Abstract
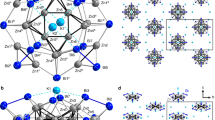
The concept of aromaticity was originally defined as a property of unsaturated, cyclic planar organic molecules like benzene, which gain stability by the inherent delocalization of 4n + 2 π-electrons over the ring atoms. Since then, π-aromaticity has been observed for a large variety of organic and inorganic non-metal compounds, yet, for molecules consisting only of metal atoms, it has remained restricted to systems with three to five atoms. Here, we present the straightforward synthesis of a metal 12-ring that exhibits 2π-aromaticity and has a ring current much stronger than that of benzene (6π) and equivalent to that of porphine (26π), despite these organic molecules having (much) larger numbers of π-electrons. Highly reducing reaction conditions allowed access to the heterometallic anion [Th@Bi12]4−, with interstitial Th4+ stabilizing a Bi128− moiety. Our results show that it is possible to design and generate substantial π-aromaticity in large metal rings, and we hope that such π-aromatic heavy-metal cycles will eventually find use in cluster-based reactions.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this Article and its Supplementary Information files. The structures of compounds 1–4 were determined by single-crystal X-ray diffraction. The crystallographic data have been deposited with the Cambridge Crystallographic Data Centre under CCDC numbers 1983070 (1), 1983072 (2), 1983073 (3) and 1983071 (4).
The optimized structures of all studied compounds are part of the Supplementary Information (separate zip file ‘Supplementary-Computational-Data.zip’): the coordinates of the optimized structures shown in Fig. 1, as well as their NICS values and ring current strengths, are provided in the Supplementary File ‘Fig1-OptimizedStructures-GIMIC-NICS.txt’. All calculated coordinates of the optimized structures of 2A and the compounds mentioned explicitly in the main text or Supplementary Information are provided in a Supplementary File ‘OptimizedStructures.txt’. The files comprise all necessary data for reproducing the values. All non-default parameters for the computational studies are given in the Supplementary Information together with the corresponding references of the used methods. For the default parameters of TURBOMOLE, such as the convergence criteria for structure optimizations, please see the manual at https://www.turbomole.org (retrieved 29 August 2020).
References
Hückel, E. Quantentheoretische Beiträge zum Benzolproblem. I. Die Elektronenkonfiguration des Benzols und verwandter Beziehungen. Z. Phys. 70, 204–286 (1931).
McNaught, A. D. & Wilkinson, A. IUPAC. Compendium of Chemical Terminology 2nd edn (Blackwell Scientific Publications, 1997); online version (2019) created by S. J. Chalk.
Schleyer, P. V. R. & Jiao, H. What is aromaticity? Pure Appl. Chem. 68, 209–221 (1996).
Gershoni-Poranne, R. & Stanger, A. Magnetic criteria of aromaticity. Chem. Soc. Rev. 44, 6597–6615 (2015).
Sundholm, D., Fliegl, H. & Berger, R. J. Calculation of magnetically induced current densities: theory and applications. Wiley Interdiscip. Rev. Comput. Mol. Sci. 6, 639–678 (2016).
Schleyer, P. V. R., Maerker, C., Dransfeld, A., Jiao, H. & Hommes, N. Nucleus-independent chemical shifts: a simple and efficient aromaticity probe. J. Am. Chem. Soc. 118, 6317–6318 (1996).
Sekiguchi, A., Matsuo, T. & Watanabe, H. Synthesis and characterization of a cyclobutadiene dianion dilithium salt: evidence for aromaticity. J. Am. Chem. Soc. 122, 5652–5653 (2000).
Wehrmann, R., Meyer, H. & Berndt, A. Diboriranides and a 1,3-diboraallyl system with B-H-B bridge. Angew. Chem. Int. Ed. 24, 788–790 (1985).
Zhu, Z., Wang, X., Olmstead, M. M. & Power, P. P. Synthesis and characterization of [Ar′GaC(Ph)CH]2 and K2[Ar′GaC(Ph)CH]2·OEt2: from digallene to digallacyclohexadiene to digallatabenzene. Angew. Chem. Int. Ed. 48, 2027–2030 (2009).
De Proft, F. et al. Ring currents as probes of the aromaticity of inorganic monocycles: P5−, As5−, S2N2, S3N3−, S4N3+, S4N42+, S5N5+, S42+ and Se42+. Chem. Eur. J. 10, 940–950 (2004).
Jin, J. et al. The [B3(NN)3]+ and [B3(CO)3]+ complexes featuring the smallest π-aromatic species B3+. Angew. Chem. Int. Ed. 55, 2078–2082 (2016).
Unverzagt, M. et al. Carbene analogues of boron stabilized by neighboring B–B moieties: doubly aromatic bishomotriboriranides. Angew. Chem. Int. Ed. 36, 1469–1472 (1997).
Präsang, C., Hofmann, M., Geiseler, G., Massa, W. & Berndt, A. Aromatic boranes with planar-tetracoordinate boron atoms and very short B–B distances. Angew. Chem. Int. Ed. 41, 1526–1529 (2002).
Maier, A., Hofmann, M., Pritzkow, H. & Siebert, W. A planar, aromatic bicyclo‐tetraborane(4). Angew. Chem. Int. Ed. 41, 1529–1532 (2002).
Takanashi, K., Lee, V. Y. & Sekiguchi, A. Tetrasilacyclobutadiene and cyclobutadiene tricarbonylruthenium complexes: η4-(tBu2MeSi)4Si4]Ru(CO)3 and [η4-(Me3Si)4C4]Ru(CO)3. Organometallics 28, 1248–1251 (2009).
Gleiter, R. Structure and bonding in cyclic sulfur–nitrogen compounds—molecular orbital considerations. Angew. Chem. Int. Ed. 20, 444–452 (1981).
Bucholz, C. F. Versuche über die Auflösung des Indigs in der Schwefelsäure, als Beytrag zur Ausmittelung des Vorgangs bey Verselben. Gehlen’s Neues J. Chem. 3, 3–29 (1804).
Gillespie, R. J. & Passmore, J. Polycations of group VI. Acc. Chem. Res. 4, 413–419 (1971).
Kraus, F., Aschenbrenner, J. C. & Korber, N. P42−: a 6π aromatic polyphosphide in dicesium cyclotetraphosphide–ammonia (1/2). Angew. Chem. Int. Ed. 42, 4030–4033 (2003).
Korber, N. & Reil, M. An isolated cyclo-tetraarsendiide: low temperature synthesis and crystal structure of bis-pentaamminesodium tetraarsendiide–ammonia (1/3) [Na(NH3)5]2As4·3NH3. Chem. Commun. 84–85 (2002); https://doi.org/10.1039/b108879c
Cisar, A. & Corbett, J. D. Polybismuth anions. Synthesis and crystal structure of a salt of the tetrabismuthide(2−) ion, Bi42−. A basis for the interpretation of the structure of some complex intermetallic phases. Inorg. Chem. 16, 2482–2487 (1977).
Critchlow, S. C. & Corbett, J. D. Homopolyatomic anions of the post transition elements. Synthesis and structure of potassium-crypt salts of the tetraantimonide(2−) and heptaantimonide(3−) anions, Sb42− and Sb73−. Inorg. Chem. 23, 770–774 (1984).
Velian, A. & Cummins, C. C. Synthesis and characterization of P2N3−: an aromatic ion composed of phosphorus and nitrogen. Science 348, 1001–1004 (2015).
Scherer, O. J. Complexes with substituent-free acyclic and cyclic phosphorus, arsenic, antimony and bismuth ligands. Angew. Chem. Int. Ed. 29, 1104–1122 (1990).
Li, X.-W., Pennington, W. T. & Robinson, G. H. A metallic system with aromatic character. Synthesis and molecular structure of Na2[(Mes2C6H3)Ga]3 (Mes = 2,4,6-Me3C6H2): the first cyclogallane. J. Am. Chem. Soc. 117, 7578–7579 (1995).
Li, X.-W. et al. Cyclogallanes and metalloaromaticity. Synthesis and molecular structure of dipotassium tris((2,6-dimesitylphenyl)cyclogallene), K2[(Mes2C6H3)Ga]3 (Mes = 2,4,6-Me3C6H2): a structural and theoretical examination. Organometallics 15, 3798–3803 (1996).
Wright, R. J., Brynda, M. & Power, P. P. Synthesis and structure of the ‘dialuminyne’ Na2[Ar′AlAlAr′] and Na2[(Ar″Al)3]: Al−Al bonding in Al2Na2 and Al3Na2 clusters. Angew. Chem. Int. Ed. 45, 5953–5956 (2006).
Li, X. L., Kuznetsov, A. E., Zhang, H.-F., Boldyrev, A. I. & Wang, L.-S. Observation of all-metal aromatic molecules. Science 291, 859–861 (2001).
Kuznetsov, A. E., Boldyrev, A. I., Li, X. & Wang, L.-S. On the aromaticity of square planar Ga42− and In42− in gaseous NaGa4− and NaIn4− clusters. J. Am. Chem. Soc. 123, 8825–8831 (2001).
Boldyrev, A. I. & Wang, L. S. All-metal aromaticity and antiaromaticity. Chem. Rev. 105, 3716–3757 (2005).
Pan, F.-X. et al. An all-metal aromatic sandwich complex [Sb3Au3Sb3]3−. J. Am. Chem. Soc. 137, 10954–10957 (2015).
Twamley, B. & Power, P. P. Synthesis of the square-planar gallium species K2[Ga4(C6H3-2,6-Trip2)2] (Trip = C6H2-2,4,6-iPr3): the role of aryl–alkali metal ion interactions in the structure of gallium clusters. Angew. Chem. Int. Ed. 39, 3500–3503 (2000).
Kuznetsov, A. E., Corbett, J. D., Wang, L. S. & Boldyrev, A. I. Aromatic mercury clusters in ancient amalgams. Angew. Chem. Int. Ed. 40, 3369–3372 (2001).
Gausa, M., Kaschner, R., Lutz, H. O., Seifert, G. & Meiwes-Broer, K.-H. Photoelectron and theoretical investigations on bismuth and antimony pentamer anions: evidence for aromatic structure. Chem. Phys. Lett. 230, 99–102 (1994).
Todorov, I. & Sevov, S. C. Heavy-metal aromatic rings: cyclopentadienyl anion analogues Sn56− and Pb56− in the Zintl phases Na8BaPb6, Na8BaSn6 and Na8EuSn6. Inorg. Chem. 43, 6490–6494 (2004).
Yong, L., Hoffmann, S. D., Fässler, T. F., Riedel, S. & Kaupp, M. [Pb5{Mo(CO)3}2]4−: a complex containing a planar Pb5 unit. Angew. Chem. Int. Ed. 44, 2092–2096 (2005).
Zubarev, D. Y., Averkiev, B. B., Zhai, H.-J., Wang, L.-S. & Boldyrev, A. I. Aromaticity and antiaromaticity in transition-metal systems. Phys. Chem. Chem. Phys. 10, 257–267 (2008).
Kuznetsov, A. E. & Boldyrev, A. I. A single π-bond captures 3, 4 and 5 atoms. Chem. Phys. Lett. 388, 452–456 (2004).
Liu, C., Popov, I. A., Chen, Z., Boldyrev, A. I. & Sun, Z.-M. Aromaticity and antiaromaticity in Zintl clusters. Chem. Eur. J. 24, 14583–14597 (2018).
Huang, X., Zhai, H. ‐J., Kiran, B. & Wang, L. ‐S. Observation of d‐orbital aromaticity. Angew. Chem. Int. Ed. 44, 7251–7254 (2005).
Popov, I. A. et al. Peculiar all-metal σ-aromaticity of the [Au2Sb16]4− anion in the solid. Angew. Chem. Int. Ed. 55, 15344–15346 (2016).
Liu, C. et al. [Co2@Ge16]4−: localized versus delocalized bonding in two isomeric intermetalloid clusters. Chem. Eur. J. 24, 699–705 (2018).
Jones, C. E. Jr. et al. AlnBi clusters: transitions between aromatic and jellium stability. J. Phys. Chem. 112, 13316–13325 (2008).
Wilson, R. J., Lichtenberger, N., Weinert, B. & Dehnen, S. Intermetalloid and heterometallic clusters combining p-block (semi)metals with d- or f-block metals. Chem. Rev. 119, 8506–8554 (2019).
Min, X. et al. All‐metal antiaromaticity in Sb4‐type lanthanocene anions. Angew. Chem. Int. Ed. 55, 5531–5535 (2016).
Lichtenberger, N. et al. Main group metal–actinide magnetic coupling and structural response upon U4+ inclusion into Bi, Tl/Bi or Pb/Bi cages. J. Am. Chem. Soc. 138, 9033–9036 (2016).
Xu, L. & Sevov, S. C. Heteroatomic deltahedral clusters of main-group elements: synthesis and structure of the Zintl ions [In4Bi5]3−, [InBi3]2− and [GaBi3]2−. Inorg. Chem. 39, 5383–5389 (2000).
Turbomole version 7.4.1 2019 and version 7.5 2020 (University of Karlsruhe and Forschungszentrum Karlsruhe GmbH, 1989–2007, TURBOMOLE GmbH since 2007); https://www.turbomole.org
Balasubramani, S. G. et al. TURBOMOLE: modular program suite for ab initio quantum-chemical and condensed-matter simulations. J. Chem. Phys. 152, 184107 (2020).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996); erratum 78, 1396 (1997).
Cao, X., Dolg, M. & Stoll, H. Valence basis sets for relativistic energy consistent small-core actinide pseudopotentials. J. Chem. Phys. 118, 487–496 (2003).
Metz, B., Stoll, H. & Dolg, M. Small-core multiconfiguration-Dirac–Hartree–Fock-adjusted pseudopotentials for post-d main group elements: application to PbH and PbO. J. Chem. Phys. 113, 2563–2569 (2000).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Schäfer, A., Klamt, A., Sattel, D., Lohrenz, J. C. W. & Eckert, F. COSMO Implementation in TURBOMOLE: extension of an efficient quantum chemical code towards liquid systems. Phys. Chem. Chem. Phys. 2, 2187–2193 (2000).
Mulliken, R. S. Electronic population analysis on LCAO–MO molecular wave functions. I. J. Chem. Phys. 23, 1833–1840 (1955).
Boys, S. F. Construction of some molecular orbitals to be approximately invariant for changes from one molecule to another. Rev. Mod. Phys. 32, 296–299 (1960).
Knizia, G. Intrinsic atomic orbitals: an unbiased bridge between quantum theory and chemical concepts. J. Chem. Theory Comput. 9, 4834–4843 (2013).
Becke, A. D. & Edgecombe, K. E. A simple measure of electron localization in atomic and molecular systems. J. Chem. Phys. 92, 5397–5403 (1990).
Jusélius, J., Sundholm, D. & Gauss, J. Calculation of current densities using gauge-including atomic orbitals. J. Chem. Phys. 121, 3952–3963 (2004); https://github.com/qmcurrents/gimic
Peng, D., Middendorf, N., Weigend, F. & Reiher, M. An efficient implementation of two-component relativistic exact-decoupling methods for large molecules. J. Chem. Phys. 138, 184105 (2013).
Franzke, Y. J., Middendorf, N. & Weigend, F. Efficient implementation of one- and two-component analytical energy gradients in exact two-component theory. J. Chem. Phys. 148, 104110 (2018).
Franzke, Y. J. & Weigend, F. NMR shielding tensors and chemical shifts in scalar-relativistic local exact two-component theory. J. Chem. Theory Comput. 15, 1028–1043 (2019).
Franzke, Y. J., Treß, R., Pazdera, T. M. & Weigend, F. Error-consistent segmented contracted all-electron relativistic basis sets of double- and triple-zeta quality for NMR shielding constants. Phys. Chem. Chem. Phys. 21, 16658–16664 (2019).
Franzke, Y. J., Sundholm, D. & Weigend, F. Calculations of current densities and aromatic pathways in cyclic porphyrin and isoporphyrin arrays. Phys. Chem. Chem. Phys. 19, 12794–12803 (2017).
Lichtenberger, N., Spang, N., Eichhöfer, A. & Dehnen, S. Between localization and delocalization: Ru(cod)2+ units in the Zintl clusters [Bi9{Ru(cod)}2]3− and [Tl2Bi6{Ru(cod)}]2−. Angew. Chem. Int. Ed. 56, 13253–13258 (2017).
Ababei, R. et al. Making practical use of the pseudo-element concept: an efficient way to ternary intermetalloid clusters by an isoelectronic Pb−–Bi combination. Chem. Commun. 48, 11295–11297 (2012).
Deubner, H. L., Rudel, S. S. & Kraus, F. A simple access to pure thorium(iv) halides (ThCl4, ThBr4 & ThI4). Z. Anorg. Allg. Chem. 643, 2005–2010 (2017).
Sheldrick, G. M. SHELXT—integrated space-group and crystal-structure determination. Acta Crystallogr. A Found. Adv. 71, 3–8 (2015).
Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. C Struct. Chem. 71, 3–8 (2015).
Reiter, K., Mack, F. & Weigend, F. Calculation of magnetic shielding constants with meta-GGA functionals employing the multipole-accelerated resolution of the identity: implementation and assessment of accuracy and efficiency. J. Chem. Theory Comput. 14, 191–197 (2018).
Acknowledgements
We thank the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG) for financial support within the framework of GRK 1782. We thank J.L. Vasco and M. Pyschik for help with the synthesis, S. Ivlev, B. Weinert, M. Marsch and R. Riedel for help with the diffraction experiments, and M. Hellwig for measuring the EDX spectra of 1. We also thank K. Reiter and F. Dehnen for discussions. N.L. acknowledges a grant from Marburg University Research Academy (MARA). Y.J.F. is grateful to Fonds der Chemischen Industrie for general support of his PhD. studies (Kekulé fellowship), to the German Academic Exchange Service (Deutscher Akademischer Austauschdienst, DAAD) for a fellowship (grant no. 57438025) and to F. Furche for hosting. R.C. acknowledges support from the University of Bordeaux, the CNRS, the Region Nouvelle Aquitaine, the MOLSPIN COST action CA15128 and the GdR MCM-2.
Author information
Authors and Affiliations
Contributions
A.R.E., N.L., R.J.W. and H.L.D. conceived and performed the synthetic experiments, collected single-crystal X-ray crystallographic data, solved and refined the structures, performed ESI-MS and prepared samples for further analyses. R.C. performed and analysed the magnetic measurements. F.W. performed the computational structure optimization and orbital analysis, and Y.J.F. studied the aromaticity and performed the TD-DFT calculations as well as the structure optimizations for Fig. 1. S.D., F.K. and F.W. supervised the work. All authors co-wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary discussion on the formation of compound 2, supplementary information on X-ray diffraction (including Supplementary Table 1 and Supplementary Figs. 1–5), supplementary information on micro-X-ray fluorescence spectroscopy (µ-XFS) (including Supplementary Table 2 and Supplementary Figs. 6–8), supplementary information on electrospray ionization (ESI) mass spectrometry (including Supplementary Figs. 9–11), supplementary information on magnetic measurements of compound 2 (including Supplementary Fig. 12), supplementary details on quantum chemical investigations (including Supplementary Figs. 13–16 and Supplementary Tables 3–12).
Supplementary Data
Crystallographic information file for compound 1.
Supplementary Data
Crystallographic information file for compound 2.
Supplementary Data
Crystallographic information file for compound 3.
Supplementary Data
Crystallographic information file for compound 4.
Supplementary Data
The zip archive comprises two ASCII files entitled ‘Fig01-OptimizedStructures-GIMIC-NICS.txt’ (providing the coordinates of the optimized structures shown in Fig. 1, as well as their NICs values and ring currents) and ‘OptimizedStructures.txt’ (providing all coordinates of the optimized structures of 2A and the compounds mentioned explicitly in the manuscript or the Supplementary Information.pdf file).
Rights and permissions
About this article
Cite this article
Eulenstein, A.R., Franzke, Y.J., Lichtenberger, N. et al. Substantial π-aromaticity in the anionic heavy-metal cluster [Th@Bi12]4−. Nat. Chem. 13, 149–155 (2021). https://doi.org/10.1038/s41557-020-00592-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-00592-z
This article is cited by
-
Thorium(iv)–antimony complexes exhibiting single, double, and triple polar covalent metal–metal bonds
Nature Chemistry (2024)
-
φ-Aromaticity in prismatic {Bi6}-based clusters
Nature Chemistry (2023)
-
Insight into the formation of bismuth-tungsten carbonyl clusters
Communications Chemistry (2023)
-
Open questions on bonding involving lanthanide atoms
Communications Chemistry (2022)
-
[{Th(C8H8)Cl2}3]2− is stable but not aromatic
Nature (2022)