Abstract
Individuals at clinical high risk for psychosis (CHR) report a maladaptive self-concept—with more negative and less positive self-beliefs—linked to clinical symptoms and functional impairment. Alterations have also been reported in brain networks associated with intrinsic (cortical midline structures, CMS) and extrinsic (sensorimotor network, SMN) self-processing. Theoretical accounts of multiple levels of self-experience in schizophrenia suggest that interactions between these networks would be relevant for self-beliefs. This study tested whether self-beliefs related to resting-state functional connectivity within and between the CMS and SMN. Participants were 56 individuals meeting CHR criteria and 59 matched healthy community participants (HC). Pearson correlations examined potential mediators and outcomes. The CHR group reported more negative and less positive self-beliefs. Greater resting-state functional connectivity between the posterior CMS (posterior cingulate cortex) and the SMN was associated with less positive self-beliefs in CHR, but more positive self-beliefs in HC. Attenuated negative symptoms and poorer social functioning were associated with CMS-SMN connectivity (trend level after FDR-correction) and self-beliefs. Reduced connectivity between the left and right PCC was associated with lower positive self-beliefs in CHR, although this effect was specific to very low levels of positive self-beliefs. Left-right PCC connectivity did not correlate with outcomes. Dynamic interactions between intrinsic and extrinsic self-processing supported positive self-beliefs in typically developing youth while undermining positive self-beliefs in CHR youth. Implications are discussed for basic self-fragmentation, narrative self-related metacognition, and global belief updating. Interventions for self-processing may be beneficial in the CHR syndrome.
Similar content being viewed by others
Introduction
Individuals with schizophrenia tend to hold unusually strong negative beliefs and few positive beliefs about themselves, forming a maladaptive self-concept with low global self-esteem1,2 and cognitive feedback loops which maintain psychotic symptoms3,4. The same pattern of maladaptive self-concept, with more negative and less positive self-beliefs, has been observed in the clinical high risk for psychosis syndrome (CHR), a high risk state that often predates the onset of psychotic disorders5,6,7. Crucially, longitudinal studies have found that self-beliefs track the pathogenic mechanisms leading toward psychotic disorders: in CHR samples, self-beliefs become increasingly maladaptive over time for individuals who experience worsening symptoms8 or convert to a psychotic disorder7. The specific processes associated with dysfunctional self-concept in the CHR syndrome are therefore likely to inform the overall progression of psychotic disorders and suggest new avenues for intervention in this critical population. However, the complex multilevel structure of self-experience, together with impaired cognitive insight9 and self-disturbances10 in the CHR syndrome, may limit the utility of self-report or introspection to access mechanisms underlying self-beliefs in this population.
Resting-state functional connectivity between relevant brain networks can shed light on neural mechanisms underlying self-beliefs, independent of individuals’ explicit insight into these processes11. Self-disturbances have been observed in psychotic disorders affecting two distinct levels of the self, with links to relevant brain networks. The basic or minimal self (i.e., the prereflective experience of subjectivity in the present moment) is fundamentally altered in psychotic disorders, chiefly in a “heightened awareness of aspects of experience that are normally tacit or implicit” and “a weakened sense of existing as a subject of awareness”10,12,13. Similar basic self-disturbances have been observed in the CHR syndrome10. The narrative or autobiographical self, by contrast, is a temporally extended consciousness of oneself as an object of experience—as an individual with traits, goals, values, social roles, and so on13,14. Disruptions in the narrative self have been observed in psychotic disorders from various perspectives including metacognitive self-awareness15,16, dialogical or dialectical self-awareness (i.e., self-understanding based on one’s position in the physical, social, and cultural environment)16,17, lifespan identity development18, and narrative identity19. Self-beliefs would most likely be located at the narrative level of self, as self-schemas20,21 or “meta-positions”17 representing one’s characteristic ways of being in the world.
Both levels of self entail some amount of integration between internal cognitive-affective experiences and interactions with the external world. The basic self is most commonly associated with processing of internal self-referential information (e.g., self-related thoughts and emotions)22. However, the basic self is “most of the time an acting self” engaged in real or imagined movement, perception, and proprioception23,24 embedded in a “self-world structure” in which self-experience is juxtaposed against the backdrop of a reliable, predictable external world13,25. Integration of intrinsic and extrinsic processing supports the normative functioning of the basic self, and fragmentation of intrinsic and extrinsic processing may underlie the self-disturbances observed in schizophrenia11. Similarly, the narrative self is built on the foundations of the basic self13, and is theorized to emerge over time as basic self-experience co-occurs with encoding and retrieval of autobiographical memories14,21,26. Therefore, narrative self-experience should also entail integration of intrinsic and extrinsic processing.
And in fact, narrative self-experience probably involves even more intrinsic-extrinsic integration than does basic self-experience. The primary ingredients of the narrative self are socially constructed autobiographical memories that define “the self, other people, and typical interactions with others and the surrounding world…drawn largely from the influences of familial and peer socialization, schooling, and religion, as well as [culture]”21. These ingredients are particularly relevant in the CHR age range, a developmental period in which youth form the initial autobiographical memories on which self-beliefs are based27,28. Empirically, personally-relevant memories in middle-late adolescence (ages 14–18) focus on academic and sports performance, physical appearance, grooming, and adherence to social norms28, changing focus to career choices, educational attainment, and dating relationships in emerging adulthood (ages 18–25)29,30. These memory categories plainly entail interactions with others in the external world.
Moreover, the metacognitive processes that assemble autobiographical memories into a narrative self are also thought to be intersubjective and dialectical. These processes first appear through dialogue, in conversations with caregivers in early childhood31. Self-referential metacognition then develops “via intersubjectivity, either formed explicitly with and in the company of others or in the context of implied or imagined others”32. Metacognition evolves into an awareness of multiple ways in which the self is positioned relative to others and the world17. This awareness then supports a sense of personal agency as individuals balance multiple self-positions to organize cognition, emotion, and behavior toward coherent personal goals17,21. The metacognitive processes that form a narrative self from basic self-experience and autobiographical memory would thus seem to rely heavily on integration of intrinsic and extrinsic self-processing.
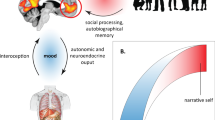
In the brain, self-referential processing of internal cognitive-affective experiences is most commonly associated with the cortical midline structures (CMS), a circuit comprising major components of the default mode network including the medial prefrontal cortex (mPFC), anterior cingulate cortex (ACC), and posterior cingulate cortex (PCC)33,34. Processing of real or imagined interactions with the external environment is most associated with the sensorimotor network (SMN), centered on the precentral gyrus (primary motor cortex) and postcentral gyrus (primary sensory cortex)11,24,35,36. Interactions between the CMS and SMN, passing through hub regions including the posterior cingulate cortex37, support a healthy integration of intrinsic- and extrinsic self-processing11. In schizophrenia, the CMS are typically found to be elevated in activity and connectivity at rest relative to control samples38,39,40,41,42, with a lack of normative task-based suppression43. These patterns relate to clinical symptoms of schizophrenia including hallucinations44,45,46 and delusions47,48, as well as associated features including aberrant salience49, faulty emotional appraisals47, impaired insight50,51, and cognitive impairment43. Similarly, SMN hypoactivity has been observed in schizophrenia and has been linked to global hallucination severity52 as well as specific experiences of alien control53,54, thought insertion54, and auditory hallucinations55. Finally, interactions between the CMS and SMN are disrupted in schizophrenia56,57,58. Functional isolation of the two networks and reduced network modularity have been theorized to result in self-fragmentation (i.e., failure to integrate intrinsic and extrinsic self-processing), which in turn contributes to positive and negative symptoms, impaired insight, and interpersonal problems11.
Resting-state functional connectivity within and between the CMS and SMN appears to be disrupted in the CHR syndrome as well, although fewer studies have examined these processes in CHR. Several CHR studies have reported increased CMS activation, increased CMS connectivity, and attenuated CMS task-based suppression, similar to common findings in schizophrenia22,59,60,61. Increased CMS resting-state connectivity has been linked directly to impaired clinical insight59, while lack of CMS task-based suppression has been linked to reality distortion and impaired cognition61. Connectivity patterns may relate to other alterations in intrinsic self-processing observed behaviorally in CHR, including in self-referential reasoning biases62, impaired emotion awareness63, and heightened self-certainty9. The SMN was also found to have reduced connectivity at rest in one CHR sample64, which may relate to deficits observed behaviorally in motor coordination65,66,67, motor agency68, dyskinesia65,69, and sensorimotor gating70,71,72. To date, interactions between the CMS and SMN have not been studied in the CHR literature.
The available evidence in schizophrenia and the CHR syndrome suggests that dysfunction in the self-concept should relate to dysconnectivity between the CMS (reflecting altered intrinsic self-processing) and the SMN (reflecting altered extrinsic self-processing). The current study aimed to test these relationships. Comparing a group meeting CHR criteria against a matched healthy comparison group, we hypothesized that a maladaptive self-concept in the CHR group would relate to: (a) increased connectivity within the CMS, reflecting increased or inefficient intrinsic self-processing; (b) reduced connectivity within the SMN, reflecting diminished extrinsic self-processing; and/or (c) altered connectivity between the CMS and SMN, reflecting faulty integration of intrinsic and extrinsic self-processing. Finally, in post hoc analyses, the current study tested relationships between connectivity findings, potential mediators (perceptual abnormalities and motor disturbances), and relevant outcomes (attenuated positive symptoms, attenuated negative symptoms, and social functioning).
Results
Participant characteristics
CHR participants (n = 56) were 39 (69%) Caucasian, 11 (20%) Hispanic/Latin, 3 (5%) Asian, and 3 (5%) other; 7 (13%) left-handed; with a mean age of 18.66 (SD = 1.74); a mean of 12.49 years of education (SD = 1.67); and a median annual household income of $40,000–59,999. As is common in CHR samples, some participants were prescribed psychiatric medication. In the CHR group, 24 participants (43%) were prescribed psychiatric medication, of whom 13 (23%) were prescribed more than one class of medication. SSRIs and stimulants were most common (n = 9, 16% each), followed by other antidepressants and antipsychotics (n = 8, 14% each) and mood stabilizers (n = 6, 11%). HC participants (n = 59) were 36 (61%) Caucasian, 12 (20%) Hispanic/Latin, 8 (14%) Asian, and 3 (5%) other; 4 (7%) left-handed; with a mean age of 18.36 (SD = 2.50); a mean of 12.39 years of education (SD = 2.48); and a median annual household income of $40,000–59,999. There were no significant differences between CHR and HC groups on any demographic variables (race, handedness, age, education, or income), confirmed by t-tests and chi-squared tests.
Group differences in self-beliefs
As shown in Fig. 1, CHR participants reported significantly more negative self-beliefs (mean = 0.92, SD = 0.85) than HC participants (mean = 0.19, SD = 0.26), t(64) = 6.16, p < 0.001, d = 1.16 [95% CI −1.59, −0.75]. CHR participants also reported significantly less positive self-beliefs (mean = 2.04, SD = 0.95) than HC participants (mean = 2.80, SD = 0.73), t(103) = −4.87, p < 0.001, d = −0.90 [95% CI −1.28, −0.51]. The distribution of negative self-beliefs suggests a likely floor effect in negative self-beliefs in the HC group.
Resting-state functional connectivity
Group contrast
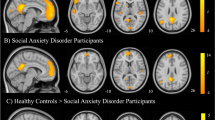
Regions of interest in the CMS and SMN are shown in Fig. 2 (see “Methods” below for more details). As shown in Table 1 and Fig. 3a, the CHR group showed a pattern of reduced interhemispheric connectivity within the CMS and SMN networks. Connectivity was reduced in CHR compared to HC between the left mPFC and right mPFC, t(55) = −3.01, pFDR = 0.039; the left PCC and right PCC, t(55) = −2.97, pFDR = 0.039; the left primary sensory cortex and right primary sensory cortex, t(55) = −2.92, pFDR = 0.039; and the left primary motor cortex and right primary motor cortex, t(55) = −2.78, pFDR = 0.044. These effects may reflect broad reductions in interhemispheric connectivity or specific reductions in connectivity within the CMS and SMN.
a Group differences in functional connectivity (CHR>HC). Colors indicate t-statistics, with blue indicating negative effects (i.e., CHR has less connectivity than HC) and red indicating positive effects (i.e., CHR has more connectivity than HC). b Group differences in the relationship between positive self-beliefs and functional connectivity (i.e., group x positive self-beliefs interaction). Effects plotted for the group (CHR>HC) x positive self-beliefs interaction. Negative effects (in blue) indicate that the relationship between positive self-beliefs and connectivity is more negative in CHR than HC, while positive effects (in red) indicate that the relationship between positive self-beliefs and connectivity is more positive in CHR than HC. In both a, b, effects were thresholded at pFDR < 0.05. Effects with pFDR > 0.05 are omitted from this figure. No effects were significant for the negative self-beliefs × group interaction.
Positive self-beliefs
As shown in Table 1, Figs. 3b, and 4, group (CHR>HC) interacted with positive self-beliefs in predicting functional connectivity between the left PCC and left primary motor area, HC r = 0.36, CHR r = −0.46, interaction t(113) = −4.66, pFDR < 0.001, and between the left PCC and left primary sensory area, HC r = 0.34, CHR r = −0.38, interaction t(113) = −4.06, pFDR = 0.001. A similar pattern was observed on the contralateral side, with self-beliefs predicting increased connectivity between the right PCC and right primary motor area, HC r = 0.28, CHR r = −0.42, interaction t(113) = −3.91, pFDR = 0.001, and between the right PCC and right primary sensory area, HC r = 0.25, CHR r = −0.43, interaction t(113) = −3.67, pFDR = 0.002. LOESS regression plots and quartile effect size plots (Fig. 4) indicated that these effects were consistent across the full range of positive self-beliefs. In sum, the groups showed opposite patterns: positive self-beliefs were associated with increased functional connectivity between CMS and SMN regions in the HC group, whereas they were associated with decreased functional connectivity between CMS and SMN regions in the CHR group.
a Interactions plotted as LOESS (locally estimated slopes) regression lines to highlight any nonlinearity. Colored bands indicate the standard error of the LOESS regression lines. b Interactions plotted as effect sizes of mean differences in connectivity (CHR>HC) in each quartile of positive self-belief scores. Effect sizes expressed as Cohen’s d, with error lines indicating 95% confidence intervals.
Group (CHR>HC) also interacted with positive self-beliefs in predicting functional connectivity between the left and right PCC, HC r = −0.29, CHR r = 0.26, t(113) = 2.75, pFDR = 0.039. LOESS regression plots and quartile effect size plots (Fig. 4) suggested that this effect was driven by large group differences when positive self-beliefs were very low.
Group did not interact with positive self-beliefs to predict functional connectivity between any other pairs of ROIs (right primary motor area – left primary motor area, right primary sensory area—left primary sensory area, right PCC—left primary motor area).
Negative self-beliefs
Group (CHR vs. HC) did not interact with negative self-beliefs in predicting functional connectivity between any pairs of ROIs.
External validators
As shown in Table 2, self-beliefs and connectivity scores did not correlate with potential mediators (perceptual abnormalities and motor disturbances, r ≤ |0.217|, pFDR ≥ 0.180). With respect to outcome variables, positive self-beliefs correlated with attenuated negative symptoms (r = −0.560, pFDR < 0.001) and social functioning (r = 0.429, pFDR = 0.004). Mean PCC – SMN connectivity also correlated at FDR-corrected trend level with attenuated negative symptoms (r = 0.275, pFDR = 0.094) and social functioning (r = −0.272, pFDR = 0.094). Self-beliefs and connectivity scores did not correlate with positive symptoms. Left–right PCC connectivity did not correlate with any potential mediators or outcomes.
Discussion
This was the first study to test interactions between the CMS and SMN in the clinical high risk for psychosis syndrome. The current study linked low positive self-beliefs to abnormal resting-state functional connectivity between brain networks involved in intrinsic and extrinsic self-processing (the CMS and SMN, respectively). Consistent with theoretical accounts of intrinsic and extrinsic processing in the basic and narrative self, CHR and HC groups showed opposite associations between positive self-beliefs and CMS-SMN functional connectivity. In the HC group, increased functional connectivity between these networks was associated with more positive self-beliefs; by contrast, in the CHR group, increased functional connectivity between these networks was associated with less positive self-beliefs.
Developmentally, in adolescence and emerging adulthood, the self-concept is shaped through engagement with the environment28,29,30. In the HC group, positive self-beliefs were highest when the posterior CMS and SMN were more connected, suggesting that increased CMS-SMN connectivity normatively has a positive effect on the self-concept. In the CHR group, by contrast, positive self-beliefs were lowest when the posterior CMS and SMN were more connected, suggesting that increased CMS-SMN connectivity has a negative effect on the self-concept in CHR. Although a variety of sensorimotor issues have been observed in other CHR samples65,66,67,68,69,71,72, the present study’s results were not explained by the severity of perceptual abnormalities or motor disturbances, implying that the results do not simply reflect faulty sensorimotor processing. Rather, CMS-SMN connectivity and self-beliefs were associated with increased negative symptoms and social functional impairment, implying that integration of intrinsic and extrinsic self-processing may prompt negative self-evaluations associated with real or imagined experiences of anhedonia, avolition, or social failure. Correlations between CMS-SMN connectivity, negative symptoms, and social function reached trend-level after FDR-correction, with effect sizes in the conventional small-medium range73, whereas correlations between self-beliefs, negative symptoms, and social function reached significance after FDR-correction with effect sizes in the conventional medium-large range73. This pattern suggests that self-beliefs may be more proximal to outcomes, perhaps even mediating relationships between connectivity and outcomes. Future studies powered for mediation analysis74 within a CHR sample would be valuable to test this hypothesis.
An interesting implication of this finding is that self-referential metacognition may be somewhat aversive to individuals at CHR. This would be in line with previous research showing that autobiographical reflection is aversive for individuals with schizotypal traits75. A global deficit in updating prior beliefs based on new sensory data has been proposed as a common mechanism underlying the experience of psychosis at multiple levels of analysis76,77, including in CHR78. In schizophrenia, low positive self-beliefs are maintained in part by a lack of updating in response to belief-inconsistent information, e.g., positive feedback about the self4,79. Deficits in belief updating also predict which individuals with schizophrenia will respond to metacognitive training80, suggesting a causal link between belief updating and metacognitive ability. If individuals at CHR experience self-referential metacognition as aversive, this may increase motivation to avoid interrogating prior beliefs, contributing to global problems with belief updating. Thus, the current findings may provide a common link tying together basic self-fragmentation, narrative self-referential metacognition, and global problems with belief updating in individuals at CHR.
In the present study, CHR individuals with low positive self-beliefs also appeared to have decreased functional connectivity between the left and right PCC. The PCC is a key posterior node of the CMS involved in directing attention toward internally- or externally-directed cognition, accessing autobiographical memory, and integrating multimodal information about the self from the CMS and other brain networks34,37. Reduced functional connectivity between the right and left PCC could plausibly accompany the processes described above. However, the left–right PCC interaction appeared to be driven by CHR participants with very low connectivity scores (see Fig. 3) and was unrelated to any potential mediators or outcomes. This finding’s theoretical and practical implications are therefore somewhat unclear based on the current study’s evidence. Interhemispheric PCC connectivity may be a valuable target for further research in the CHR syndrome.
The current study has some clinical implications. Cognitive-behavioral treatments have been suggested to target self-beliefs in psychosis4,7,79,81,82,83,84. The current study suggests that changes to self-beliefs in the CHR syndrome may require both interactions with the environment and reflection on those interactions. Individuals meeting CHR criteria experience more stressful or stigmatizing interactions with the environment than their normatively developing peers85,86. A key process for psychosocial interventions may be to support deliberate reflection on positive interactions with the environment. For instance, behavioral activation could be combined with mindfulness-based or metacognitive approaches. The emotion exposure component of the unified protocol for treatment of emotional disorders87 also combines these elements. The unified protocol has shown early promise in treatment for CHR, both in its original format88 and when enhanced with a mobile application89, and the current study provides further support for continued research and application of the unified protocol.
This study’s main strengths include its sample size, inclusion of a normatively developing comparison group, administration of a measure of specific positive and negative self-beliefs, and examination of multiple potential brain networks to test theoretically informed mechanisms linking internetwork connectivity to phenomenology and behavior. This study’s main limitations include its cross-sectional design, which limits causal inferences, and its reliance on a single self-report measure of self-beliefs. Future studies could include measures of other relevant self-concept processes or experimental tasks engaging self-referential processing. Similarly, we found no significant effects for negative self-beliefs. It may be that positive self-beliefs are uniquely tied to intrinsic and extrinsic self-processing. However, the HC group reported very few negative self-beliefs (see Fig. 1), and a statistical floor effect may have suppressed true associations between negative self-beliefs and functional connectivity. The BCSS was developed for use in adult schizophrenia populations, and the strong negative self-beliefs captured by the BCSS (e.g., “I am weak”) may not capture subtler changes in negative self-concept for CHR individuals and typically developing youth. Future studies may wish to employ more sensitive measures of negative self-beliefs.
In conclusion, the present study found alterations in functional connectivity between the CMS and SMN in the CHR syndrome and showed that these alterations track maladaptive self-beliefs. These results suggest mechanisms that integrate theoretical models of basic self-fragmentation, narrative self-referential metacognition, and global problems with belief updating in the development of psychosis. Treatments to enhance positive self-concept may be valuable in this population.
Methods
Participants
Participants were 56 help-seeking individuals classified as CHR based on the Structured Interview for Psychosis-Risk Syndromes (SIPS)90 and 59 matched healthy comparison (HC) youth. Participants were recruited for a study of psychosis risk in an academic research lab via newspaper, bus, and Craigslist ads, e-mail postings, and community professional referrals. Following SIPS criteria, participants were classified as CHR based on the presence of attenuated psychotic symptoms, brief psychotic experiences, or genetic risk and functional decline within the past year. Psychotic disorders were ruled out by administering the Structured Interview for DSM-IV Axis I Disorders91. This study’s participants were a subset of the participants in a prior study on core beliefs in CHR youth6, for whom resting-state fMRI data were available. All participants from that larger sample (N = 73 CHR and 73 HC) with complete resting-state fMRI data were included in the current study.
Materials
CHR status was assessed by the Structured Interview for Prodromal Syndromes (SIPS)90 The SIPS is a clinical interview which assesses positive, negative, disorganized, and general attenuated psychotic symptoms. The SIPS is widely used in CHR research92. Several variables were also extracted from the SIPS to test potential mediators and outcome variables. Single items assessed perceptual abnormalities (item P4) and motor disturbances (item G3) as potential mediators. Total scores on positive and negative symptom scales assessed overall symptom severity. Social functioning was also included as an outcome variable, assessed by the Global Functioning Scale—Social93, a clinician-rated scale of social functioning designed and validated for use with CHR samples.
Positive and negative self-beliefs were assessed by the Brief Core Schema Scales1. The BCSS is a 24-item self-report scale, with items for positive or negative beliefs about the self or others (e.g., “I am valuable”, “I am weak”, “others are hostile”) rated on a 5-point scale from 0 = “I do not believe this” to 4 = “I believe this totally”. Items are grouped into positive-self, negative-self, positive-other, and negative-other subscales, with 6 items on each subscale. The current study used only the positive-self and negative-self subscales, as beliefs about others were not relevant to study hypotheses. The BCSS have been validated in adult psychosis populations1, and have been used increasingly in CHR research e.g., refs. 5,7.
Procedures
Clinical assessment
Clinical interviews and self-report questionnaires were administered in person as part of a baseline assessment battery. Clinical interviews were administered by advanced graduate students trained to reliability standards of κ ≥ 0.80 through gold standard videos, observation, and supervised interviews.
MRI acquisition parameters
Subjects were instructed to rest and close their eyes during a T2*-weighted echo planar imaging functional protocol (5 min 34 s; 3.8 × 3.8 × 3.5 mm voxels; 33 slices; FOV = 240 mm; TR = 2000 ms; TE = 29 ms; FA = 75°). MR images using a standard 12-channel head coil with a 3-Tesla Siemens Tim Trio MRI Scanner (Siemens AG, Munich, Germany). For registration to Montreal Neurological Institute (MNI) template space, structural images were collected using a T1-weighted 3D magnetization sequence (0.512 mm isotropic voxels; 224 interleaved slices; Field of View [FOV] 256 mm; time to repetition [TR] 2400 ms; time to echo [TE] 2.01 ms; GRAPPA Factor 2; flip angle [FA] 8; collection orientation: sagittal plane).
Resting-state data processing
The FMRIB Software Library (FSL v6.0)94 was used to complete data processing. FSL was used to strip the skulls from images using brain extraction, high-pass filtering (100 s), and spatial smoothing (6-mm FWHM). Functional images were aligned to the Montreal Neurological Institute (MNI) 2-mm brain template. Temporal derivative regressors were calculated with artifact detection software (ART)95,96,97,98. Three translation and three rotation parameters were derived, as well as additional image specific confound regressors based on brain activation and framewise movement. Brain activation outliers were calculated using the mean global brain activity, i.e., the z-normalized mean signal across all voxels as a function of time. Outliers were defined as any frames where global mean signal exceeded 3 SD. Framewise motion measures (composite measure of total motion, or maximum voxel displacement, across translation and rotation) were used to identify motion outliers, defined as frames where the absolute value of motion exceeded 1 mm. The resultant motion regressors were entered into the model as a temporal derivative nuisance covariate at the subject level. Anatomical images were segmented into gray matter, white matter, and CSF with SPM12 to create masks for signal extraction. The Conn toolbox extracts five temporal components from the segmented cerebrospinal fluid (CSF) and white matter, which were entered as confound regressors in the subject‐level GLM. The temporal derivative of composite motion outliers (as described above) was calculated in the ART toolbox and included as a nuisance regressor. To reduce noise each subject’s anatomical image was segmented into gray matter, white matter, and CSF to reduce global signal.
Data analysis
Chi-squared tests (categorical variables) and two-tailed t-tests (continuous variables) tested group difference on demographic variables and self-beliefs.
CONN toolbox v.18.b99 and SPM12 (http://www.fil.ion.ucl.ac.uk/spm/software/spm12/) were used to conduct ROI-to-ROI analyses across regions in the CMS and SMN. CMS regions were the medial prefrontal cortex (combined mask of medial orbitofrontal and rostral anterior cingulate in the Desikan brain atlas) and posterior cingulate cortex. SMN regions were the primary motor cortex and primary sensory cortex (see Fig. 2). ROIs were defined in FreeSurfer100, which uses cortical surface landmarks to delineate cortical areas defined in the Desikan atlas101. A separate mask was defined for each ROI in both hemispheres, resulting in a total of 8 masks.
Connectivity between ROIs was calculated by averaging across voxel in each ROI to create an ROI level time course. The connection between regions was then calculated with Fischer-transformed bivariate coefficient by correlating each ROI time course.
ROI-to-ROI connectivity matrices were then extracted and analyzed. Group contrasts compared connectivity patterns between groups (CHR vs. HC). Then, between-group interactions tested whether the CHR group’s unusual distribution of self-beliefs was associated with unique patterns of connectivity which were not present in the HC group. Interaction effects were defined in a general linear model in which the interaction of group and self-beliefs predicted connectivity between all eight ROIs. Significant effects were defined at FDR-corrected p < 0.05 for seed-level two-tailed t-tests. Self-belief scores were grand-mean centered prior to analyses.
Subject-level ROI-to-ROI connectivity coefficients were extracted from the model and plotted to examine significant interaction effects. Interactions were plotted as LOESS regression plots and quartile effect size plots to show any potential nonlinearity in these effects. As a post hoc analysis of external validators, Pearson correlations between connectivity, self-beliefs, potential mediators (perceptual abnormalities, motor disturbances), and outcomes (attenuated positive symptoms, attenuated negative symptoms, and social functioning) were examined in the CHR group. To limit the number of correlations tested and increase their interpretability, a single PCC-SMN connectivity score was calculated for the post hoc analyses. This score was calculated as the mean of connectivity between CMS and SMN regions of interest with significant group x self-beliefs interactions (L PCC – L Sensory, L PCC – L Motor, R PCC – R Sensory, and R PCC – R Motor).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Fowler, D. et al. The Brief Core Schema Scales (BCSS): psychometric properties and associations with paranoia and grandiosity in non-clinical and psychosis samples. Psychol. Med. 36, 749–759 (2006).
Smith, B. et al. Emotion and psychosis: links between depression, self-esteem, negative schematic beliefs and delusions and hallucinations. Schizophr. Res. 86, 181–188 (2006).
Garety, P. A., Kuipers, E., Fowler, D., Freeman, D. & Bebbington, P. E. A cognitive model of the positive symptoms of psychosis. Psychol. Med. 31, 189–195 (2001).
Rector, N. A., Beck, A. T. & Stolar, N. The negative symptoms of schizophrenia: a cognitive perspective. Can. J. Psychiatry 50, 247–257 (2005).
Addington, J. & Tran, L. Using the brief core schema scales with individuals at clinical high risk of psychosis. Behav. Cogn. Psychother. 37, 227–231 (2009).
Cowan, H. R., McAdams, D. P. & Mittal, V. A. Core beliefs in healthy youth and youth at ultra high-risk for psychosis: Dimensionality and links to depression, anxiety, and attenuated psychotic symptoms. Dev. Psychopathol. 31, 379–392 (2019).
Stowkowy, J. et al. Core schemas in youth at clinical high risk for psychosis. Behav. Cogn. Psychother. 44, 203–213 (2016).
Patton, H. N., Cowan, H. R. & Mittal, V. A. Changes in core beliefs over time predict symptoms and functioning in clinical high risk for psychosis. Early Interv. Psychiatry 16, 311–315 (2022).
Dondé, C. et al. Cognitive insight in individuals with an at-risk mental state for psychosis: a meta-analysis. Early Interv. Psychiatry 15, 449–456 (2021).
Raballo, A., Poletti, M., Preti, A. & Parnas, J. The self in the spectrum: a meta-analysis of the evidence linking basic self-disorders and schizophrenia. Schizophr. Bull. 47, 1007–1017 (2021).
Ebisch, S. J. H. & Aleman, A. The fragmented self: imbalance between intrinsic and extrinsic self-networks in psychotic disorders. Lancet Psychiatry 3, 784–790 (2016).
Sass, L. A. & Parnas, J. Schizophrenia, consciousness, and the self. Schizophr. Bull. 29, 427–444 (2003).
Nelson, B., Parnas, J. & Sass, L. A. Disturbance of minimal self (Ipseity) in schizophrenia: clarification and current status. Schizophr. Bull. 40, 479–482 (2014).
Gallagher, S. Philosophical conceptions of the self: implications for cognitive science. Trends Cogn. Sci. 4, 14–21 (2000).
Lysaker, P.H. & Klion, R.E. Recovery, Meaning-Making, and Severe Mental Illness: A Comprehensive Guide to Metacognitive Reflection and Insight Therapy. (Routledge, 2017).
Mishara, A. L., Lysaker, P. H. & Schwartz, M. A. Self-disturbances in schizophrenia: history, phenomenology, and relevant findings from research on metacognition. Schizophr. Bull. 40, 5–12 (2014).
Lysaker, P. H., Buck, B. & Lysaker, J. T. Schizophrenia and alterations in the experience of self and agency: comparisons of dialogical and phenomenological views. Theory Psychol. 22, 738–755 (2012).
Conneely, M. et al. Understanding identity changes in psychosis: a systematic review and narrative synthesis. Schizophr. Bull. (2020) https://doi.org/10.1093/schbul/sbaa124.
Cowan, H. R., Mittal, V. A. & McAdams, D. P. Narrative identity in the psychosis spectrum: a systematic review and developmental model. Clin. Psychol. Review 88, 102067 (2021).
Markus, H. Self-schemata and processing information about the self. J. Pers. Soc. Psychol. 35, 63–78 (1977).
Conway, M. A., Singer, J. A. & Tagini, A. The self and autobiographical memory: correspondence and coherence. Soc. Cogn. 22, 491–529 (2004).
Damme, K. S. F., Pelletier‐Baldelli, A., Cowan, H. R., Orr, J. M. & Mittal, V. A. Distinct and opposite profiles of connectivity during self‐reference task and rest in youth at clinical high risk for psychosis. Hum. Brain Mapp. https://doi.org/10.1002/hbm.24595 (2019).
Jeannerod, M. The mechanism of self-recognition in humans. Behav. Brain Res. 142, 1–15 (2003).
Ferri, F., Frassinetti, F., Ardizzi, M., Costantini, M. & Gallese, V. A sensorimotor network for the bodily self. J. Cogn. Neurosci. 24, 1584–1595 (2012).
Henriksen, M. G. & Parnas, J. Self-disorders and schizophrenia: a phenomenological reappraisal of poor insight and noncompliance. Schizophr. Bull. 40, 542–547 (2014).
Damasio, A. The Feeling of What Happens: Body and Emotion in the Making of Consciousness. (Harcourt, 1999).
Sebastian, C., Burnett, S. & Blakemore, S.-J. Development of the self-concept during adolescence. Trends Cogn. Sci. 12, 441–446 (2008).
Cole, D. et al. The development of multiple domains of child and adolescent self-concept: a cohort sequential longitudinal design. Child Dev. 72, 1723–1746 (2001).
Schwartz, S. J., Zamboanga, B. L., Luyckx, K., Meca, A. & Ritchie, R. A. Identity in emerging adulthood: reviewing the field and looking forward. Emerg. Adulth. 1, 96–113 (2013).
Wood, D. et al. Emerging adulthood as a critical stage in the life course. in Handbook of Life Course Health Development (eds. Halfon, N., Forrest, C. B., Lerner, R. M. & Faustman, E. M.) 123–143 (Springer International Publishing, 2018). https://doi.org/10.1007/978-3-319-47143-3.
Nelson, K. & Fivush, R. The development of autobiographical memory, autobiographical narratives, and autobiographical consciousness. Psychol. Rep. 123, 71–96 (2020).
Lysaker, P. H. et al. Metacognitive function and fragmentation in schizophrenia: Relationship to cognition, self-experience and developing treatments. Schizophr. Res.: Cogn. 19, 100142 (2020).
Northoff, G. & Bermpohl, F. Cortical midline structures and the self. Trends Cogn. Sci. 8, 102–107 (2004).
van der Meer, L., Costafreda, S., Aleman, A. & David, A. S. Self-reflection and the brain: a theoretical review and meta-analysis of neuroimaging studies with implications for schizophrenia. Neurosci. Biobehav. Rev. 34, 935–946 (2010).
Christoff, K., Cosmelli, D., Legrand, D. & Thompson, E. Specifying the self for cognitive neuroscience. Trends Cogn. Sci. 15, 104–112 (2011).
Gallese, V. & Sinigaglia, C. The bodily self as power for action. Neuropsychologia 48, 746–755 (2010).
Leech, R. & Sharp, D. J. The role of the posterior cingulate cortex in cognition and disease. Brain 137, 12–32 (2014).
Anticevic, A. et al. The role of default network deactivation in cognition and disease. Trends Cogn. Sci. 16, 584–592 (2012).
Kindler, J. et al. Static and dynamic characteristics of cerebral blood flow during the resting state in schizophrenia. Schizophr. Bull. 41, 163–170 (2015).
Penner, J. et al. Medial prefrontal and anterior insular connectivity in early schizophrenia and major depressive disorder: a resting functional MRI evaluation of large-scale brain network models. Front. Hum. Neurosci. 10, 132 (2016).
Rikandi, E. et al. Connectivity of the precuneus-posterior cingulate cortex with the anterior cingulate cortex-medial prefrontal cortex differs consistently between control subjects and first-episode psychosis patients during a movie stimulus. Schizophr. Res. 199, 235–242 (2018).
Whitfield-Gabrieli, S. & Ford, J. M. Default mode network activity and connectivity in psychopathology. Annu. Rev. Clin. Psychol. 8, 49–76 (2012).
Zhou, L. et al. Inefficient DMN suppression in schizophrenia patients with impaired cognitive function but not patients with preserved cognitive function. Sci. Rep. 6, 21657 (2016).
Garrity, A. G. et al. Aberrant “default mode” functional connectivity in schizophrenia. Am. J. Psychiatry 164, 450–457 (2007).
Northoff, G. & Qin, P. How can the brain’s resting state activity generate hallucinations? A ‘resting state hypothesis’ of auditory verbal hallucinations. Schizophr. Res. 127, 202–214 (2011).
Wolf, N. D. et al. Dysconnectivity of multiple resting-state networks in patients with schizophrenia who have persistent auditory verbal hallucinations. J. Psychiatry Neurosci. 36, 366–374 (2011).
Holt, D. J. et al. Dysfunction of a cortical midline network during emotional appraisals in schizophrenia. Schizophr. Bull. 37, 164–176 (2011).
Larivière, S. et al. Altered functional connectivity in brain networks underlying self-referential processing in delusions of reference in schizophrenia. Psychiatry Res. Neuroimaging 263, 32–43 (2017).
Pankow, A. et al. Aberrant salience is related to dysfunctional self-referential processing in psychosis. Schizophr. Bull. 42, 67–76 (2016).
Raij, T. T., Riekki, T. J. J. & Hari, R. Association of poor insight in schizophrenia with structure and function of cortical midline structures and frontopolar cortex. Schizophr. Res. 139, 27–32 (2012).
van der Meer, L. et al. Insight in schizophrenia: involvement of self-reflection networks? Schizophr. Bull. 39, 1288–1295 (2013).
Shergill, S. S. et al. Functional magnetic resonance imaging of impaired sensory prediction in schizophrenia. JAMA Psychiatry 71, 28 (2014).
Spence, S. A. et al. A PET study of voluntary movement in schizophrenic patients experiencing passivity phenomena (delusions of alien control). Brain 120(Pt 11), 1997–2011 (1997).
Walsh, E., Oakley, D. A., Halligan, P. W., Mehta, M. A. & Deeley, Q. The functional anatomy and connectivity of thought insertion and alien control of movement. Cortex 64, 380–393 (2015).
Raij, T. T. & Riekki, T. J. J. Poor supplementary motor area activation differentiates auditory verbal hallucination from imagining the hallucination. Neuroimage Clin. 1, 75–80 (2012).
Alderson-Day, B. et al. Auditory hallucinations and the brain’s resting-state networks: findings and methodological observations. Schizophr. Bull. 42, 1110–1123 (2016).
Alonso-Solís, A. et al. Resting-state functional connectivity alterations in the default network of schizophrenia patients with persistent auditory verbal hallucinations. Schizophr. Res. 161, 261–268 (2015).
Schilbach, L. et al. Transdiagnostic commonalities and differences in resting state functional connectivity of the default mode network in schizophrenia and major depression. NeuroImage Clin. 10, 326–335 (2016).
Clark, S. V. et al. Stronger default mode network connectivity is associated with poorer clinical insight in youth at ultra high-risk for psychotic disorders. Schizophr. Res. 193, 244–250 (2018).
Shim, G. et al. Altered resting-state connectivity in subjects at ultra-high risk for psychosis: an fMRI study. Behav. Brain Functions 6, 1–11 (2010).
Wotruba, D. et al. Aberrant coupling within and across the default mode, task-positive, and salience network in subjects at risk for psychosis. Schizophr. Bull. 40, 1095–1104 (2014).
Broome, M. R. et al. Delusion formation and reasoning biases in those at clinical high risk for psychosis. Br. J. Psychiatry 191, s38–s42 (2007).
Kimhy, D. et al. The impact of emotion awareness and regulation on social functioning in individuals at clinical high risk for psychosis. Psychol. Med. 46, 2907–2918 (2016).
Du, Y. et al. Identifying functional network changing patterns in individuals at clinical high-risk for psychosis and patients with early illness schizophrenia: a group ICA study. Neuroimage-Clin. 17, 335–346 (2018).
Damme, K. S. F., Schiffman, J., Ellman, L. M. & Mittal, V. A. Sensorimotor and Activity Psychosis-Risk (SMAP-R) scale: an exploration of scale structure with replication and validation. Schizophr. Bull. 47, 332–343 (2021).
Dean, D. J., Walther, S., Bernard, J. A. & Mittal, V. A. Motor clusters reveal differences in risk for psychosis, cognitive functioning, and thalamocortical connectivity: evidence for vulnerability subtypes. Clin. Psychol. Sci. 6, 721–734 (2018).
Masucci, M. D., Lister, A., Corcoran, C. M., Brucato, G. & Girgis, R. R. Motor dysfunction as a risk factor for conversion to psychosis independent of medication use in a psychosis-risk cohort. J. Nerv. Ment. Dis. 206, 356–361 (2018).
Wilquin, H. & Delevoye-Turrell, Y. Motor agency: a new and highly sensitive measure to reveal agency disturbances in early psychosis. Plos One 7, e30449 (2012).
Mittal, V. A. et al. Markers of basal ganglia dysfunction and conversion to psychosis: neurocognitive deficits and dyskinesias in the prodromal period. Biol. Psychiatry 68, 93–99 (2010).
Bo, Q. et al. Impaired sensorimotor gating using the acoustic prepulse inhibition paradigm in individuals at a clinical high risk for psychosis. Schizophr. Bull. 47, 128–137 (2021).
Khan, A. & Powell, S. B. Sensorimotor gating deficits in “two-hit” models of schizophrenia risk factors. Schizophr. Res. 198, 68–83 (2018).
Quednow, B. B. et al. Impaired sensorimotor gating of the acoustic startle response in the prodrome of schizophrenia. Biol. Psychiatry 64, 766–773 (2008).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences. (Routledge, 1988). https://doi.org/10.4324/9780203771587.
Fritz, M. S. & MacKinnon, D. P. Required sample size to detect the mediated effect. Psychol. Sci. 18, 233–239 (2007).
Cicero, D. C. & Kerns, J. G. Unpleasant and pleasant referential thinking: relations with self-processing, paranoia, and other schizotypal traits. J. Res. Pers. 45, 208–218 (2011).
Heinz, A. et al. Towards a unifying cognitive, neurophysiological, and computational neuroscience account of schizophrenia. Schizophr. Bull. 45, 1092–1100 (2019).
Katthagen, T., Fromm, S., Wieland, L. & Schlagenhauf, F. Models of dynamic belief updating in psychosis—a review across different computational approaches. Front. Psychiatry 13, 814111 (2022).
Cole, D. M. et al. Atypical processing of uncertainty in individuals at risk for psychosis. NeuroImage: Clin. 26, 102239 (2020).
Strauss, G. P. & Gold, J. M. A new perspective on anhedonia in schizophrenia. Am. J. Psychiatry 169, 364–373 (2012).
Hauke, D. et al. Increased belief instability in psychotic disorders predicts treatment response to metacognitive training. Schizophr. Bull 48, 826–838 (2022).
Grant, P. M. & Beck, A. T. Defeatist beliefs as a mediator of cognitive impairment, negative symptoms, and functioning in schizophrenia. Schizophr. Bull. 35, 798–806 (2009).
Kesting, M.-L. & Lincoln, T. M. The relevance of self-esteem and self-schemas to persecutory delusions: a systematic review. Compr. Psychiatry 54, 766–789 (2013).
Palmier‐Claus, J., Dunn, G., Drake, R. & Lewis, S. The negative and positive self: a longitudinal study examining self-esteem, paranoia and negative symptoms in individuals with first-episode psychosis. Early Interv. Psychiatry 5, 150–155 (2011).
Taylor, H. E. et al. Core schemas across the continuum of psychosis: a comparison of clinical and non-clinical groups. Behav. Cogn. Psychother. 42, 718–730 (2014).
Saleem, M. M. et al. Perceived discrimination in those at clinical high risk for psychosis. Early Interv. Psychiatry 8, 77–81 (2014).
Trotman, H. D. et al. Stress exposure and sensitivity in the clinical high-risk syndrome: Initial findings from the North American Prodrome Longitudinal Study (NAPLS). Schizophr. Res. 160, 104–109 (2014).
Barlow, D. H., Harris, B. A., Eustis, E. H. & Farchione, T. J. The unified protocol for transdiagnostic treatment of emotional disorders. World Psychiatry 19, 245–246 (2020).
Breitborde, N. J. K. et al. The Ohio State University Early Psychosis Intervention Center (EPICENTER) step-based care programme for individuals at clinical high risk for psychosis: study protocol for an observational study. BMJ Open 10, e034031 (2020).
Weintraub, M. J. et al. App-enhanced transdiagnostic CBT for adolescents with mood or psychotic spectrum disorders. J. Affect. Disord. 311, 319–326 (2022).
Miller, T. J. et al. Symptom assessment in schizophrenic prodromal states. Psychiatr. Q 70, 273–287 (1999).
First, M., Spitzer, M., Gibbon, M. & Williams, J. Structural Clinical Interview for DSM-IV Axis I Disorders (SCID-IV). (New York State Psychiatric Institute, Biometrics Research, 1997).
Fusar-Poli, P. et al. The psychosis high-risk state: a comprehensive state-of-the-art review. JAMA Psychiatry 70, 107–120 (2013).
Cornblatt, B. A. et al. Preliminary findings for two new measures of social and role functioning in the prodromal phase of schizophrenia. Schizophr. Bull. 33, 688–702 (2007).
Jenkinson, M., Beckmann, C. F., Behrens, T. E. J., Woolrich, M. W. & Smith, S. M. FSL. NeuroImage 62, 782–790 (2012).
Adolphs, R. Neural systems for recognizing emotion. Curr. Opin. Neurobiol. 12, 169–177 (2002).
Gallese, V. & Goldman, A. Mirror neurons and the simulation theory of mind-reading. Trends Cogn. Sci. 2, 493–501 (1998).
Mitchell, J. P., Macrae, C. N. & Banaji, M. R. Dissociable medial prefrontal contributions to judgments of similar and dissimilar others. Neuron 50, 655–663 (2006).
Nickerson, R. S. How we know—and sometimes misjudge—what others know: imputing one’s own knowledge to others. Psychol. Bull. 125, 737–759 (1999).
Whitfield-Gabrieli, S. & Nieto-Castanon, A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2, 125–141 (2012).
Fischl, B. FreeSurfer. NeuroImage 62, 774–781 (2012).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 31, 968–980 (2006).
Acknowledgements
This work was supported in part by the Canadian Institutes of Health Research (DFS-152268 to H.R.C.) and by the National Institutes of Health (R01MH112545-01, R01MH116039-01A1, and R01MH094650 to V.A.M.). The study’s sponsors were not involved in design, data collection, analysis, or report writing.
Author information
Authors and Affiliations
Contributions
All authors participated in the study design. H.R.C. and K.S.F.D. conducted analyses under the supervision of V.A.M. H.R.C. wrote the first draft of the manuscript. All authors contributed to the final manuscript and have approved it in its current version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cowan, H.R., Damme, K.S.F. & Mittal, V.A. Interactions between the cortical midline structures and sensorimotor network track maladaptive self-beliefs in clinical high risk for psychosis. Schizophr 8, 74 (2022). https://doi.org/10.1038/s41537-022-00279-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-022-00279-z