Abstract
Background
Vitamin D is a modulator of immune functions. Investigations on the mechanisms of vitamin D action and pathogenesis of Hashimoto’s thyroiditis (HT) have revealed that vitamin D can reduce damages to thyroid cells caused by autoreactive immune cells.
Methods
Totally, 48 female patients with HT disease were introduced to the study by endocrinologists. Patients were divided into two major groups of 24 individuals and treated weekly with 50,000 IU of cholecalciferol (vitamin D group) or placebo (placebo group) using oral administration for 3 months. Eventually, 17 of the 24 patients in each group finished the study. Before and after supplementation, frequencies of Th1, Th17, Th2 and Tr1 cells and mean fluorescent intensity (MFI) of the associated cytokines, including IFN-γ, IL-17, IL-4 and IL-10, were assessed using flow cytometry. Furthermore, gene expression of IL-10 was assessed using real-time PCR.
Results
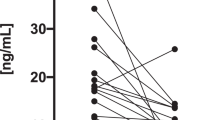
Results of this study showed that cholecalciferol supplementation caused a significant decrease in Th17/Tr1 ratio. The proportion and MFI of Th1, Th2, Tr1 and Th17 cells included no significant changes in vitamin D group, compared to those in placebo group. Expression rate and MFI of IL-10 increased in both groups. This increase was higher in vitamin D group than placebo group with no significance.
Conclusions
In this novel preliminary clinical trial study, supplementation with cholecalciferol in HT patients for 3 months changed the balance of CD4+ T-cell subsets to improve the disease control. However, further studies are necessary to investigate effects of vitamin D on immune functions in HT patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Caturegli P, De Remigis A, Rose NR. Hashimoto thyroiditis: clinical and diagnostic criteria. Autoimmun Rev. 2014;13:391–7.
Ben-Skowronek I, Szewczyk L, Ciechanek R, Korobowicz E. Interactions of lymphocytes, thyrocytes and fibroblasts in Hashimoto’s thyroiditis: an immunohistochemical and ultrastructural study. Horm Res Paediatr. 2011;76:335–42.
Zhang L, Li H, Ji QH, Zhu YX, Wang ZY, Wang Y, et al. The clinical features of papillary thyroid cancer in Hashimoto’s thyroiditis patients from an area with a high prevalence of Hashimoto’s disease. BMC Cancer. 2012;12:610.
Azizi G, Keller JM, Lewis M, Piper K, Puett D, Rivenbark KM, et al. Association of Hashimoto’s thyroiditis with thyroid cancer. Endocr Relat Cancer. 2014;21:845–52.
Jiskra J. [Management of hypothyroidism and hyperthyroidism]. Vnitr Lek. 2015;61:868–72.
Bogner U, Wall SH Jr. Antibody-dependent cell mediated cytotoxicity against human thyroid cells in Hashimoto’s thyroiditis but not Graves’ disease. J Clin Endocrinol Metab. 1984;59:734–8.
Cogni G, Chiovato L. An overview of the pathogenesis of thyroid autoimmunity. Hormones. 2013;12:19–29.
Bock G, Prietl B, Mader JK, Höller E, Wolf M, Pilz S, et al. The effect of vitamin D supplementation on peripheral regulatory T cells and β cell function in healthy humans: a randomized controlled trial. Diabetes Metab Res Rev. 2011;27:942–5.
Palmer MT, Lee YK, Maynard CL, Oliver JR, Bikle DD, Jetten AM, et al. Lineage-specific effects of 1,25-dihydroxyvitamin D(3) on the development of effector CD4 T cells. J Biol Chem. 2011;286:997–1004.
Mishima Y, Liu B, Hansen JJ, Sartor RB. Resident bacteria-stimulated interleukin-10-sSecreting B cells ameliorate T-cell-mediated colitis by inducing T-rRegulatory-1 cells that require interleukin-27 sSignaling. Cell Mol Gastroenterol Hepatol. 2015;1:295–310.
Gonzalez-Amaro R, Marazuela M. T regulatory (Treg) and T helper 17 (Th17) lymphocytes in thyroid autoimmunity. Endocrine. 2016;52:30–8.
Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5:2502–21.
Soleimani M, Jameie SB, Mehdizadeh M, Keradi M, Masoumipoor M, Mehrabi S. Vitamin D3 influence the Th1/Th2 ratio in C57BL/6 induced model of experimental autoimmune encephalomyelitis. Iran J Basic Med Sci. 2014;17:785–92.
Hayes CE, Hubler SL, Moore JR, Barta LE, Praska CE, Nashold FE. Vitamin D actions on CD4(+) T cells in autoimmune disease. Front Immunol. 2015;6:1664–3224.
Cantorna MT. Mechanisms underlying the effect of vitamin D on the immune system. Proc Nutr Soc. 2010;69:286–9.
Giovinazzo S, Vicchio TM, Certo R, Alibrandi A, Palmieri O, Campenni A, et al. Vitamin D receptor gene polymorphisms/haplotypes and serum 25(OH)D3 levels in Hashimoto’s thyroiditis. Endocrine. 2017;55:599–606.
Ke W, Sun T, Zhang Y, He L, Wu Q, Liu J, et al. 25-Hydroxyvitamin D serum level in Hashimoto’s thyroiditis, but not Graves’ disease is relatively deficient. Endocr J. 2017;64:581–7.
Kim D. Low vitamin D status is associated with hypothyroid Hashimoto’s thyroiditis. Hormones. 2016;15:385–93.
Mazokopakis EE, Papadomanolaki MG, Tsekouras KC, Evangelopoulos AD, Kotsiris DA, Tzortzinis AA. Is vitamin D related to pathogenesis and treatment of Hashimoto’s thyroiditis? Hell J Nucl Med. 2015;18:222–7.
Bozkurt NC, Karbek B, Ucan B, Sahin M, Cakal E, Ozbek M, et al. The association between severity of vitamin D deficiency and Hashimoto’s thyroiditis. Endocr Pract. 2013;19:479–84.
Chen W, Lin H, Wang M. Immune intervention effects on the induction of experimental autoimmune thyroiditis. J Huazhong Univ Sci Technol Med Sci. 2002;22:343–5, 354.
Liu S, Xiong F, Liu EM, Zhu M, Lei PY. [Effects of 1,25-dihydroxyvitamin D3 in rats with experimental autoimmune thyroiditis]. Nan Fang Yi Ke Da Xue Xue Bao. 2010;30:1573–6.
Jeffery LE, Raza K, Hewison M. Vitamin D in rheumatoid arthritis-towards clinical application. Nat Rev Rheumatol. 2016;12:201–10.
Hawrylowicz CM, O’Garra A. Potential role of interleukin-10-secreting regulatory T cells in allergy and asthma. Nat Rev Immunol. 2005;5:271–83.
Tourneur L, Damotte D, Marion S, Mistou S, Chiocchia G. IL-10 is necessary for FasL-induced protection from experimental autoimmune thyroiditis but not for FasL-induced immune deviation. Eur J Immunol. 2002;32:1292–9.
Demirbilek H, Kandemir N, Gonc EN, Ozon A, Alikasifoglu A, Yordam N. Hashimoto’s thyroiditis in children and adolescents: a retrospective study on clinical, epidemiological and laboratory properties of the disease. J Pediatr Endocrinol Metab. 2007;20:1199–205.
Chen S, Sims GP, Chen XX, Gu YY, Chen S, Lipsky PE. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J Immunol. 2007;179:1634–47.
Das M, Tomar N, Sreenivas V, Gupta N, Goswami R. Effect of vitamin D supplementation on cathelicidin, IFN-gamma, IL-4 and Th1/Th2 transcription factors in young healthy females. Eur J Clin Nutr. 2014;68:338–43.
Joshi S, Pantalena LC, Liu XK, Gaffen SL, Liu H, Rohowsky-Kochan C, et al. 1,25-dihydroxyvitamin D(3) ameliorates Th17 autoimmunity via transcriptional modulation of interleukin-17A. Mol Cell Biol. 2011;31:3653–69.
Zhang X, Wu X, Xiong L, Yi Z, He Q, He X, et al. Role of vitamin D3 in regulation of T helper cell 17 and regulatory T-cell balance in rats with immunoglobulin A nephropathy. Iran J Kidney Dis. 2014;8:363–70.
Farsani ZS, Behmanesh M, Sahraian MA. Interleukin-10 but not transforming growth factor-beta1 gene expression is up-regulated by vitamin D treatment in multiple sclerosis patients. J Neurol Sci. 2015;350:18–23.
Marinho A, Carvalho C, Boleixa D, Bettencourt A, Leal B, Guimaraes J, et al. Vitamin D supplementation effects on FoxP3 expression in T cells and FoxP3+/IL-17A ratio and clinical course in systemic lupus erythematosus patients: a study in a Portuguese cohort. Immunol Res. 2017;65:197–206.
Rafiee M, Gharagozloo M, Ghahiri A, Mehrabian F, Maracy MR, Kouhpayeh S, et al. Altered Th17/Treg ratio in recurrent miscarriage after treatment with paternal lymphocytes and vitamin D3: a double-blind placebo-controlled study. Iran J Immunol. 2015;12:252–62.
Giacomet V, Vigano A, Manfredini V, Cerini C, Bedogni G, Mora S, et al. Cholecalciferol supplementation in HIV-infected youth with vitamin D insufficiency: effects on vitamin D status and T-cell phenotype: a randomized controlled trial. HIV Clin Trials. 2013;14:51–60.
Kongsbak M, von Essen MR, Levring TB, Schjerling P, Woetmann A, Odum N, et al. Vitamin D-binding protein controls T cell responses to vitamin D. BMC Immunol. 2014;15:35.
Jeffery LE, Henley P, Marium N, Filer A, Sansom DM, Hewison M, et al. Decreased sensitivity to 1,25-dihydroxyvitamin D3 in T cells from the rheumatoid joint. J Autoimmun. 2018;88:50–60.
Terrier B, Derian N, Schoindre Y, Chaara W, Geri G, Zahr N, et al. Restoration of regulatory and effector T cell balance and B cell homeostasis in systemic lupus erythematosus patients through vitamin D supplementation. Arthritis Res Ther. 2012;14:R221.
Gonzalez AM, Sell KM, Ghigiarelli JJ, Spitz RW, Accetta MR, Mangine GT. Effect of multi-ingredient supplement containing satiereal, naringin, and vitamin D on body composition, mood, and satiety in overweight adults. J Diet Suppl. 2018;15:965–76.
Jerzynska J, Stelmach W, Rychlik B, Majak P, Podlecka D, Woicka-Kolejwa K, et al. Clinical and immunological effects of vitamin D supplementation during the pollen season in children with allergic rhinitis. Arch Med Sci. 2018;14:122–31.
Raed A, Bhagatwala J, Zhu H, Pollock NK, Parikh SJ, Huang Y, et al. Dose responses of vitamin D3 supplementation on arterial stiffness in overweight African Americans with vitamin D deficiency: a placebo controlled randomized trial. PLoS ONE. 2017;12:e0188424.
Acknowledgements
The authors acknowledge the School of Public Health, Tehran University of Medical Sciences, Tehran, Iran, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Iran, and Mazandaran University of Medical Sciences, Sari, Iran, for providing the facilities and funds via grant number 95-01-27-30905. The authors also wish to thank Ms. Salehi for her helpful comments and discussions and Mr. Ghanavati for his assistance in FACS analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This study was financially supported by grant no. 95-01-27-30905 from Research Administration Department, Tehran University of Medical Sciences (TUMS), Tehran, Iran and grant no. 2353 from Mazandaran University of Medical Sciences, Sari, Iran.
Ethical approval
The current study was approved by the Ethics Committee of Tehran University of Medical Sciences, Tehran, Iran (approval no. IRCT2016110130644N1).
Informed consent
Written informed consents were signed by all the patients.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nodehi, M., Ajami, A., Izad, M. et al. Effects of vitamin D supplements on frequency of CD4+ T-cell subsets in women with Hashimoto’s thyroiditis: a double-blind placebo-controlled study. Eur J Clin Nutr 73, 1236–1243 (2019). https://doi.org/10.1038/s41430-019-0395-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-019-0395-z
This article is cited by
-
Hypovitaminosis D in persons with Down syndrome and autism spectrum disorder
Journal of Neurodevelopmental Disorders (2023)
-
How does Hashimoto’s thyroiditis affect bone metabolism?
Reviews in Endocrine and Metabolic Disorders (2023)
-
Assessment of the reporting quality of randomised controlled trials for vitamin D supplementation in autoimmune thyroid disorders based on the CONSORT statement
Endocrine (2022)