Abstract
Background
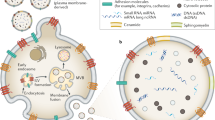
Urinary extracellular vesicles (uEVs) secreted from bladder cancer contain cancer-specific proteins that are potential diagnostic biomarkers. We identified and evaluated a uEV-based protein biomarker for bladder cancer diagnosis and analysed its functions.
Methods
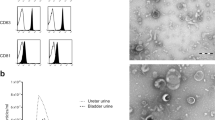
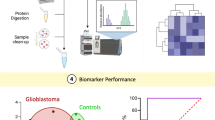
Biomarker candidates, selected by shotgun proteomics, were validated using targeted proteomics of uEVs obtained from 49 patients with and 48 individuals without bladder cancer, including patients with non-malignant haematuria. We developed an enzyme-linked immunosorbent assay (ELISA) for quantifying the uEV protein biomarker without ultracentrifugation and evaluated urine samples from 36 patients with and 36 patients without bladder cancer.
Results
Thirteen membrane proteins were significantly upregulated in the uEVs from patients with bladder cancer in shotgun proteomics. Among them, eight proteins were validated by target proteomics, and Ephrin type-A receptor 2 (EphA2) was the only protein significantly upregulated in the uEVs of patients with bladder cancer, compared with that of patients with non-malignant haematuria. The EV-EphA2-CD9 ELISA demonstrated good diagnostic performance (sensitivity: 61.1%, specificity: 97.2%). We showed that EphA2 promotes proliferation, invasion and migration and EV-EphA2 promotes the invasion and migration of bladder cancer cells.
Conclusions
We established EV-EphA2-CD9 ELISA for uEV-EphA2 detection for the non-invasive early clinical diagnosis of bladder cancer.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Babjuk M. Trends in bladder cancer incidence and mortality: success or disappointment? Eur Urol. 2017;71:109–10. https://doi.org/10.1016/j.eururo.2016.06.040.
Yoshida T, Kates M, Fujita K, Bivalacqua TJ, McConkey DJ. Predictive biomarkers for drug response in bladder cancer. Int J Urol. 2019;26:1044–53.
Lotan Y, Roehrborn CG. Sensitivity and specificity of commonly available bladder tumor markers versus cytology: results of a comprehensive literature review and meta-analyses. Urology. 2003;61:109–18.
Schmitz-Dräger BJ, Droller M, Lokeshwar VB, Lotan Y, Hudson MA, Van Rhijn BW, et al. Molecular markers for bladder cancer screening, early diagnosis, and surveillance: The WHO/ICUD consensus. Urol Int. 2015;94:1–24.
Oeyen E, Hoekx L, De Wachter S, Baldewijns M, Ameye F, Mertens I. Bladder cancer diagnosis and follow-up: the current status and possible role of extracellular vesicles. Int J Mol Sci. 2019;20:821.
Yáñez-Mó M, Siljander PRM, Andreu Z, Zavec AB, Borràs FE, Buzas EI, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015;4:1–60.
Liu YR, Ortiz-Bonilla CJ, Lee YF. Extracellular vesicles in bladder cancer: biomarkers and beyond. Int J Mol Sci. 2018;19:2822.
Fujita K, Nonomura N. Urinary biomarkers of prostate cancer. Int J Urol. 2018;25:770–9.
Fujita K, Kume H, Matsuzaki K, Kawashima A, Ujike T, Nagahara A, et al. Proteomic analysis of urinary extracellular vesicles from high Gleason score prostate cancer. Sci Rep. 2017;7:1–9.
Linxweiler J, Junker K. Extracellular vesicles in urological malignancies: an update. Nat Rev Urol. 2020;17:11–27. https://doi.org/10.1038/s41585-019-0261-8.
Matsuzaki K, Fujita K, Jingushi K, Kawashima A, Ujike T, Nagahara A, et al. MiR-21-5p in urinary extracellular vesicles is a novel biomarker of urothelial carcinoma. Oncotarget. 2017;8:24668–78.
Tomiyama E, Matsuzaki K, Fujita K, Shiromizu T, Narumi R, Jingushi K, et al. Proteomic analysis of urinary and tissue-exudative extracellular vesicles to discover novel bladder cancer biomarkers. Cancer Sci. 2021;112:2033–45.
Lee J, McKinney KQ, Pavlopoulos AJ, Niu M, Kang JW, Oh JW, et al. Altered proteome of extracellular vesicles derived from bladder cancer patients urine. Mol Cells. 2018;41:179–87.
Smalley DM, Sheman NE, Nelson K, Theodorescu D. Isolation and identification of potential urinary microparticle biomarkers of bladder cancer. J Proteome Res. 2008;7:2088–96.
Chen CL, Lai YF, Tang P, Chien KY, Yu JS, Tsai CH, et al. Comparative and targeted proteomic analyses of urinary microparticles from bladder cancer and hernia patients. J Proteome Res. 2012;11:5611–29.
Lin SY, Chang CH, Wu HC, Lin CC, Chang KP, Yang CR, et al. Proteome profiling of urinary exosomes identifies alpha 1-antitrypsin and H2B1K as diagnostic and prognostic biomarkers for urothelial carcinoma. Sci Rep [Internet]. 2016;6:1–12. https://doi.org/10.1038/srep34446.
Tomiyama E, Fujita K, Nonomura N. Urinary extracellular vesicles. In: Salvi S, Casadio V (eds). Urinary Biomarkers: Methods and Protocols, Methods in Molecular Biology, vol. 2292. (New York: Humana Press; 2021) pp 173–81.
Nakai W, Yoshida T, Diez D, Miyatake Y, Nishibu T, Imawaka N, et al. A novel affinity-based method for the isolation of highly purified extracellular vesicles. Sci Rep. 2016;6:1–11.
Tomiyama E, Fujita K, Rodriguez Pena MDC, Taheri D, Banno E, Kato T, et al. Expression of nectin-4 and pd-l1 in upper tract urothelial carcinoma. Int J Mol Sci. 2020;21:1–13.
Dadhania V, Zhang M, Zhang L, Bondaruk J, Majewski T, Siefker-Radtke A, et al. Meta-analysis of the luminal and basal subtypes of bladder cancer and the identification of signature immunohistochemical markers for clinical use. EBioMedicine. 2016;12:105–17.
Tomiyama E, Fujita K, Hashimoto M, Miyamoto H, Netto GJ, Nonomura N. Programmed cell death-ligand 1 expression in different molecular subtypes of upper tract urothelial carcinoma. Int J Urol. 2022;29:89–90.
Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, Zhang F. Genome engineering using the CRISPR-Cas9 system. Nat Protoc. 2013;8:2281–308.
Gonzales P, Pisitkun T, Knepper MA. Urinary exosomes: is there a future? Nephrol Dial Transpl. 2008;23:1799–801.
Rood IM, Deegens JKJ, Merchant ML, Tamboer WPM, Wilkey DW, Wetzels JFM, et al. Comparison of three methods for isolation of urinary microvesicles to identify biomarkers of nephrotic syndrome. Kidney Int [Internet]. 2010;78:810–6. https://doi.org/10.1038/ki.2010.262
Dhondt B, Geeurickx E, Tulkens J, Van Deun J, Vergauwen G, Lippens L, et al. Unravelling the proteomic landscape of extracellular vesicles in prostate cancer by density-based fractionation of urine. J Extracell Vesicles. 2020. https://doi.org/10.1080/20013078.2020.1736935
Wei Q, Wei L, Zhang J, Li Z, Feng H, Ren L. EphA2-enriched exosomes promote cell migration and are a potential diagnostic serum marker in pancreatic cancer. Mol Med Rep. 2020;22:2941–7.
Gao Z, Han X, Zhu Y, Zhang H, Tian R, Wang Z. Drug-resistant cancer cell-derived exosomal EphA2 promotes breast cancer metastasis via the EphA2-Ephrin A1 reverse signaling. Cell Death Dis. 2021. https://doi.org/10.1038/s41419-021-03692-x
BioLegend, Inc. Purified anti-human EphA2 Antibody. 2013. https://www.biolegend.com/ja-jp/products/purified-anti-human-epha2-antibody-8365?GroupID=BLG15664. Accessed 25 May 2022.
Pasquale EB. Eph-ephrin bidirectional signaling in physiology and disease. Cell. 2008;133:38–52.
Wilson K, Shiuan E, Brantley-Sieders DM. Oncogenic functions and therapeutic targeting of EphA2 in cancer. Oncogene. 2021;40:2483–95. https://doi.org/10.1038/s41388-021-01714-8
Dunne PD, Dasgupta S, Blayney JK, McArt DG, Redmond KL, Weir JA, et al. EphA2 expression is a key driver of migration and invasion and a poor prognostic marker in colorectal cancer. Clin Cancer Res. 2016;22:230–42.
Brannan JM, Dong W, Prudkin L, Behrens C, Lotan R, Bekele BN, et al. Expression of the receptor tyrosine kinase EphA2 is increased in smokers and predicts poor survival in non -small cell lung cancer. Clin Cancer Res. 2009;15:4423–30.
Zelinski DP, Zantek ND, Stewart JC, Irizarry AR, Kinch MS. EphA2 overexpression causes tumorigenesis of mammary epithelial cells. Cancer Res. 2001;61:2301–6.
Brantley-Sieders DM, Zhuang G, Hicks D, Wei BF, Hwang Y, Cates JMM, et al. The receptor tyrosine kinase EphA2 promotes mammary adenocarcinoma tumorigenesis and metastatic progression in mice by amplifying ErbB2 signaling. J Clin Invest. 2008;118:64–78.
Yuan WJ, Ge J, Chen ZK, Wu SB, Shen H, Yang P, et al. Over-expression of EphA2 and ephrina-1 in human gastric adenocarcinoma and its prognostic value for postoperative patients. Dig Dis Sci. 2009;54:2410–7.
Miyazaki T, Kato H, Fukuchi M, Nakajima M, Kuwano H. EphA2 overexpression correlates with poor prognosis in esophageal squamous cell carcinoma. Int J Cancer. 2003;103:657–63.
Mudali SV, Fu B, Lakkur SS, Luo M, Embuscado EE, Iacobuzio-Donahue CA. Patterns of EphA2 protein expression in primary and metastatic pancreatic carcinoma and correlation with genetic status. Clin Exp Metastasis. 2006;23:357–65.
Zeng G, Hu Z, Kinch MS, Pan CX, Flockhart DA, Kao C, et al. High-level expression of EphA2 receptor tyrosine kinase in prostatic intraepithelial neoplasia. Am J Pathol. 2003;163:2271–6. https://doi.org/10.1016/S0002-9440(10)63584-5
Liu Y, Zhang X, Qiu Y, Huang D, Zhang S, Xie L, et al. Clinical significance of EphA2 expression in squamous-cell carcinoma of the head and neck. J Cancer Res Clin Oncol. 2011;137:761–9.
Wang LF, Fokas E, Juricko J, You A, Rose F, Pagenstecher A, et al. Increased expression of EphA7 correlates with adverse outcome in primary and recurrent glioblastoma multiforme patients. BMC Cancer. 2008;8:151–6.
Thaker PH, Deavers M, Celestino J, Thornton A, Fletcher MS, Landen CN, et al. EphA2 expression is associated with aggressive features in ovarian carcinoma. Clin Cancer Res. 2004;10:5145–50.
Abraham S, Knapp DW, Cheng L, Snyder PW, Mittal SK, Bangari DS, et al. Expression of EphA2 and Ephrin A-1 in carcinoma of the urinary bladder. Clin Cancer Res. 2006;12:353–60.
Kamoun W, Swindell E, Pien C, Luus L, Cain J, Pham M, et al. Targeting epha2 in bladder cancer using a novel antibody-directed nanotherapeutic. Pharmaceutics. 2020;12:1–18.
Mo J, Zhao X, Dong X, Liu T, Zhao N, Zhang D, et al. Effect of EphA2 knockdown on melanoma metastasis depends on intrinsic ephrinA1 level. Cell Oncol. 2020;43:655–67.
Hess AR, Seftor EA, Gardner LMG, Carles-Kinch K, Schneider GB, Seftor REB, et al. Molecular regulation of tumor cell vasculogenic mimicry by tyrosine phosphorylation: role of epithelial cell kinase (Eck/Epha2). Cancer Res. 2001;61:3250–5.
Takasugi M, Okada R, Takahashi A, Virya Chen D, Watanabe S, Hara E. Small extracellular vesicles secreted from senescent cells promote cancer cell proliferation through EphA2. Nat Commun. 2017;8:1–11. https://doi.org/10.1038/ncomms15728
Acknowledgements
We thank Sadamu Ozaki, Takahiro Nishibu and Shigeaki Nakazawa for their help in constructing the ELISA and Mutsumi Tsuchiya and Atsuko Yasumoto for their technical support.
Funding
This study was supported by the Japan Society for the Promotion of Science under KAKENHI (grant numbers: 17K16788 and 20K18140) and the Japan Agency for Medical Research and Development under Translational Research (grant number: A102).
Author information
Authors and Affiliations
Contributions
Study conception and design: ET and KF; data acquisition: ET, KM and RN; data analysis: ET, KM, RN, AY, TU, GY, YK, MM and YH; provision of resources: ET, KM, YH, MH, EB, RU, TT and ST; drafting the manuscript and figures: ET and KF; reviewing the manuscript: TK, KH, AK, MU, HU, JA, TT and NN. All authors have read and approved the final draft for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Approval was obtained from the Institutional Review Board (Osaka University Hospital Institutional Review Board, Protocol Number: 13397-11) before initiating the study, and all patients provided written informed consent. The study was performed in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Tomiyama, E., Fujita, K., Matsuzaki, K. et al. EphA2 on urinary extracellular vesicles as a novel biomarker for bladder cancer diagnosis and its effect on the invasiveness of bladder cancer. Br J Cancer 127, 1312–1323 (2022). https://doi.org/10.1038/s41416-022-01860-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-022-01860-0
This article is cited by
-
Targeting cancer-derived extracellular vesicles by combining CD147 inhibition with tissue factor pathway inhibitor for the management of urothelial cancer cells
Cell Communication and Signaling (2024)
-
Recent progress in mass spectrometry-based urinary proteomics
Clinical Proteomics (2024)
-
Exosome, the glass slipper for Cinderella of cancer—bladder cancer?
Journal of Nanobiotechnology (2023)
-
Insight into extracellular vesicles in vascular diseases: intercellular communication role and clinical application potential
Cell Communication and Signaling (2023)