requires lifelong treatment [2, 3]. Anticholinergic (AC) agents are currently the first-line pharmacological therapy for NLUTD with evidence of poor detrusor compliance and/or neurogenic detrusor overactivity. AC agents competitively but non-selectively, inhibit binding of the neurotransmitter, acetylcholine to all (M1–M5) muscarinic cholinergic receptors. The blocking of M3 receptors located at neuromuscular junctions in the human bladder detrusor muscle relaxes the detrusor smooth muscle and increases bladder capacity; however, due to the wide distribution of muscarinic cholinergic receptor subtypes (M1–M5) in the central nervous system and body, AC agents can have undesirable side effects including xerostomia, dry eye syndrome, blurred vision, constipation, as well as delirium or cognitive impairment/memory loss, especially in older persons.
Older SCI persons are particularly susceptible to the adverse cognitive effects of AC medications due to: (1) reduced cholinergic activity related to decreased acetylcholine synthesis and/or a decrease in acetylcholine receptors [4]; (2) increased blood–brain barrier permeability [5]; and (3) lower p-glycoprotein activity, which is an efflux central nervous system transporter [6]. Overall, these age-related changes increase the probability that AC medications will cause cognitive dysfunction. Furthermore, in 2003, Perry et al. [7] reported that AC use is associated with increased Alzheimer’s disease-related pathology. In the first prospective cohort study, Gray et al. demonstrated an association between higher cumulative AC medication use and increased risk for dementia [8]. In 2016, Risacher et al. [9] showed that AC medication use is associated with increased brain atrophy, reduced brain glucose metabolism, and decline in both general cognition and specific cognitive domains (memory and executive function) in older adults. According to the World Health Organization, AC agent use is one of the few modifiable risk factors that has been identified for dementia [10] and therefore recommend minimizing the use of AC agents. The American Geriatrics Society Beers Criteria advise providers against using AC agents in the older [11]. Due to the foregoing, AC agents appear to be best avoided in older persons if at all possible [12].
In 2012, a new Beta-3 adrenoreceptor agonist (Mirabegron) was FDA-approved for the treatment of overactive bladder which is functionally similar to neurogenic detrusor overactivity from NLUTD but is not neurogenic in etiology [13]. Mirabegron stimulates B3-adrenoreceptors and thus affects detrusor smooth muscle relaxation, decreases afferent signaling from the bladder, and increases bladder capacity. Therefore, mirabegron is not only expected to be effective in overactive bladder, consistent with FDA approval but also NLUT dysfunction associated with poor detrusor compliance and neurogenic detrusor overactivity [14]. Given this agent’s different mechanism of action, it is unsurprising that the adverse effect profile is different than AC-associated adverse effects, e.g., no cognitive impairment. Preliminary trials (N = 15) of off-label use of mirabegron in persons with SCI and NLUTD suggest that it is as effective and tolerable as AC agents for incontinence, increasing bladder capacity, decreasing voiding frequency, and detrusor compliance [15,16,17], thus, mirabegron appears to hold promise for NLUTD in patients with SCI.
Based on the foregoing, we hypothesized that cognition in older persons with SCI taking AC agents for NLUTD would improve when switched to mirabegron. Three main domains of cognitive function were assessed by neuropsychological measures: (1) memory, the processes of encoding information, information storage, and retrieval; 2) attention, the ability to maintain awareness of specific stimuli, sensations, or thoughts while disregarding others; and (3) executive function, the ability to critically assess situations, manipulate abstract information to solve complex problems, and direct behavior towards a goal. Secondarily, we hypothesized that mirabegron efficacy in treating NLUTD symptoms would be comparable or better than AC. Finally, to assess safety, cardiovascular parameters (i.e. heart rate (HR), mean arterial pressure, and electrical conduction) were monitored.
Materials and methods
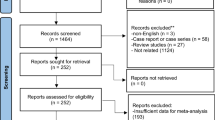
43 persons were screened using the inclusion and exclusion criteria in Table 1.
After screening, 20 were enrolled (19 male, 1 female). Demographics are reported in Table 2. The most frequent cause of exclusion was cardiac dysrhythmias (Fig. 1). After visit 1, the AC agent was discontinued and replaced with 25 mg mirabegron.
In a pre–post study design, all (primary and secondary) outcome measures were initially collected while on AC for baseline/control data (i.e.; 0 months). Participants were then switched to mirabegron. Outcome measures were repeated at various intervals while on 6 months of mirabegron treatment per the visit schedule outlined in Table 3.
Primary outcome measures
Cognitive tests
Measured a range of cognitive functions including memory, executive function, and attention. Tests included: (1) Weschler Memory Scale IV [18] (WMS-IV); (2) Saint Louis University Mental Status Exam (SLUMS) [19]; (3) The Telephone Executive Assessment Scale [20] (TEXAS); (4) Symbol Digit Modality Test [21] (SDMT); and (5) Stroop Color-Word Test [22] (SCWT) (Table 4 for descriptions). These tests were conducted at 0 months on AC and after 6 months of mirabegron therapy.
All cognitive assessments were administered by a clinical research nurse, who was trained and approved for testing by a geriatric neuropsychologist. Cognitive assessments were only conducted twice within a 6-month spanning interval to avoid confounding (i.e.; learning) effects of repeated testing. Some aspects of executive function tests that require a motor function that is compromised in SCI persons (i.e. hand function), therefore, the cognitive assessments chosen were those that did not require upper arm motor function, consistent with past SCI cognition studies [23].
Secondary outcome measures
Efficacy
To ensure comparable efficacy to AC, bladder function assessments (i.e. bladder diary (BD) and Neurogenic Bladder Symptom Score (NBSS)) were performed monthly.
BD was specifically developed for this study. Measured the frequency of bladder evacuation (BE)/24 h, frequency of incontinence (FI)/24 h, number of symptomatic urinary tract infections (SUTI)/month, urine output volume (UOP) (ml) per void, and average urine output (ml) per 24 h (UOP24).
Neurogenic Bladder Symptom Score
This validated quality of life (QOL) measure for urological problems in SCI specifically assesses symptoms and bladder-related consequences for NLUTD in SCI through 24 questions, divided into 4 subsections of Total incontinence (TI), total storage and voiding (TSV), total consequences (TC) and urinary specific QOL [24, 25]. These questions elucidate the frequency and quantity of urine leakage, the impact on health and everyday activities, the frequency and severity of UTIs, and the frequency of bladder and kidney stones. The total score can range from 0 (minimal symptoms) to 74 (maximum symptoms).
Safety
As mirabegron is a beta-3 agonist, to assess potential off-target beta-adrenergic effects, HR and mean arterial blood pressure (MAP) were recorded monthly. All MAP and HR measurements were conducted at the same time of day (within the hour) for each patient at Visit 1, 4, and 7. One BP and one HR recording were taken each visit. A standard 12 lead electrocardiogram with a rhythm strip was obtained every 3 months to assess changes in HR, rhythm, or conduction and was interpreted by a cardiologist. In addition, some studies in elderly persons on mirabegron have reported constipation could worsen due to beta-3 bowel receptors [26]. The validated neurogenic bowel dysfunction (NBD) survey [27] has two sections, “severity of dysfunction” (0–6 = Very Minor, 7–9 = Minor, 10–13 = Moderate, 14+ = Severe) and “general satisfaction” (0 = perfect and 10= total dissatisfaction). The NBD survey was collected monthly.
After baseline data were collected, participants were started on mirabegron 25 mg daily. After 3 weeks, participants were called and queried for any significant adverse changes in BD measures as listed above. If there were any adverse changes in BD measures (e.g. increased FI), the dose was increased to 50 mg daily. After this optimal dosing was determined, participants remained on that mirabegron dose for the remainder of the study (~5 months) (Table 1).
Statistical analysis
Primary outcomes
Raw mean scores of each cognitive test (WMS-IV story A and B, SLUMS, SDMT, SWCT, and TEXAS) were calculated then analyzed via a paired samples t-test to evaluate for significant differences in scores at V1 vs. V7.
Secondary outcomes
BD measures including frequency of BE/24 h, FI/24 h, number of SUTI/month, UOP (ml) per void, and average urine output (ml) per 24 h (UOP24) and NBSS data were analyzed via repeated-measures ANOVA. Each of the first three measures on sheet NBBS were analyzed separately with a mixed model ANOVA followed by Dunnett’s test to compare each visit with V1. Ordinal measures were analyzed with one-way repeated ordinal regression using the CLMM package in R. Before analysis, each ordinal measure was recoded with rank numbers ranging from low to high (the recoded measures are indicated by a lower case r). NBD interval measures were analyzed with a mixed model ANOVA. For all analyses, the statistical threshold for significance was set at p < 0.05.
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research. The VA-affiliate, University of Texas Health San Antonio Institutional Review Board (#20180376H) approved the study.
Results
Cognitive tests (primary outcome measures)
Memory
SLUMS scores showed no significant difference (p = 0.39) between V1 and V7 (Fig. 2). Similarly, WMS-IV Story A first immediate recall and Story B immediate recall did not show significant differences (p = 0.09 and p = 0.22, respectively) between V1 and V7. On the other hand, the second immediate Story A recall significantly differed between V1 and V7 (p = 0.01). Delayed recall of Story A and B scores also were significantly improved on AC vs mirabegron (p = 0.01 and 0.004, respectively) between V1 and V7, respectively.
Logical Memory subset (A–E) and Saint Louis University Mental Status (SLUMS) (F) at V1 vs. V7. A Story A first immediate; B Story A second immediate, C Story A delayed recall, D Story B immediate recall, E Story B delayed recall and F SLUMS. In each panel, the right-hand figure shows the mean difference between visits and its 95% confidence interval.
Executive function
SDMT “correct” scores showed no significant difference (p = 0.12) between V1 and V7. SDMT “incorrect” scores also showed no significant difference (p = 0.16) between V1 and V7 (data not shown). On the other hand, TEXAS scores significantly improved (p = 0.04) from V1 to V7 (Fig. 3).
Attention
SCWT scores were not significantly different between V1 and V7 on any page (pages 1–3) (p = 0.21, 0.18, 0.14, respectively).
Bladder and bowel function
Bladder diary
Within the BD data, the FI was the only measure that showed a significant effect of visit (p = 0.02) and a statistically significant decrease between V1 (mean rank number 1.50) and V7 (mean rank number 1.15) (p = 0.03). All other measures (BE, SUTI, UOP, UOP24) did not differ statistically over the 6 months.
NBSS scores
TI, TSV, and TC showed a statistically significant overall effect of visit (p = 0.01, 0.01, 0.01), with TSV and TC being significantly lower at V7 than V1 and at V4 and V3, respectively. NBSS QOL domain (0 = pleased, 4 = unhappy) showed a significant effect of visit (p = 0.03) and a significant difference between V1 and V7 (p = 0.04) (Fig. 4).
NBD
Neither “severity” (p = 0.13) or “general satisfaction” (p = 0.28) showed statistically significant effects of visit.
Cardiovascular parameters
HR and MAP did not significantly differ between visit (i.e. V1 (baseline/AC agent) vs. V4 (mirabegron) and V7 (mirabegron)) (HR: p = 0.31; MAP: p = 0.22) (Table 5). EKG rhythms were unchanged in all participants from V1 to V7. Post hoc analysis of HR and MAP data showed neither a statistically significant effect of dose (25 mg vs. 50 mg) (p = 0.59), visit (V1, V4, V7) (p = 0.42) nor a significant dose × visit interaction (p = 0.57).
Discussion
Given that persons with SCI have an increased risk of cognitive impairment, about 13 times higher than age-matched healthy controls [28, 29]; and the knowledge that AC agents carry a higher risk of dementia [9], identifying AC treatment alternatives in persons with SCI and NLUTD is desirable. This is the first study to examine the cognitive impact of removing AC agents and substituting mirabegron in persons with SCI and the comparative efficacy (NLUTD symptoms) and potential side effects on bowel and cardiovascular functions. It is noteworthy that documented detrimental effects of AC agents on cognition led the American Urological Association to update its guidelines in 2015 to include mirabegron as an alternative first-line agent for overactive bladder treatment. This study was conducted in hopes to demonstrate a lower side effect profile of mirabegron (compared to AC) with similar efficacy in persons with SCI with NLUTD. We found cognitive benefits in the realms of memory and executive function with mirabegron compared to AC treatment for NLUTD while improving efficacy and maintaining safety.
Memory
The Weschler Memory Scale-IV has been used as the main outcome measure in studies of cognitive decline in older persons without SCI [9]. We chose this measure to evaluate cognition in our older population with SCI. We used the Weschler Memory Scale-Revised Logical Memory Immediate Recall whose subsection of Story A and B did not require motor function. Interestingly, there was a significant improvement in the Wechsler logical memory stories on immediate and delayed recall. While the stories are strongly related to memory function, they also have an executive function component due to their structure with a beginning, middle, and end, which a respondent can utilize to organize their responses.
The second memory exam we used was SLUMS which just like the Mini-Mental Status Exam (MMSE) predicts mortality and institutionalization for male patients screened as positive for dementia [30]. SLUMS is suggested to be more sensitive than MMSE [30]. We found no significant change in the SLUMS scores which is not unexpected as the SLUMS is a screening assessment for cognitive impairment that has a limited number of memory items as well as other items related to general cognition. It is not conceptualized as a memory test, but more as a brief general cognitive screen, which even over a year re-test interval following medication or cognitive intervention, often do not show significant change [31].
Executive function
The TEXAS [20] tests executive function and is comprised of five items from the Exit Interview (EXIT25) [32] that can be administered by telephone. Four of those appear on the Quick EXIT and EXIT15. Switching from AC to mirabegron significantly improved the TEXAS score. This could be explained as these test items were extracted from the EXIT25 as the strongest items correlated with the total EXIT score representing an executive function [20, 32].
The SDMT assesses divided attention, visual scanning, tracking, and motor speed using a symbol/digit substitution task. It is sensitive to change in neurocognitive status, thus making it useful for evaluating interventions with good test–retest (r = 0.76) and alternate forms (r = 0.82, r = 0.84) reliability [33]. It also distinguishes between individuals with depression and those with organic dementia. There was no statistically significant change in SDMT scores after switching from AC to mirabegron. The lack of change in the SDMT could be explained by interpreting it as a complex multifactorial task that involves many components of executive function that include visual selective attention, visual scanning, processing speed, and even memory. Finally, A total raw score likely does not reflect independent contributions of these different components which could have improved.
Attention/psychomotor speed
SCWT assesses the ability to inhibit cognitive interference that occurs when the processing of a specific stimulus feature impedes the simultaneous processing of a second stimulus attribute. There was no significant change in SCWT after switching from AC to mirabegron. This finding could be related to the selective visual attention requirement and cognitive flexibility that both could result in poor performance due to cognitive slowing seen in normal aging. Furthermore, deficiencies in reading aloud may also be influencing decreased Stroop performance related to low education, language problems (articulation and verbal fluency), bilingualism, and even respiratory and mood issues. All participants were English speaking however education level was not assessed which is a limitation.
Efficacy and safety outcomes
NLUTD symptoms
NLUTD function measured by NBSS (median score is 19 out of a possible 74 with higher scores relating to worse symptoms) and BD both demonstrated no worsening of bladder function with improvement in many subscores (TC, TSV, TI, FI) over time. TC subscore improvement means there was overall less frequent and severe UTIs, less pain with voiding, fewer kidney/bladder stones, and bladder medication efficacy. TSV subscore improvement suggests mirabegron had an overall positive impact on the frequency of urgency, nocturia, the interval between emptying, stream, straining, and post-void fullness. TI and FI score improvements show mirabegron collectively improved frequency of daytime and nighttime incontinence, degree of saturation and number of pads used daily, oral fluid restriction, skin problems, and limitation of activities. Notably, urinary specific QOL on NBSS improved which is significant as the recovery of bladder function is one of the highest priorities of persons with SCI [34]. This subjective data suggests that mirabegron is either as effective or more effective than AC in the treatment of NLUTD. However, objective data from urodynamic studies (filling and voiding pressures) should be collected in future studies to most objectively compare the efficacy of AC vs. mirabegron.
Safety
The NBD assessment did not show any change in bowel function. The most concerning potential adverse effect was a change in HR or MAP; however, no significant changes were found. EKG monitoring also showed no change in rhythm. Thus, the safety (NBD, HR, and MAP) and efficacy (NBSS and BD) data gathered in this study revealed no major adverse effects caused by the switch from AC to mirabegron.
Limitations
Practice effects on all cognitive tests have been observed so a follow-up larger and longer study (1–2 years) may be needed to show significant findings consistent with these results that can then be interpreted as reliable cognitive indicators of significant change related to mirabegron. In addition, It should be noted that both the SDMT and SCWT tests have one common characteristic that involves a participant using controlled eye movements that are integrated with frontal eye fields and visual pathways that connect to the occipital lobe as part of the participant’s task in order to respond. Ocular-motor integration is an active neurocognitive area of study relating to visual perception and executive function but was not specifically examined in this study. Finally, the small sample size of this pilot study could also explain some non-statistically significant findings.
Conclusion
Substituting mirabegron for AC agents in older persons with SCI provides some cognitive benefit in the realms of executive function and memory as evidenced by significant improvement in scores of TEXAS and of the WMS-IV logical memory recall stories A and B, respectively. In addition, some therapeutic benefit in subjective NLUTD symptomatology (e.g. FI, TSV, TC) was also found. These benefits come without adverse impacts on bowel function or cardiovascular changes (HR, MAP, and EKG). Given the intrinsic impaired cognitive function of persons with SCI compared to age-matched controls, risk of dementia with chronic AC use, and the intrinsic risk of dementia in the older (>60 y/o) population, use of mirabegron should be considered prior to AC for NLUTD treatment in the older population with SCI to preserve cognition.
Data archiving
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
National Spinal Cord Injury Statistical Center. Facts and figures at a glance. Birmingham, AL: National Spinal Cord Injury Statistical Center; 2017. Accessed.
Ku JH. The management of neurogenic bladder and quality of life in spinal cord injury. BJU Int. 2006;98:739–45.
Stover SL. Review of forty years of rehabilitation issues in spinal cord injury. J Spinal Cord Med. 1995;18:175–82.
Bowie MW, Slattum PW. Pharmacodynamics in older adults: a review. Am J Geriatr Pharmacother. 2007;5:263–303.
Montagne A, Barnes SR, Sweeney MD, Halliday MR, Sagare AP, Zhao Z, et al. Blood–brain barrier breakdown in the aging human hippocampus. Neuron 2015;85:296–302.
van Assema DM, Lubberink M, Boellaard R, Schuit RC, Windhorst AD, Scheltens P, et al. P-glycoprotein function at the blood–brain barrier: effects of age and gender. Mol Imaging Biol. 2012;14:771–6.
Perry EK, Kilford L, Lees AJ, Burn DJ, Perry RH. Increased Alzheimer pathology in Parkinson’s disease related to antimuscarinic drugs. Ann Neurol. 2003;54:235–8.
Gray SL, Anderson ML, Hanlon JT, Dublin S, Walker RL, Hubbard RA, et al. Exposure to strong anticholinergic medications and dementia-related neuropathology in a community-based autopsy cohort. J Alzheimers Dis. 2018;65:607–16.
Risacher SL, McDonald BC, Tallman EF, West JD, Farlow MR, Unverzagt FW, et al. Association between anticholinergic medication use and cognition, brain metabolism, and brain atrophy in cognitively normal older adults. JAMA Neurol. 2016;73:721–32.
Gray SL, Hanlon JT. Anticholinergic medication use and dementia: latest evidence and clinical implications. Ther Adv Drug Saf. 2016;7:217–24.
By the American Geriatrics Society Beers Criteria Update Expert P. American Geriatrics Society. 2015 Updated beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227–46.
Gormley EA, Lightner DJ, Faraday M, Vasavada SP.American Urological A, Society of Urodynamics FPM Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline amendment. J Urol. 2015;193:1572–80.
Welk B. Mirabegron: a decade of study. Transl Androl Urol. 2020;9:2286–8.
Welk B, Hickling D, McKibbon M, Radomski S, Ethans K. A pilot randomized-controlled trial of the urodynamic efficacy of mirabegron for patients with neurogenic lower urinary tract dysfunction. Neurourol Urodyn. 2018;37:2810–7.
Wollner J, Pannek J. Initial experience with the treatment of neurogenic detrusor overactivity with a new beta-3 agonist (mirabegron) in patients with spinal cord injury. Spinal Cord. 2016;54:78–82.
Wada N, Okazaki S, Kobayashi S, Hashizume K, Kita M, Matsumoto S, et al. [Efficacy of combination therapy with mirabegron for anticholinergic-resistant neurogenic bladder: videourodynamic evaluation]. Hinyokika Kiyo. 2015;61:7–11.
Kamei J, Furuta A, Akiyama Y, Niimi A, Ichihara K, Fujimura T, et al. Video-urodynamic effects of mirabegron, a beta3-adrenoceptor agonist, in patients with low-compliance bladder. Int J Urol. 2015;22:956–61.
Drozdick LW, Holdnack JA, Salthouse T, Cullum CM. Assessing cognition in older adults with the WAIS-IV, WMS-IV, and ACS. In: WAIS-IV, WMS-IV, and ACS: Advanced clinical interpretation. San Diego, CA, USA: Elsevier Academic Press; 2013, p. 407–83. https://doi.org/10.1016/B978-0-12-386934-0.00009-2.
Szczesniak D, Rymaszewska J. The usfulness of the SLUMS test for diagnosis of mild cognitive impairment and dementia. Psychiatr Pol. 2016;50:457–72.
Salazar R, Velez CE, Royall DR. Telephone screening for mild cognitive impairment in hispanics using the Alzheimer’s questionnaire. Exp Aging Res. 2014;40:129–39.
Smith A. Symbol digit modalities test: manual. Los Angeles, CA, USA: Western Psychological Services; 1982.
Golden C, Stroop S. Color word test. Chicago: Stoelting Co.; 1978.
Lee J, Dudley-Javoroski S, Shields RK. Motor demands of cognitive testing may artificially reduce executive function scores in individuals with spinal cord injury. J Spinal Cord Med. 2021;44:253–61.https://doi.org/10.1080/10790268.2019.1597482.
Welk B, Morrow S, Madarasz W, Baverstock R, Macnab J, Sequeira K. The validity and reliability of the Neurogenic Bladder Symptom Score. J Urol. 2014;192:452–7.
Welk B, Lenherr S, Elliott S, Stoffel J, Presson A, Zhang C, et al. The Neurogenic Bladder Symptom Score (NBSS): a secondary assessment of its validity, reliability among people with a spinal cord injury. Spinal Cord. 2018;56:259–64.
Schemann M, Hafsi N, Michel K, Kober O, Wollmann J, Li Q, et al. The beta3-adrenoceptor agonist GW427353 (Solabegron) decreases excitability of human enteric neurons via release of somatostatin. Gastroenterology 2010;138:266–74.
Cameron AP, Rodriguez GM, Gursky A, He C, Clemens JQ, Stoffel JT. The severity of bowel dysfunction in patients with neurogenic bladder. J Urol. 2015;194:1336–41.
Craig A, Guest R, Tran Y, Middleton J. Cognitive impairment and mood states after spinal cord injury. J Neurotrauma. 2017;34:1156–63.
Chiaravalloti ND, Weber E, Wylie G, Dyson-Hudson T, Wecht JM. Patterns of cognitive deficits in persons with spinal cord injury as compared with both age-matched and older individuals without spinal cord injury. J Spinal Cord Med. 2020;43:88–97.
Cruz-Oliver DM, Malmstrom TK, Roegner M, Tumosa N, Grossberg GT. Cognitive deficit reversal as shown by changes in the Veterans Affairs Saint Louis University Mental Status (SLUMS) examination scores 7.5 years later. J Am Med Dir Assoc. 2014;15:687e685–610.
Tombaugh TN. Test–retest reliable coefficients and 5-year change scores for the MMSE and 3MS. Arch Clin Neuropsychol. 2005;20:485–503.
Royall DR, Mahurin RK, Gray KF. Bedside assessment of executive cognitive impairment: the executive interview. J Am Geriatr Soc. 1992;40:1221–6.
Kiely KM, Butterworth P, Watson N, Wooden M. The Symbol Digit Modalities Test: normative data from a large nationally representative sample of Australians. Arch Clin Neuropsychol. 2014;29:767–75.
Anderson KD. Targeting recovery: priorities of the spinal cord-injured population. J Neurotrauma. 2004;21:1371–83.
Acknowledgements
This material is the result of work supported with resources and the use of facilities at the Audie L. Murphy Veteran’s Affairs Hospital in San Antonio, TX.
Funding
Funding is provided by Claude D. Pepper Older Americans Idependence Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
While SK (author) does acknowledge conflict of interest with the manufacturer of Mirabegron (Astellas), he did not have access to data from the study that could have potentially created bias in results.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Trbovich, M., Romo, T., Polk, M. et al. The treatment of neurogenic lower urinary tract dysfunction in persons with spinal cord injury: An open label, pilot study of anticholinergic agent vs. mirabegron to evaluate cognitive impact and efficacy. Spinal Cord Ser Cases 7, 50 (2021). https://doi.org/10.1038/s41394-021-00413-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41394-021-00413-6
This article is cited by
-
Impact of Neurogenic Lower Urinary Tract Dysfunction on Bowel Symptoms
Current Bladder Dysfunction Reports (2023)
-
Efficacy and safety of mirabegron for treatment of neurogenic detrusor overactivity in adults with spinal cord injury or multiple sclerosis: a systematic review
Spinal Cord (2022)
-
Urological Care After Spinal Cord Injury
Current Physical Medicine and Rehabilitation Reports (2022)