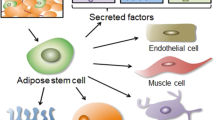
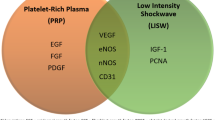
Abstract
The prevalence of erectile dysfunction (ED) has increased in recent decades. Although many treatments offer some benefits for patients with ED, unmet therapeutic needs remain, and promising new approaches are under investigation. One of these approaches is the use of stem-cell (SC) therapy for ED. We comprehensively reviewed the published literature and ongoing phase 1 and phase 2 trials and identified 27 trials by using SC therapy to treat ED. Of the 27 trials, three have been withdrawn, nine have published results, six are complete but without published results, and nine trials are ongoing or have an “unknown” status. Our analysis revealed that SC therapy represents a promising option to treat ED, although published data exist for less than 100 patients. Large placebo-controlled trials with longer follow-up are needed to confirm the long-term safety and efficacy of SC therapy for ED.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nicolosi A, Moreira ED Jr., Shirai M, Bin Mohd Tambi MI, Glasser DB. Epidemiology of erectile dysfunction in four countries: cross-national study of the prevalence and correlates of erectile dysfunction. Urology. 2003;61:201–6.
Korneyev IA, Alexeeva TA, Al-Shukri SH, Bernikov AN, Erkovich AA, Kamalov AA, et al. Prevalence and risk factors for erectile dysfunction and lower urinary tract symptoms in Russian Federation men: analysis from a national population-based multicenter study. Int J Impot Res. 2016;28:74–9.
Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol. 1994;151:54–61.
Bacon CG, Mittleman MA, Kawachi I, Giovannucci E, Glasser DB, Rimm EB. A prospective study of risk factors for erectile dysfunction. J Urol. 2006;176:217–21.
Selvin E, Burnett AL, Platz EA. Prevalence and risk factors for erectile dysfunction in the US. Am J Med. 2007;120:151–7.
McCabe MP, Sharlip ID, Lewis R, Atalla E, Balon R, Fisher AD, et al. Risk factors for sexual dysfunction among women and men: a consensus statement from the fourth international consultation on sexual medicine 2015. J Sex Med. 2016;13:153–67.
Mangir N, Turkeri L. Stem cell therapies in post-prostatectomy erectile dysfunction: a critical review. Can J Urol. 2017;24:8609–19.
Garaffa G, Trost LW, Serefoglu EC, Ralph D, Hellstrom WJ. Understanding the course of Peyronie’s disease. Int J Clin Pract. 2013;67:781–8.
Lopez JA, Jarow JP. Penile vascular evaluation of men with Peyronie’s disease. J Urol. 1993;149:53–5.
Burnett AL, Nehra A, Breau RH, Culkin DJ, Faraday MM, Hakim LS, et al. Erectile dysfunction: AUA Guideline. J Urol. 2018;200:633–41.
Melnik T, Soares BG, Nasselo AG. The effectiveness of psychological interventions for the treatment of erectile dysfunction: systematic review and meta-analysis, including comparisons to sildenafil treatment, intracavernosal injection, and vacuum devices. J Sex Med. 2008;5:2562–74.
McMahon CN, Smith CJ, Shabsigh R. Treating erectile dysfunction when PDE5 inhibitors fail. BMJ. 2006;332:589–92.
Sun DZ, Abelson B, Babbar P, Damaser MS. Harnessing the mesenchymal stem cell secretome for regenerative urology. Nat Rev Urol. 2019;16:363–75.
Wu J, Izpisua Belmonte JC. Stem cells: a renaissance in human biology research. Cell. 2016;165:1572–85.
Wagers AJ, Weissman IL. Plasticity of adult stem cells. Cell. 2004;116:639–48.
Friedenstein AJ, Chailakhyan RK, Latsinik NV, Panasyuk AF, Keiliss-Borok IV. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation. 1974;17:331–40.
Ding DC, Shyu WC, Lin SZ. Mesenchymal stem cells. Cell Transplant. 2011;20:5–14.
Caplan AI. Mesenchymal stem cells. J Orthop Res. 1991;9:641–50.
Caplan AI, New MSC. MSCs as pericytes are sentinels and gatekeepers. J Orthop Res. 2017;35:1151–9.
Bochinski D, Lin GT, Nunes L, Carrion R, Rahman N, Lin CS, et al. The effect of neural embryonic stem cell therapy in a rat model of cavernosal nerve injury. BJU Int. 2004;94:904–9.
Hou QL, Ge MY, Zhang CD, Tian DD, Wang LK, Tian HZ, et al. Adipose tissue-derived stem cell therapy for erectile dysfunction in rats: a systematic review and meta-analysis. Int Urol Nephrol. 2017;49:1127–37.
Kim IG, Piao S, Lee JY, Hong SH, Hwang TK, Kim SW, et al. Effect of an adipose-derived stem cell and nerve growth factor-incorporated hydrogel on recovery of erectile function in a rat model of cavernous nerve injury. Tissue Eng Part A. 2013;19:14–23.
Lee SH, Kim IG, Jung AR, Shrestha KR, Lee JH, Park KD, et al. Combined effects of brain-derived neurotrophic factor immobilized poly-lactic-co-glycolic acid membrane with human adipose-derived stem cells and basic fibroblast growth factor hydrogel on recovery of erectile dysfunction. Tissue Eng Part A. 2014;20:2446–54.
Levy JA, Marchand M, Iorio L, Zribi G, Zahalsky MP. Effects of stem cell treatment in human patients with Peyronie disease. J Am Osteopath Assoc. 2015;115:e8–13.
Lander EB, Berman MH, See JR. Stromal vascular fraction combined with shock wave for the treatment of Peyronie’s disease. Plast Reconstr Surg Glob Open. 2016;4:e631.
Bahk JY, Jung JH, Han H, Min SK, Lee YS. Treatment of diabetic impotence with umbilical cord blood stem cell intracavernosal transplant: preliminary report of 7 cases. Exp Clin Transplant. 2010;8:150–60.
Levy JA, Marchand M, Iorio L, Cassini W, Zahalsky MP. Determining the feasibility of managing erectile dysfunction in humans with placental-derived stem cells. J Am Osteopath Assoc. 2016;116:e1–5.
Yiou R, Hamidou L, Birebent B, Bitari D, Le Corvoisier P, Contremoulins I, et al. Intracavernous injections of bone marrow mononucleated cells for postradical prostatectomy erectile dysfunction: final results of the INSTIN clinical trial. Eur Urol Focus. 2017;3:643–5.
Yiou R, Hamidou L, Birebent B, Bitari D, Lecorvoisier P, Contremoulins I, et al. Safety of intracavernous bone marrow-mononuclear cells for postradical prostatectomy erectile dysfunction: an open dose-escalation pilot study. Eur Urol. 2016;69:988–91.
Haahr MK, Jensen CH, Toyserkani NM, Andersen DC, Damkier P, Sorensen JA, et al. Safety and potential effect of a single intracavernous injection of autologous adipose-derived regenerative cells in patients with erectile dysfunction following radical prostatectomy: an open-label phase I clinical trial. EBioMedicine. 2016;5:204–10.
Al Demour S, Jafar H, Adwan S, AlSharif A, Alhawari H, Alrabadi A, et al. Safety and potential therapeutic effect of two intracavernous autologous bone marrow derived mesenchymal stem cells injections in diabetic patients with erectile dysfunction: an open label phase I Clinical Trial. Urol Int. 2018;101:358–65.
Protogerou V, Michalopoulos E, Mallis P, Gontika I, Dimou Z, Liakouras C, et al. Administration of adipose derived mesenchymal stem cells and platelet lysate in erectile dysfunction: a single center pilot study. Bioengineering. 2019;6:21.
Albersen M, Fandel TM, Lin G, Wang G, Banie L, Lin CS, et al. Injections of adipose tissue-derived stem cells and stem cell lysate improve recovery of erectile function in a rat model of cavernous nerve injury. J Sex Med. 2010;7:3331–40.
Sun C, Lin H, Yu W, Li X, Chen Y, Qiu X, et al. Neurotrophic effect of bone marrow mesenchymal stem cells for erectile dysfunction in diabetic rats. Int J Androl. 2012;35:601–7.
Breitbach M, Bostani T, Roell W, Xia Y, Dewald O, Nygren JM, et al. Potential risks of bone marrow cell transplantation into infarcted hearts. Blood. 2007;110:1362–9.
Kunter U, Rong S, Boor P, Eitner F, Muller-Newen G, Djuric Z, et al. Mesenchymal stem cells prevent progressive experimental renal failure but maldifferentiate into glomerular adipocytes. J Am Soc Nephrol. 2007;18:1754–64.
Foudah D, Redaelli S, Donzelli E, Bentivegna A, Miloso M, Dalpra L, et al. Monitoring the genomic stability of in vitro cultured rat bone-marrow-derived mesenchymal stem cells. Chromosome Res. 2009;17:1025–39.
Jeong JO, Han JW, Kim JM, Cho HJ, Park C, Lee N, et al. Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ Res. 2011;108:1340–7.
Shauly O, Gould DJ, Patel KM. Emerging nonsurgical and surgical techniques to treat erectile dysfunction: a systematic review of treatment options and published outcomes. J Plast Reconstr Aesthet Surg. 2019;72:532–8.
Gur S, Abdel-Mageed AB, Sikka SC, Hellstrom WJG. Advances in stem cell therapy for erectile dysfunction. Expert Opin Biol Ther. 2018;18:1137–50.
Knoepfler PS. From bench to FDA to bedside: US regulatory trends for new stem cell therapies. Adv Drug Deliv Rev. 2015;82-83:192–6.
Daley GQ, Hyun I, Apperley JF, Barker RA, Benvenisty N, Bredenoord AL, et al. Setting global standards for stem cell research and clinical translation: the 2016 ISSCR guidelines. Stem Cell Rep. 2016;6:787–97.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial affiliation or involvement with any organization with a financial interest/conflict associated with the studies discussed in this paper. This includes expert testimony, employment, honoraria, consultancies, stock options or ownership, grants or patents pending/received, or royalties.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, M., von Schwarz, E.R. Stem-cell therapy for erectile dysfunction: a review of clinical outcomes. Int J Impot Res 33, 271–277 (2021). https://doi.org/10.1038/s41443-020-0279-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41443-020-0279-8