Abstract
Background
Adverse event (AE) reporting in early-phase clinical trials is essential in determining the tolerability of experimental anticancer therapies. The patient-reported outcome version of the CTCAE (PRO-CTCAE) evaluates AE components such as severity and interference in daily life. The aim of this study was to correlate the grade of clinician-reported AEs with patients’ reported experience of these toxicities using PRO-CTCAE.
Methods
Patients with advanced solid tumours enrolled on Phase I clinical trials were surveyed using the PRO-CTCAE. Symptomatic AEs were recorded by physicians using the CTCAE. A logistic regression model was used to assess associations between CTCAE grade and PRO responses.
Results
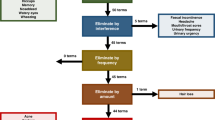
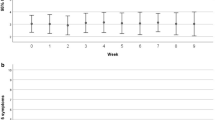
Of 219 evaluable patients, 81 experienced a high-grade (3/4) clinician-reported symptom, and of these, only 32 (40%) and 26 (32%) patients concordantly reported these as either severe or very severe, and interfering with daily life either ‘quite a bit’ or ‘very much’, respectively. Of the 137 patients who experienced a low-grade (1/2) clinician-reported AE as their worst symptom, 98 (72%) and 118 (86%) patients concordantly reported these as either mild–moderate severity and minimally interfering with daily life, respectively. There was a statistically significant association between clinician-reported AE grade and interference. Interference scores were also associated with dose reductions.
Conclusion
This is the first study to explore patient-reported severity and interference from symptomatic toxicities and compare clinician grading of the same toxicities. The study provided further evidence to support the added value of the PRO-CTCAE in Phase I oncology trials, which would make AE reporting patient-centred. Further work is needed to determine how this would affect the assessment of tolerability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Basch E, Dueck AC, Rogak LJ, Minasian LM, Kelly WK, O’Mara AM, et al. Feasibility assessment of patient reporting of symptomatic adverse events in multicenter cancer clinical trials. JAMA Oncol. 2017;3:1043–50.
Basch E, Reeve BB, Mitchell SA, Clauser SB, Minasian LM, Dueck AC, et al. Development of the National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). J Natl Cancer Inst. 2014;106:dju244.
Dueck AC, Mendoza TR, Mitchell SA, Reeve BB, Castro KM, Rogak LJ, et al. Validity and reliability of the US National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). JAMA Oncol. 2015;1:1051–9.
Cook N, Hansen AR, Siu LL, Abdul, Razak AR. Early phase clinical trials to identify optimal dosing and safety. Mol Oncol. 2015;9:997–1007.
Lai-Kwon JE, Yin Z, Minchom AR, Yap C. 585P trends in patient-reported outcome (PRO) use in early phase oncology trials. Ann Oncol. 2020;31:S495.
Fiteni F, Ray IL, Ousmen A, Isambert N, Anota A, Bonnetain F. Health-related quality of life as an endpoint in oncology phase I trials: a systematic review. BMC Cancer. 2019;19:361.
Sedhom R, Ferrell B, Ruel N, Koczywas M, Chung V, Smith TJ. Using patient-reported outcomes to describe the patient experience on phase I clinical trials. JNCI cancer Spectr. 2020;4:pkaa067–pkaa.
Veitch ZW, Shepshelovich D, Gallagher C, Wang L, Abdul Razak AR, Spreafico A, et al. Underreporting of symptomatic adverse events in phase I clinical trials. J Natl Cancer Institute. 2021;113:980–8.
Tang E, Ravaud P, Riveros C, Perrodeau E, Dechartres A. Comparison of serious adverse events posted at ClinicalTrials.gov and published in corresponding journal articles. BMC Med. 2015;13:189.
Ioannidis JP, Lau J. Completeness of safety reporting in randomized trials: an evaluation of 7 medical areas. J Am Med Assoc.2001;285:437–43.
Scharf O, Colevas AD. Adverse event reporting in publications compared with sponsor database for cancer clinical trials. J Clin Oncol. 2006;24:3933–8.
Atkinson TM, Ryan SJ, Bennett AV, Stover AM, Saracino RM, Rogak LJ, et al. The association between clinician-based common terminology criteria for adverse events (CTCAE) and patient-reported outcomes (PRO): a systematic review. Support Care Cancer. 2016;24:3669–76.
Cirillo M, Venturini M, Ciccarelli L, Coati F, Bortolami O, Verlato G. Clinician versus nurse symptom reporting using the National Cancer Institute—common terminology criteria for adverse events during chemotherapy: results of a comparison based on patient’s self-reported questionnaire. Ann Oncol. 2009;20:1929–35.
Atkinson TM, Dueck AC, Satele DV, Thanarajasingam G, Lafky JM, Sloan JA, et al. Clinician vs patient reporting of baseline and postbaseline symptoms for adverse event assessment in cancer clinical trials. JAMA Oncol. 2020;6:437–9.
Bennett AV, Dueck AC, Mitchell SA, Mendoza TR, Reeve BB, Atkinson TM, et al. Mode equivalence and acceptability of tablet computer-, interactive voice response system-, and paper-based administration of the U.S. National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Health Qual Life Outcomes. 2016;14:24.
Jacobsen P. National Cancer Institute, Division of Cancer control andPopulation Sciences, Healthcare Delivery and Research Program. Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAETM). 2019. http://healthcaredelivery.cancer.gov/pro-ctcae/.
Atkinson TM, Hay JL, Dueck AC, Mitchell SA, Mendoza TR, Rogak LJ, et al. What do “none”, “mild”, “moderate”, “severe”, and “very severe” mean to patients with cancer? Content validity of PRO-CTCAE™ response scales. J Pain Symptom Manag. 2018;55:e3–e6.
Health UDo, Services H. Common terminology criteria for adverse events. Version 5.0. Published November 27, 2017. 2020.
Chung AE, Shoenbill K, Mitchell SA, Dueck AC, Schrag D, Bruner DW, et al. Patient free text reporting of symptomatic adverse events in cancer clinical research using the National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). J Am Med Inf Assoc. 2019;26:276–85.
Basch E, Rogak LJ, Dueck AC. Methods for Implementing and reporting patient-reported outcome (PRO) measures of symptomatic adverse events in cancer clinical trials. Clin Ther. 2016;38:821–30.
Basch E, Becker C, Rogak LJ, Schrag D, Reeve BB, Spears P, et al. Composite grading algorithm for the National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Clin Trials. 2021;18:104–14.
Gresham G, Diniz MA, Razaee ZS, Luu M, Kim S, Hays RD, et al. Evaluating treatment tolerability in cancer clinical trials using the toxicity index. J Natl Cancer Institute. 2020;112:1266–74.
Goldberg RM, Sargent DJ, Morton RF, Mahoney MR, Krook JE, O’Connell MJ. Early detection of toxicity and adjustment of ongoing clinical trials: the history and performance of the North Central Cancer Treatment Group’s real-time toxicity monitoring program. J Clin Oncol. 2002;20:4591–6.
Acknowledgements
The study group would like to acknowledge the patients and their families who contributed to and made this research possible.
Funding
No funding was procured for this study.
Author information
Authors and Affiliations
Contributions
Study concepts and design: DS and ARH. Data acquisition—GW, ZWV and DS. Quality control of data and algorithms: GW, DS, ZWV and ZAL. Data analysis and interpretation—GW, ZWV, ZAL and ARH. Statistical analysis: ZAL, GW and ZWV. Manuscript preparation: GW and ARH. Manuscript editing: GW, ZWV, ARH, DS, ZAL, AAR, AS, PLB, LLS and LM.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This protocol was approved by the Princess Margaret Cancer Centre Research Ethics Board (REB) and conformed to the Helsinki Declaration. All participants provided written informed consent prior to study enrolment.
Competing interests
GW has received travels grants from BMS and Abbvie, and received honoraria from Pfizer. ZWV has received an honorarium from Pfizer and Genomic Health. He has also served on an advisory board for Genomic Health. PLB reports grants from Bristol-Myers-Squibb, Sanofi, Genentech/Roche Novartis, GlaxoSmithKline, Nektar Therapeutics, Merck, Lilly, Servier, PTC Therapeutics, Seattle Genetics, Mersana outside the submitted work; and Uncompensated advisory boards for Bristol-Myers-Squibb, Sanofi, Pfizer, Genentech/Roche. LLS reports compensated consultancy/advisory board participation for Merck, Pfizer, Celgene, AstraZeneca/Medimmune, Morphosys, Roche, GeneSeeq, Loxo, Oncorus, Symphogen, Seattle Genetics, Glaxo-Smith-Kline, Voronoi, Treadwell Therapeutics, Arvinas, Tessa and Navire; and reports institutional support for clinical trials conduct from Novartis, Bristol-Myers Squibb, Pfizer, Boehringer-Ingelheim, GlaxoSmithKline, Roche/Genentech, Karyopharm, AstraZeneca/Medimmune, Merck, Celgene, Astellas, Bayer, AbbVie, Amgen, Symphogen, Intensity Therapeutics, Mirati, Shattucks and Avid. Spouse hold stock in Agios and Treadwell Therapeutics. ARH reports institutional support for clinical trials to conduct by Novartis, Bristol-Myers Squibb, Pfizer, Boehringer-Ingelheim, GlaxoSmithKline, Roche/Genentech, Karyopharm, AstraZeneca/Medimmune, Merck, Astellas and Bayer; and compensated consulting/advisory boards for AstraZeneca, Merck and GlaxoSmithKline. ARAR has received research funding from Roche, Genentech, Eli Lilly, Merck, Boehringer-Ingelheim, Novartis, AbbVie, Deciphera, Karyopharm, AstraZeneca, Medimmune, Blueprint, Bristol-Myers Squibb, GSK, Entremed/Casi Pharmaceuticals, Adaptimmune and BetaCat. He also has also served the advisory board for Eli Lilly, Merck, Adaptimmune, Boehringer-Ingelheim. AS has served as an advisory board consultant for Merck (compensated), Bristol-Myers Squibb (compensated), Novartis (compensated), Oncorus (compensated), Janssen (compensated). She has also received research funding from Novartis, Bristol-Myers Squibb, Symphogen AstraZeneca/Medimmune, Merck, Bayer, Surface Oncology, Northern Biologics, Janssen Oncology/Johnson & Johnson, Roche, Regeneron, Alkermes, Array Biopharma, GSK. DS, ZAL, LM report no competing interests.
Consent to publish
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Watson, G.A., Veitch, Z.W., Shepshelovich, D. et al. Evaluation of the patient experience of symptomatic adverse events on Phase I clinical trials using PRO-CTCAE. Br J Cancer 127, 1629–1635 (2022). https://doi.org/10.1038/s41416-022-01926-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-022-01926-z
This article is cited by
-
Adverse event signal extraction from cancer patients’ narratives focusing on impact on their daily-life activities
Scientific Reports (2023)
-
A tailored phase I-specific patient-reported outcome (PRO) survey to capture the patient experience of symptomatic adverse events
British Journal of Cancer (2023)