Abstract
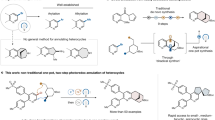
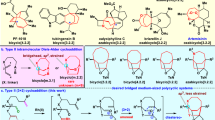
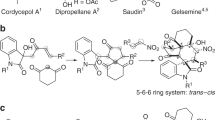
Bridged frameworks are of high chemical and biological significance, being ubiquitous in pharmaceutical molecules and natural products. Specific structures are usually preformed to build these rigid segments at the middle or late stage in the synthesis of polycyclic molecules, resulting in decreased synthetic efficiency and target-specific syntheses. As a logically distinct synthetic strategy, we constructed an allene/ketone-equipped morphan core at the outset through an enantioselective α-allenylation of ketones. Experimental and theoretical results revealed that the high reactivity and enantioselectivity of this reaction are attributed to the cooperative effects of the organocatalyst and metal catalyst. The bridged backbone generated was employed as a structural platform to guide and facilitate the assembly of up to five fusing rings, and the allene and ketone groups thereon were used to precisely install various functionalities at C16 and C20 at the late stage, leading to a concise, collective total synthesis of nine strychnan alkaloids.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All relevant data supporting the findings of this study, including experimental procedures, compound characterizations and theoretical calculations are available within the Article and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2195671 (9b), 2195676 (10h), 2195681 (15) and 2195680 (24). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Atanasov, A. G., Zotchev, S. B., Dirsch, V. M., the International Natural Product Sciences Taskforce, & Supuran, C. T. Natural products in drug discovery: advances and opportunities. Nat. Rev. Drug Discovery 20, 200–216 2021).
Rodrigues, T., Reker, D., Schneider, P. & Schneider, G. Counting on natural products for drug design. Nat. Chem. 8, 531–541 (2016).
Newman, D. J. & Cragg, G. M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 83, 770–803 (2020).
Ricarte, A., Dalton, J. A. R. & Giraldo, J. Structural assessment of agonist efficacy in the μ‑opioid receptor: morphine and fentanyl elicit different activation patterns. J. Chem. Inf. Model. 61, 1251–1274 (2021).
Zhao, S., Sirasani, G. & Andrade, R. B. in The Alkaloids: Chemistry and Biology Vol. 86 (ed. Knölker, H. J.) 1−143 (Academic Press, 2021).
Sirasani, G. & Andrade, R. B. in Strategies and Tactics in Organic Synthesis Vol. 9 (ed. Harmata, M.) Ch. 1 (Academic Press, 2013).
He, W., Wang, P., Chen, J. & Xie, W. Recent progress in the total synthesis of Strychnos alkaloids. Org. Biomol. Chem. 18, 1046–1056 (2020).
Cannon, J. S. & Overman, L. E. Is there no end to the total syntheses of strychnine? Lessons learned in strategy and tactics in total synthesis. Angew. Chem. Int. Ed. 51, 4288–4311 (2012).
Krause, N., Stephen, A. & Hashmi, K. Modern Allene Chemistry (Wiley, 2004).
Yu, S. & Ma, S. Allenes in catalytic asymmetric synthesis and natural product syntheses. Angew. Chem. Int. Ed. 51, 3074–3112 (2012).
Watson, A. & MacMillan, D. in Science of Synthesis: Stereoselective Synthesis 3; Stereoselective Pericyclic Reactions Cross-coupling, and C−H and C−X Activation (ed. Evans, P. A.) 675–745 (Stuttgart, 2011).
Liu, Y. et al. Nickel(II)-catalyzed asymmetric propargyl and allyl claisen rearrangements to allenyl- and allyl-substituted β-ketoesters. Angew. Chem. Int. Ed. 53, 11579–11582 (2014).
Cao, T., Deitch, J., Linton, E. C. & Kozlowski, M. C. Asymmetric synthesis of allenyl oxindoles and spirooxindoles by a catalytic enantioselective Saucy–Marbet Claisen rearrangement. Angew. Chem. Int. Ed. 51, 2448–2451 (2012).
Qian, D., Wu, L., Lin, Z. & Sun, J. Organocatalytic synthesis of chiral tetrasubstituted allenes from racemic propargylic alcohols. Nat. Commun. 8, 567 (2017).
Loui, H. J. & Schneider, C. Cooperative palladium/Brønsted acid catalysis toward the highly enantioselective allenylation of β-keto esters. Org. Lett. 24, 1496–1501 (2022).
J. Zhou, Ed. Multicatalyst System in Asymmetric Catalysis (Wiley, 2014).
Allen, A. E. & Macmillan, D. W. C. Synergistic catalysis: a powerful synthetic strategy for new reaction development. Chem. Sci. 3, 633–658 (2012).
Bonjoch, J., Diaba, F. & Bradshaw, B. Synthesis of 2-azabicyclo[3.3.1]nonanes. Synthesis 7, 993–1018 (2011).
Xu, Y. et al. Organocatalytic enantioselective Conia-ene-type carbocyclization of ynamide cyclohexanones: regiodivergent synthesis of morphans and normorphans. Angew. Chem. Int. Ed. 58, 16252–16259 (2019).
Manzano, R., Datta, S., Paton, R. S. & Dixon, D. J. Enantioselective silver and amine co-catalyzed desymmetrizing cycloisomerization of alkyne-linked cyclohexanones. Angew. Chem. Int. Ed. 56, 5834–5838 (2017).
Liu, R. et al. Palladium/L‑proline-catalyzed enantioselective α‑arylative desymmetrization of cyclohexanones. J. Am. Chem. Soc. 138, 5198–5201 (2016).
Gammack Yamagata, A. D. et al. Enantioselective desymmetrization of prochiral cyclohexanones by organocatalytic intramolecular michael additions to α,β-unsaturated esters. Angew. Chem. Int. Ed. 54, 4899–4903 (2015).
Frisch, M. J. et al. Gaussian 09, Revision D.01 (Gaussian, Inc., 2013).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B. 37, 785–789 (1988).
Zhao, Y. & Truhlar, D. G. The Mo6 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four Mo6-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Hutchings-Goetz, L. S., Yang, C., Fyfe, J. W. B. & Snaddon, T. N. Enantioselective syntheses of Strychnos and Chelidonium alkaloids through regio- and stereocontrolled cooperative catalysis. Angew. Chem. Int. Ed. 59, 17556–17564 (2020).
He, L. et al. Asymmetric total synthesis of (+)-strychnine. Org. Lett. 21, 252–255 (2019).
Lee, G. S., Namkoong, G., Park, J. & Chen, D. Y. Total synthesis of strychnine. Chem. Eur. J. 23, 16189–16193 (2017).
Jones, S. B., Simmons, B., Mastracchio, A. & MacMillan, D. W. C. Collective synthesis of natural products by means of organocascade catalysis. Nature 475, 183–188 (2011).
Feng, L. et al. Reaction of donor-acceptor cyclobutanes with indoles: a general protocol for the formal total synthesis of (±)-strychnine and the total synthesis of (±)-akuammicine. Angew. Chem. Int. Ed. 56, 3055–3058 (2017).
Sirasani, G., Paul, T., Dougherty, W., Kassel, S. & Andrade, R. B. Concise total syntheses of (±)-strychnine and (±)-akuammicine. J. Org. Chem. 75, 3529–3532 (2010).
Mori, M., Nakanishi, M., Kajishima, D. & Sato, Y. A novel and general synthetic pathway to Strychnos indole alkaloids: total syntheses of (–)-tubifoline, (–)-dehydrotubifoline, and (–)-strychnine using palladium-catalyzed asymmetric allylic substitution. J. Am. Chem. Soc. 125, 9801–9807 (2003).
Ito, M., Clark, C. W., Mortimore, M. J., Goh, B. & Martin, S. F. Biogenetically inspired approach to the Strychnos alkaloids. Concise syntheses of (±)-akuammicine and (±)-strychnine. J. Am. Chem. Soc. 123, 8003–8010 (2001).
Knight, S. D., Overman, L. E. & Pairaudeau, G. Asymmetric total syntheses of (–)- and (+)-strychnine and the Wieland–Gumlich aldehyde. J. Am. Chem. Soc. 117, 5776–5788 (1995).
Rawal, V. H. & Iwasa, S. Short, stereocontrolled synthesis of strychnine. J. Org. Chem. 59, 2685–2686 (1994).
Woodward, R. B. et al. The total synthesis of strychnine. J. Am. Chem. Soc. 76, 4749–4751 (1954).
Mbeunkui, F. et al. In vitro antiplasmodial activity of indole alkaloids from the stem bark of Geissospermum vellosii. J. Ethnopharmacol. 139, 471–477 (2012).
Maertens, G., Deruer, E., Denis, M. & Canesi, S. Common strategy for the synthesis of some Strychnos indole alkaloids. J. Org. Chem. 85, 6098–6108 (2020).
Maertens, G. & Canesi, S. Synthesis of the Strychnos alkaloid (−)-strychnopivotine and confirmation of its absolute configuration. Chem. Eur. J. 22, 7090–7093 (2016).
Boonsombat, J., Zhang, H., Chughtai, M. J., Hartung, J. & Padwa, A. A general synthetic entry to the pentacyclic Strychnos alkaloid family, using a [4+2]-cycloaddition/rearrangement cascade sequence. J. Org. Chem. 73, 3539–3550 (2008).
He, W. et al. Highly enantioselective tandem Michael addition of tryptamine-derived oxindoles to alkynones: concise synthesis of Strychnos alkaloids. Angew. Chem. Int. Ed. 57, 3806–3809 (2018).
Hong, A. Y. & Vanderwal, C. D. A synthesis of alsmaphorazine B demonstrates the chemical feasibility of a new biogenetic hypothesis. J. Am. Chem. Soc. 137, 7306–7309 (2015).
Bonjoch, J., Sole, D. & Bosch, J. Studies on the synthesis of Strychnos indole alkaloids. Synthesis of (±)-dehydrotubifoline. J. Am. Chem. Soc. 117, 11017–11018 (1995).
Rawal, V. H., Michoud, C. & Monestel, R. General strategy for the stereocontrolled synthesis of Strychnos alkaloids: a concise synthesis of (±)-dehydrotubifoline. J. Am. Chem. Soc. 115, 3030–3031 (1993).
Fevig, J. M., Marquis, R. W. & Overman, L. E. New approach to Strychnos alkaloids. Stereocontrolled total synthesis of (±)-dehydrotubifoline. J. Am. Chem. Soc. 113, 5085–5086 (1991).
Teijaro, C. N. et al. Synthesis and biological evaluation of pentacyclic Strychnos alkaloids as selective modulators of the ABCC10 (MRP7) efflux pump. J. Med. Chem. 57, 10383–10390 (2014).
Yuan, W. & Ma, S. Ligand controlled highly selective copper-catalyzed borylcuprations of allenes with bis(pinacolato)diboron. Adv. Synth. Catal. 354, 1867–1872 (2012).
Anet, F. A. L. & Robinson, R. Conversion of the Wieland–Gumlich aldehyde into strychnine. Chem. Ind. 245–245 (1953).
Bestmann, H. J. & Sandmeier, D. Simple synthesis of ketenylidenetriphenylphosphorane and its thioanalogs. Angew. Chem. Int. Ed. 14, 634–634 (1975).
Kokkonda, P. et al. Biomimetic total syntheses of (–)-leucoridines A and C through the dimerization of (–)-dihydrovalparicine. Angew. Chem. Int. Ed. 54, 12632–12635 (2015).
Yang, Z. et al. Asymmetric total synthesis of sarpagine and koumine alkaloids. Angew. Chem. Int. Ed. 60, 13105–13111 (2021).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21922102, 21871033, 22271033), the Fundamental Research Funds for the Central Universities (2022CDJQY-001, 2020CDJQY-Z002) and Chongqing Science and Technology Commission (CSTB2022NSCQLZX0036) to M.Z. The numerical computations were performed at Hefei advanced computing centre. We are grateful to X. Gong from Analytical and Testing Center of Chongqing University for X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
M.Z. and Y.L. conceived and directed the project. W.Z. and S.X. performed the experiments and analysed the experimental data with the help of D.J., J.Y. and S.L. Y.L. and H.C. conducted the theoretical studies. M.Z. and Y.L. wrote the manuscript with W.Z., S.X., H.C., H.Q. and L.H. W.Z., S.X. and H.C. prepared the Supplementary Information. All authors discussed the results and gave their approval of the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Christoph Schneider, Sylvain Canesi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–23, Figs. 1 and 2, experimental data, synthesis and characterization data, NMR spectra, X-ray crystallographic data and density functional theory calculation data.
Supplementary Data 1
Crystallographic data for compound 10h; CCDC reference 2195676.
Supplementary Data 2
Crystallographic data for compound 9b; CCDC reference 2195671.
Supplementary Data 3
Crystallographic data for compound 15; CCDC reference 2195681.
Supplementary Data 4
Crystallographic data for compound 24; CCDC reference 2195680.
Supplementary Data 5
Structure factors for compound 10h; CCDC reference 2195676.
Supplementary Data 6
Structure factors for compound 9b; CCDC reference 2195671.
Supplementary Data 7
Structure factors for compound 15; CCDC reference 2195681.
Supplementary Data 8
Structure factors for compound 24; CCDC reference 2195680.
Supplementary Data 9
Cartesian coordinates for all optimized structures.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, W., Xi, S., Chen, H. et al. A bridged backbone strategy enables collective synthesis of strychnan alkaloids. Nat. Chem. 15, 1074–1082 (2023). https://doi.org/10.1038/s41557-023-01264-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01264-4
This article is cited by
-
Total synthesis of strychnan alkaloids
Nature Chemistry (2023)