Abstract
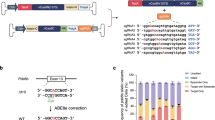
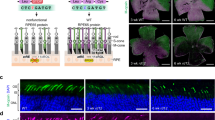
Cytosine base editors and adenine base editors (ABEs) can correct point mutations predictably and independent of Cas9-induced double-stranded DNA breaks (which causes substantial indel formation) and homology-directed repair (which typically leads to low editing efficiency). Here, we show, in adult mice, that a subretinal injection of a lentivirus expressing an ABE and a single-guide RNA targeting a de novo nonsense mutation in the Rpe65 gene corrects the pathogenic mutation with up to 29% efficiency and with minimal formation of indel and off-target mutations, despite the absence of the canonical NGG sequence as a protospacer-adjacent motif. The ABE-treated mice displayed restored RPE65 expression and retinoid isomerase activity, and near-normal levels of retinal and visual functions. Our findings motivate the further testing of ABEs for the treatment of inherited retinal diseases and for the correction of pathological mutations with non-canonical protospacer-adjacent motifs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the paper and its Supplementary Information. The deep-sequencing data are available from the Sequence Read Archive under accession number PRJNA644016.
Change history
12 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41551-020-00652-2
References
Cremers, F. P., van den Hurk, J. A. & den Hollander, A. I. Molecular genetics of Leber congenital amaurosis. Hum. Mol. Genet. 11, 1169–1176 (2002).
Den Hollander, A. I., Roepman, R., Koenekoop, R. K. & Cremers, F. P. M. Leber congenital amaurosis: genes, proteins and disease mechanisms. Prog. Retin. Eye Res. 27, 391–419 (2008).
Miraldi Utz, V., Coussa, R. G., Antaki, F. & Traboulsi, E. I. Gene therapy for RPE65-related retinal disease. Ophthalmic Genet. 39, 671–677 (2018).
Maguire, A. M. et al. Efficacy, safety, and durability of voretigene neparvovec-rzyl in RPE65 mutation-associated inherited retinal dystrophy: results of phase 1 and 3 trials. Ophthalmology 126, 1273–1285 (2019).
Bainbridge, J. W. et al. Long-term effect of gene therapy on Leber’s congenital amaurosis. N. Engl. J. Med. 372, 1887–1897 (2015).
Jacobson, S. G. et al. Improvement and decline in vision with gene therapy in childhood blindness. N. Engl. J. Med. 372, 1920–1926 (2015).
Cideciyan, A. V. et al. Human retinal gene therapy for Leber congenital amaurosis shows advancing retinal degeneration despite enduring visual improvement. Proc. Natl Acad. Sci. USA 110, E517–E525 (2013).
Gardiner, K. L. et al. Long-term structural outcomes of late-stage RPE65 gene therapy. Mol. Ther. 28, 266–278 (2020).
Hsu, P. D., Lander, E. S. & Zhang, F. Development and applications of CRISPR–Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Doudna, J. A. & Charpentier, E. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Chapman, J. R., Taylor, M. R. & Boulton, S. J. Playing the end game: DNA double-strand break repair pathway choice. Mol. Cell 47, 497–510 (2012).
Lin, S., Staahl, B. T., Alla, R. K. & Doudna, J. A. Enhanced homology-directed human genome engineering by controlled timing of CRISPR/Cas9 delivery. eLife 3, e04766 (2014).
Cox, D. B., Platt, R. J. & Zhang, F. Therapeutic genome editing: prospects and challenges. Nat. Med. 21, 121–131 (2015).
Rees, H. A. & Liu, D. R. Base editing: precision chemistry on the genome and transcriptome of living cells. Nat. Rev. Genet. 19, 770–788 (2018).
Gaudelli, N. M. et al. Programmable base editing of A•T to G•C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Villiger, L. et al. Treatment of a metabolic liver disease by in vivo genome base editing in adult mice. Nat. Med. 24, 1519–1525 (2018).
Rossidis, A. C. et al. In utero CRISPR-mediated therapeutic editing of metabolic genes. Nat. Med. 24, 1513–1518 (2018).
Osborn, M. J. et al. Base editor correction of COL7A1 in recessive dystrophic epidermolysis bullosa patient-derived fibroblasts and iPSCs. J. Invest. Dermatol. 140, 338–347 (2020).
Ryu, S.-M. et al. Adenine base editing in mouse embryos and an adult mouse model of Duchenne muscular dystrophy. Nat. Biotechnol. 36, 536–539 (2018).
Pang, J. J. et al. Retinal degeneration 12 (rd12): a new, spontaneously arising mouse model for human Leber congenital amaurosis (LCA). Mol. Vis. 11, 152–162 (2005).
Zhong, Z. et al. Seven novel variants expand the spectrum of RPE65-related Leber congenital amaurosis in the Chinese population. Mol. Vis. 25, 204–214 (2019).
Redmond, T. M. et al. Rpe65 is necessary for production of 11-cis-vitamin A in the retinal visual cycle. Nat. Genet. 20, 344–351 (1998).
Jo, D. H. et al. CRISPR–Cas9-mediated therapeutic editing of Rpe65 ameliorates the disease phenotypes in a mouse model of Leber congenital amaurosis. Sci. Adv. 5, eaax1210 (2019).
Hu, J. H. et al. Evolved Cas9 variants with broad PAM compatibility and high DNA specificity. Nature 556, 57–63 (2018).
Koblan, L. W. et al. Improving cytidine and adenine base editors by expression optimization and ancestral reconstruction. Nat. Biotechnol. 36, 843–846 (2018).
Hsu, P. D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 31, 827–832 (2013).
Jiang, W., Bikard, D., Cox, D., Zhang, F. & Marraffini, L. A. RNA-guided editing of bacterial genomes using CRISPR–Cas systems. Nat. Biotechnol. 31, 233–239 (2013).
Zhang, Y. et al. Comparison of non-canonical PAMs for CRISPR/Cas9-mediated DNA cleavage in human cells. Sci. Rep. 4, 5405 (2014).
Levy, J. M. et al. Cytosine and adenine base editing of the brain, liver, retina, heart and skeletal muscle of mice via adeno-associated viruses. Nat. Biomed. Eng. 4, 97–110 (2020).
Puppo, A. et al. Retinal transduction profiles by high-capacity viral vectors. Gene Ther. 21, 855–865 (2014).
Tsai, S. Q. et al. CIRCLE-seq: a highly sensitive in vitro screen for genome-wide CRISPR–Cas9 nuclease off-targets. Nat. Methods 14, 607–614 (2017).
Xin, H., Wan, T. & Ping, Y. Off-targeting of base editors: BE3 but not ABE induces substantial off-target single nucleotide variants. Signal Transduct. Target. Ther. 4, 9 (2019).
Jin, S. et al. Cytosine, but not adenine, base editors induce genome-wide off-target mutations in rice. Science 364, 292–295 (2019).
Kiser, P. D., Golczak, M. & Palczewski, K. Chemistry of the retinoid (visual) cycle. Chem. Rev. 114, 194–232 (2014).
Benchorin, G., Calton, M. A., Beaulieu, M. O. & Vollrath, D. Assessment of murine retinal function by electroretinography. Bio Protoc. 7, e2218 (2017).
Leinonen, H. & Tanila, H. Vision in laboratory rodents—tools to measure it and implications for behavioral research. Behav. Brain Res. 352, 172–182 (2018).
Hofbauer, A. & Dräger, U. C. Depth segregation of retinal ganglion cells projecting to mouse superior colliculus. J. Comp. Neurol. 234, 465–474 (1985).
Wang, L., Sarnaik, R., Rangarajan, K., Liu, X. & Cang, J. Visual receptive field properties of neurons in the superficial superior colliculus of the mouse. J. Neurosci. 30, 16573–16584 (2010).
Kretschmer, F., Sajgo, S., Kretschmer, V. & Badea, T. C. A system to measure the optokinetic and optomotor response in mice. J. Neurosci. Methods 256, 91–105 (2015).
Miller, S. M. et al. Continuous evolution of SpCas9 variants compatible with non-G PAMs. Nat. Biotechnol. 38, 471–481 (2020).
Walton, R. T., Christie, K. A., Whittaker, M. N. & Kleinstiver, B. P. Unconstrained genome targeting with near-PAMless engineered CRISPR-Cas9 variants. Science 368, 290–296 (2020).
Lee, C. et al. CRISPR-pass: gene rescue of nonsense mutations using adenine base editors. Mol. Ther. 27, 1364–1371 (2019).
Acland, G. M. et al. Gene therapy restores vision in a canine model of childhood blindness. Nat. Genet. 28, 92–95 (2001).
Acland, G. M. et al. Long-term restoration of rod and cone vision by single dose rAAV-mediated gene transfer to the retina in a canine model of childhood blindness. Mol. Ther. 12, 1072–1082 (2005).
Jacobson, S. G. et al. Safety of recombinant adeno-associated virus type 2–RPE65 vector delivered by ocular subretinal injection. Mol. Ther. 13, 1074–1084 (2006).
Chelstowska, S., Widjaja-Adhi, M. A. K., Silvaroli, J. A. & Golczak, M. Impact of LCA-associated E14L LRAT mutation on protein stability and retinoid homeostasis. Biochemistry 56, 4489–4499 (2017).
Kitamura, T. et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp. Hematol. 31, 1007–1014 (2003).
Perez-Pinera, P. et al. RNA-guided gene activation by CRISPR–Cas9-based transcription factors. Nat. Methods 10, 973–976 (2013).
Walter, D. M. et al. Systematic in vivo inactivation of chromatin-regulating enzymes identifies Setd2 as a potent tumor suppressor in lung adenocarcinoma. Cancer Res. 77, 1719–1729 (2017).
Golczak, M., Kiser, P. D., Lodowski, D. T., Maeda, A. & Palczewski, K. Importance of membrane structural integrity for RPE65 retinoid isomerization activity. J. Biol. Chem. 285, 9667–9682 (2010).
Foik, A. T. et al. Retinal origin of electrically evoked potentials in response to transcorneal alternating current stimulation in the rat. Invest. Ophthalmol. Vis. Sci. 56, 1711–1718 (2015).
Brainard, D. H. The psychophysics toolbox. Spat. Vis. 10, 433–436 (1997).
Foik, A. T. et al. Detailed visual cortical responses generated by retinal sheet transplants in rats with severe retinal degeneration. J. Neurosci. 38, 10709–10724 (2018).
Acknowledgements
We thank A. Daruwalla, H. Hashimoto, A. Lewin and A. Browne for technical assistance and providing materials. We are also grateful to members of the Palczewski laboratory for helpful comments regarding this study. K.P. is the Leopold Chair of Ophthalmology at the Gavin Herbert Eye Institute, Department of Ophthalmology, University of California, Irvine. This research was supported in part by grants to K.P. from the National Institutes of Health (NIH) (nos. EY009339, EY027283, EY025451 and EY019312) and the Research to Prevent Blindness Stein Innovation Award. S.S. was supported by NIH grant nos. F30EY029136, T32GM007250 and T32EY024236. E.H.C. was supported by NIH grant nos. T32GM007250 and T32GM008803. H.L. was supported by Fight for Sight, the Eye and Tissue Bank Foundation (Finland), The Finnish Cultural Foundation and the Orion Research Foundation. G.A.N. was supported as a Howard Hughes Medical Institute fellow of the Helen Hay Whitney Foundation. P.D.K. was supported by the US Department of Veterans Affairs (I01BX004939). We also acknowledge support from a Research to Prevent Blindness unrestricted grant to the Department of Ophthalmology, University of California, Irvine.
Author information
Authors and Affiliations
Contributions
S.S., E.H.C. and K.P. conceived of the strategy and designed the experiments. S.S. and E.H.C. designed, performed and analysed the in vitro experiments. S.S., E.H.C., H.L., A.T.F., D.C.L., Z.D. and P.D.K. designed, performed and analysed the in vivo experiments. G.A.N., W.-H.Y. and D.R.L. performed and analysed the off-target activity. S.S., E.H.C. and K.P. wrote the manuscript. All authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
D.R.L. is a consultant and co-founder of Beam Therapeutics, Prime Medicine, Editas Medicine and Pairwise Plants, companies that use genome editing.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Figs. 1–9, Tables 1–4 and references.
Rights and permissions
About this article
Cite this article
Suh, S., Choi, E.H., Leinonen, H. et al. Restoration of visual function in adult mice with an inherited retinal disease via adenine base editing. Nat Biomed Eng 5, 169–178 (2021). https://doi.org/10.1038/s41551-020-00632-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-00632-6
This article is cited by
-
Engineered virus-like particles for transient delivery of prime editor ribonucleoprotein complexes in vivo
Nature Biotechnology (2024)
-
Precise genome-editing in human diseases: mechanisms, strategies and applications
Signal Transduction and Targeted Therapy (2024)
-
Rare genetic disorders in India: Current status, challenges, and CRISPR-based therapy
Journal of Biosciences (2024)
-
In vivo adenine base editing corrects newborn murine model of Hurler syndrome
Molecular Biomedicine (2023)
-
Comparison of retinal degeneration treatment with four types of different mesenchymal stem cells, human induced pluripotent stem cells and RPE cells in a rat retinal degeneration model
Journal of Translational Medicine (2023)